Abstract
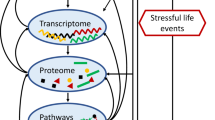
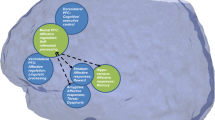
Unravelling the pathophysiology of depression is a unique challenge. Not only are depressive syndromes heterogeneous and their aetiologies diverse, but symptoms such as guilt and suicidality are impossible to reproduce in animal models. Nevertheless, other symptoms have been accurately modelled, and these, together with clinical data, are providing insight into the neurobiology of depression. Recent studies combining behavioural, molecular and electrophysiological techniques reveal that certain aspects of depression result from maladaptive stress-induced neuroplastic changes in specific neural circuits. They also show that understanding the mechanisms of resilience to stress offers a crucial new dimension for the development of fundamentally novel antidepressant treatments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kessler, R. C. et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry 62, 593–602 (2005).
Nestler, E. J. et al. Neurobiology of depression. Neuron 34, 13–25 (2002).
Knol, M. J. et al. Depression as a risk factor for the onset of type 2 diabetes mellitus. A meta-analysis. Diabetologia 49, 837–845 (2006).
Evans, D. L. et al. Mood disorders in the medically ill: scientific review and recommendations. Biol. Psychiatry 58, 175–189 (2005).
Gildengers, A. G. et al. Medical burden in late-life bipolar and major depressive disorders. Am. J. Geriatr. Psychiatry 16, 194–200 (2008).
Phelps, E. A. & LeDoux, J. E. Contributions of the amygdala to emotion processing: from animal models to human behavior. Neuron 48, 175–187 (2005).
Drevets, W. C. Neuroimaging and neuropathological studies of depression: implications for the cognitive-emotional features of mood disorders. Curr. Opin. Neurobiol. 11, 240–249 (2001).
Lopez-Leon, S. et al. Meta-analyses of genetic studies on major depressive disorder. Mol. Psychiatry 13, 772–785 (2007).
Rush, A. J. The varied clinical presentations of major depressive disorder. J. Clin. Psychiatry 68 (Suppl. 8), 4–10 (2007).
Kendler, K. S., Karkowski, L. M. & Prescott, C. A. Causal relationship between stressful life events and the onset of major depression. Am. J. Psychiatry 156, 837–841 (1999).
Mill, J. & Petronis, A. Molecular studies of major depressive disorder: the epigenetic perspective. Mol. Psychiatry 12, 799–814 (2007).
Hasler, G., Drevets, W. C., Manji, H. K. & Charney, D. S. Discovering endophenotypes for major depression. Neuropsychopharmacology 29, 1765–1781 (2004).
Ressler, K. J. & Mayberg, H. S. Targeting abnormal neural circuits in mood and anxiety disorders: from the laboratory to the clinic. Nature Neurosci. 10, 1116–1124 (2007).
Berton, O. & Nestler, E. J. New approaches to antidepressant drug discovery: beyond monoamines. Nature Rev. Neurosci. 7, 137–151 (2006).
Sheline, Y. I. Neuroimaging studies of mood disorder effects on the brain. Biol. Psychiatry 54, 338–352 (2003).
Harrison, P. J. The neuropathology of primary mood disorder. Brain 125, 1428–1449 (2002).
Mayberg, H. S. et al. Deep brain stimulation for treatment-resistant depression. Neuron 45, 651–660 (2005). This paper gives the first demonstration of the therapeutic efficacy of deep brain stimulation applied to the subgenual cingulate gyrus for treatment-refractory depression.
Schlaepfer, T. E. et al. Deep brain stimulation to reward circuitry alleviates anhedonia in refractory major depression. Neuropsychopharmacology 33, 368–377 (2008).
Nestler, E. J. & Carlezon, W. A. Jr . The mesolimbic dopamine reward circuit in depression. Biol. Psychiatry 59, 1151–1159 (2006).
Pittenger, C. & Duman, R. S. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology 33, 88–109 (2008).
Trivedi, M. H. et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am. J. Psychiatry 163, 28–40 (2006).
Ansorge, M. S., Hen, R. & Gingrich, J. A. Neurodevelopmental origins of depressive disorders. Curr. Opin. Pharmacol. 7, 8–17 (2007).
