Abstract
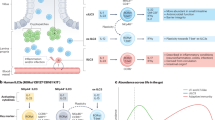
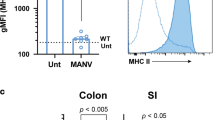
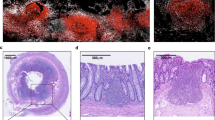
Intestinal homeostasis is critical for efficient energy extraction from food and protection from pathogens. Its disruption can lead to an array of severe illnesses with major impacts on public health, such as inflammatory bowel disease characterized by self-destructive intestinal immunity. However, the mechanisms regulating the equilibrium between the large bacterial flora and the immune system remain unclear. Intestinal lymphoid tissues generate flora-reactive IgA-producing B cells, and include Peyer's patches and mesenteric lymph nodes, as well as numerous isolated lymphoid follicles (ILFs)1,2. Here we show that peptidoglycan from Gram-negative bacteria is necessary and sufficient to induce the genesis of ILFs in mice through recognition by the NOD1 (nucleotide-binding oligomerization domain containing 1) innate receptor in epithelial cells, and β-defensin 3- and CCL20-mediated signalling through the chemokine receptor CCR6. Maturation of ILFs into large B-cell clusters requires subsequent detection of bacteria by toll-like receptors. In the absence of ILFs, the composition of the intestinal bacterial community is profoundly altered. Our results demonstrate that intestinal bacterial commensals and the immune system communicate through an innate detection system to generate adaptive lymphoid tissues and maintain intestinal homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hamada, H. et al. Identification of multiple isolated lymphoid follicles on the antimesenteric wall of the mouse small intestine. J. Immunol. 168, 57–64 (2002)
Tsuji, M. et al. Requirement for lymphoid tissue-inducer cells in isolated follicle formation and T cell-independent immunoglobulin A generation in the gut. Immunity 29, 261–271 