Ruhe, H. G., Mason, N. S. & Schene, A. H. Mood is indirectly related to serotonin, norepinephrine and dopamine levels in humans: a meta-analysis of monoamine depletion studies. Mol. Psychiatry 12, 331–359 (2007).
Hu, H. et al. Emotion enhances learning via norepinephrine regulation of AMPA-receptor trafficking. Cell 131, 160–173 (2007).
Krishnan, V. et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 131, 391–404 (2007).This paper adapts the social-defeat model to study resilient behaviour and identifies active neurobiological mechanisms that maintain normal functioning in the face of chronic stress.
Svenningsson, P. et al. Alterations in 5-HT1B receptor function by p11 in depression-like states. Science 311, 77–80 (2006).
Mathew, S. J., Manji, H. K. & Charney, D. S. Novel drugs and therapeutic targets for severe mood disorders. Neuropsychopharmacology 33, 2080–2092 (2008). This paper provides an up-to-date and comprehensive list of new antidepressant drugs currently in various stages of clinical trials.
Lucas, G. et al. Serotonin4 (5-HT4) receptor agonists are putative antidepressants with a rapid onset of action. Neuron 55, 712–725 (2007).
Uhr, M. et al. Polymorphisms in the drug transporter gene ABCB1 predict antidepressant treatment response in depression. Neuron 57, 203–209 (2008).
Holsboer, F. How can we realize the promise of personalized antidepressant medicines? Nature Rev. Neurosci. 9, 638–646 (2008).
Duman, R. S. & Monteggia, L. M. A neurotrophic model for stress-related mood disorders. Biol. Psychiatry 59, 1116–1127 (2006).
Monteggia, L. M. et al. Essential role of brain-derived neurotrophic factor in adult hippocampal function. Proc. Natl Acad. Sci. USA 101, 10827–10832 (2004).
Karege, F., Vaudan, G., Schwald, M., Perroud, N. & La Harpe, R. Neurotrophin levels in postmortem brains of suicide victims and the effects of antemortem diagnosis and psychotropic drugs. Brain Res. Mol. Brain Res. 136, 29–37 (2005).
Shirayama, Y., Chen, A. C., Nakagawa, S., Russell, D. S. & Duman, R. S. Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J. Neurosci. 22, 3251–3261 (2002).
Monteggia, L. M. et al. Brain-derived neurotrophic factor conditional knockouts show gender differences in depression-related behaviors. Biol. Psychiatry 61, 187–197 (2007).
Groves, J. O. Is it time to reassess the BDNF hypothesis of depression? Mol. Psychiatry 12, 1079–1088 (2007).
Martinowich, K., Manji, H. & Lu, B. New insights into BDNF function in depression and anxiety. Nature Neurosci. 10, 1089–1093 (2007).
Zorner, B. et al. Forebrain-specific trkB-receptor knockout mice: behaviorally more hyperactive than 'depressive'. Biol. Psychiatry 54, 972–982 (2003).
Berton, O. et al. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science 311, 864–868 (2006). By using the social-defeat model, this paper characterizes the potent, pro-depressant effects of BDNF in the mesolimbic dopamine pathway, which is opposite to the well-described antidepressant-like effects of BDNF in hippocampal circuits.
Eisch, A. J. et al. Brain–derived neurotrophic factor in the ventral midbrain-nucleus accumbens pathway: a role in depression. Biol. Psychiatry 54, 994–1005 (2003).
Chen, Z. Y. et al. Genetic variant BDNF (Val66Met) polymorphism alters anxiety-related behavior. Science 314, 140–143 (2006).
Egan, M. F. et al. The BDNF Val66Met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112, 257–269 (2003). This multidisciplinary study demonstrates how the polymorphism in BDNF in which methionine is substituted for valine at position 66 causes deficits in episodic memory, alters hippocampal activation and decreases activity-dependent BDNF secretion.
Szeszko, P. R. et al. Brain-derived neurotrophic factor Val66Met polymorphism and volume of the hippocampal formation. Mol. Psychiatry 10, 631–636 (2005).
Gratacos, M. et al. Brain-derived neurotrophic factor Val66Met and psychiatric disorders: meta-analysis of case-control studies confirm association to substance-related disorders, eating disorders, and schizophrenia. Biol. Psychiatry 61, 911–922 (2007).
Kaufman, J. et al. Brain-derived neurotrophic factor-5–HTTLPR gene interactions and environmental modifiers of depression in children. Biol. Psychiatry 59, 673–680 (2006).
Kim, J. M. et al. Interactions between life stressors and susceptibility genes (5-HTTLPR and BDNF) on depression in Korean elders. Biol. Psychiatry 62, 423–428 (2007).
Pezawas, L. et al. Evidence of biologic epistasis between BDNF and SLC6A4 and implications for depression. Mol. Psychiatry 13, 709–716 (2008).
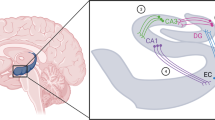
Sahay, A. & Hen, R. Adult hippocampal neurogenesis in depression. Nature Neurosci. 10, 1110–1115 (2007).
Sairanen, M., Lucas, G., Ernfors, P., Castren, M. & Castren, E. Brain-derived neurotrophic factor and antidepressant drugs have different but coordinated effects on neuronal turnover, proliferation, and survival in the adult dentate gyrus. J. Neurosci. 25, 1089–1094 (2005).
Hunsberger, J. G. et al. Antidepressant actions of the exercise-regulated gene VGF. Nature Med. 13, 1476–1482 (2007). This paper characterizes the antidepressant effects of VGF, an exercise-regulated neurotrophic factor in the hippocampus, and identifies VGF-mediated signalling as a potential therapeutic target.
Thakker-Varia, S. et al. The neuropeptide VGF produces antidepressant-like behavioral effects and enhances proliferation in the hippocampus. J. Neurosci. 27, 12156–12167 (2007).
Warner-Schmidt, J. L. & Duman, R. S. VEGF is an essential mediator of the neurogenic and behavioral actions of antidepressants. Proc. Natl Acad. Sci. USA 104, 4647–4652 (2007).
Airan, R. D. et al. High-speed imaging reveals neurophysiological links to behavior in an animal model of depression. Science 317, 819–823 (2007). This study uses quantitative voltage-sensitive dye imaging to explore the contribution of antidepressant-induced neurogenesis to local hippocampal network dynamics.
Kempermann, G. The neurogenic reserve hypothesis: what is adult hippocampal neurogenesis good for? Trends Neurosci. 31, 163–169 (2008).
Surget, A. et al. Drug-dependent requirement of hippocampal neurogenesis in a model of depression and of antidepressant reversal. Biol. Psychiatry 64, 293–301 (2008).
Santarelli, L. et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 301, 805–809 (2003).
Zhao, C., Deng, W. & Gage, F. H. Mechanisms and functional implications of adult neurogenesis. Cell 132, 645–660 (2008).
Parker, K. J., Schatzberg, A. F. & Lyons, D. M. Neuroendocrine aspects of hypercortisolism in major depression. Horm. Behav. 43, 60–66 (2003).
Raison, C. L. & Miller, A. H. When not enough is too much: the role of insufficient glucocorticoid signaling in the pathophysiology of stress-related disorders. Am. J. Psychiatry 160, 1554–1565 (2003).
Gourley, S. L. et al. Regionally specific regulation of ERK MAP kinase in a model of antidepressant-sensitive chronic depression. Biol. Psychiatry 63, 353–359 (2007).
McEwen, B. S. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol. Rev. 87, 873–904 (2007).
Brown, E. S., Varghese, F. P. & McEwen, B. S. Association of depression with medical illness: does cortisol play a role? Biol. Psychiatry 55, 1–9 (2004).
Nemeroff, C. B. & Owens, M. J. Treatment of mood disorders. Nature Neurosci. 5 (Suppl.), 1068–1070 (2002).
de Kloet, E. R., Joels, M. & Holsboer, F. Stress and the brain: from adaptation to disease. Nature Rev. Neurosci. 6, 463–475 (2005).
Brouwer, J. P. et al. Thyroid and adrenal axis in major depression: a controlled study in outpatients. Eur. J. Endocrinol. 152, 185–191 (2005).
Schatzberg, A. F. & Lindley, S. Glucocorticoid antagonists in neuropsychotic disorders. Eur. J. Pharmacol. 583, 358–364 (2008).
Gold, P. W. & Chrousos, G. P. Organization of the stress system and its dysregulation in melancholic and atypical depression: high vs low CRH/NE states. Mol. Psychiatry 7, 254–275 (2002).
Heim, C., Ehlert, U. & Hellhammer, D. H. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology 25, 1–35 (2000).
Dantzer, R., O'Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nature Rev. Neurosci. 9, 46–56 (2008).
Dunn, A. J., Swiergiel, A. H. & de Beaurepaire, R. Cytokines as mediators of depression: what can we learn from animal studies? Neurosci. Biobehav. Rev. 29, 891–909 (2005).
Loftis, J. M. & Hauser, P. The phenomenology and treatment of interferon-induced depression. J. Affect. Disord. 82, 175–190 (2004).
Chourbaji, S. et al. IL-6 knockout mice exhibit resistance to stress-induced development of depression-like behaviors. Neurobiol. Dis. 23, 587–594 (2006).
Simen, B. B., Duman, C. H., Simen, A. A. & Duman, R. S. TNFα signaling in depression and anxiety: behavioral consequences of individual receptor targeting. Biol. Psychiatry 59, 775–785 (2006).
Koo, J. W. & Duman, R. S. IL-1β is an essential mediator of the antineurogenic and anhedonic effects of stress. Proc. Natl Acad. Sci. USA 105, 751–756 (2008).
Tsankova, N., Renthal, W., Kumar, A. & Nestler, E. J. Epigenetic regulation in psychiatric disorders. Nature Rev. Neurosci. 8, 355–367 (2007).
Szyf, M., Weaver, I. & Meaney, M. Maternal care, the epigenome and phenotypic differences in behavior. Reprod. Toxicol. 24, 9–19 (2007).
Weaver, I. C. et al. Epigenetic programming by maternal behavior. Nature Neurosci. 7, 847–854 (2004).
Tsankova, N. M., Kumar, A. & Nestler, E. J. Histone modifications at gene promoter regions in rat hippocampus after acute and chronic electroconvulsive seizures. J. Neurosci. 24, 5603–5610 (2004).
Tsankova, N. M. et al. Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nature Neurosci. 9, 519–525 (2006). This paper contains one of the first demonstrations of the role of epigenetic modifications in stress-induced effects on the brain and their reversal by antidepressant treatments, as well as the therapeutic potential of HDAC inhibitors in depression.
Schroeder, F. A., Lin, C. L., Crusio, W. E. & Akbarian, S. Antidepressant-like effects of the histone deacetylase inhibitor, sodium butyrate, in the mouse. Biol. Psychiatry 62, 55–64 (2007).
Renthal, W. et al. Histone deacetylase 5 epigenetically controls behavioral adaptations to chronic emotional stimuli. Neuron 56, 517–529 (2007).
Yehuda, R. Risk and resilience in posttraumatic stress disorder. J. Clin. Psychiatry 65 (Suppl. 1), 29–36 (2004).
Charney, D. S. Psychobiological mechanisms of resilience and vulnerability: implications for successful adaptation to extreme stress. Am. J. Psychiatry 161, 195–216 (2004).
Berton, O. et al. Induction of ΔFosB in the periaqueductal gray by stress promotes active coping responses. Neuron 55, 289–300 (2007). In this paper, the learned-helplessness model is used to illustrate the pro-resilient effects of the transcription factor ΔFOSB within the dorsal raphe nucleus.
Strekalova, T., Spanagel, R., Bartsch, D., Henn, F. A. & Gass, P. Stress-induced anhedonia in mice is associated with deficits in forced swimming and exploration. Neuropsychopharmacology 29, 2007–2017 (2004).
Sajdyk, T. J. et al. Neuropeptide Y in the amygdala induces long-term resilience to stress-induced reductions in social responses but not hypothalamic–adrenal–pituitary axis activity or hyperthermia. J. Neurosci. 28, 893–903 (2008).
Peaston, A. E. & Whitelaw, E. Epigenetics and phenotypic variation in mammals. Mamm. Genome 17, 365–374 (2006).
Bergstrom, A., Jayatissa, M. N., Thykjaer, T. & Wiborg, O. Molecular pathways associated with stress resilience and drug resistance in the chronic mild stress rat model of depression: a gene expression study. J. Mol. Neurosci. 33, 201–215 (2007).
Zarate, C. A. Jr et al. A randomized trial of an N-methyl-d-aspartate antagonist in treatment-resistant major depression. Arch. Gen. Psychiatry 63, 856–864 (2006).
Maeng, S. et al. Cellular mechanisms underlying the antidepressant effects of ketamine: role of α-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors. Biol. Psychiatry 63, 349–352 (2008).
Garcia, L. S. et al. Acute administration of ketamine induces antidepressant-like effects in the forced swimming test and increases BDNF levels in the rat hippocampus. Prog. Neuropsychopharmacol. Biol. Psychiatry 32, 140–144 (2008).
Maeng, S. & Zarate, C. A. Jr . The role of glutamate in mood disorders: results from the ketamine in major depression study and the presumed cellular mechanism underlying its antidepressant effects. Curr. Psychiatry Rep. 9, 467–474 (2007).
Roy, M., David, N., Cueva, M. & Giorgetti, M. A study of the involvement of melanin-concentrating hormone receptor 1 (MCHR1) in murine models of depression. Biol. Psychiatry 61, 174–180 (2007).
Georgescu, D. et al. The hypothalamic neuropeptide melanin-concentrating hormone acts in the nucleus accumbens to modulate feeding behavior and forced-swim performance. J. Neurosci. 25, 2933–2940 (2005).
Lutter, M. et al. The orexigenic hormone ghrelin defends against depressive symptoms of chronic stress. Nature Neurosci. 11, 752–753 (2008).
Lu, X. Y., Kim, C. S., Frazer, A. & Zhang, W. Leptin: a potential novel antidepressant. Proc. Natl Acad. Sci. USA 103, 1593–1598 (2006).
Kishi, T. & Elmquist, J. K. Body weight is regulated by the brain: a link between feeding and emotion. Mol. Psychiatry 10, 132–146 (2005).
Kaplitt, M. G. et al. Safety and tolerability of gene therapy with an adeno-associated virus (AAV) borne GAD gene for Parkinson's disease: an open label, phase I trial. Lancet 369, 2097–2105 (2007). This paper demonstrates the efficacy and safety of stereotactic viral-mediated gene therapy in the treatment of severe Parkinson's disease.
Cryan, J. F., Markou, A. & Lucki, I. Assessing antidepressant activity in rodents: recent developments and future needs. Trends Pharmacol. Sci. 23, 238–245 (2002).
Acknowledgements
Work in E.J.N.'s laboratory was supported by grants from the National Institute of Mental Health.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
E.J.N. receives income for consulting for Merck Research Laboratories, Neurologix, and PsychoGenics, as well as research support from AstraZeneca.
Additional information
Reprints and permissions information is available at http://www.nature.com/reprints.
Correspondence should be addressed to E.J.N. (eric.nestler@mssm.edu).
Rights and permissions
About this article
Cite this article
Krishnan, V., Nestler, E. The molecular neurobiology of depression. Nature 455, 894–902 (2008). https://doi.org/10.1038/nature07455
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07455
This article is cited by
-
Comparing the effectiveness of transdiagnostic treatment with acceptance and commitment therapy on emotional disorders, rumination, and life satisfaction in patients with irritable bowel syndrome: a randomized clinical trial
BMC Gastroenterology (2024)
-
Advancing preclinical chronic stress models to promote therapeutic discovery for human stress disorders
Neuropsychopharmacology (2024)
-
Integrating Schema Therapy with Kleinman's Cultural Explanatory Model: A Case Study
Psychological Studies (2024)
-
Hypothalamic Gene Expression in a Rat Model of Chronic Unpredictable Mild Stress Treated with Electroacupuncture
Neurochemical Research (2024)
-
Simvastatin ameliorates synaptic plasticity impairment in chronic mild stress-induced depressed mice by modulating hippocampal NMDA receptor
Psychopharmacology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.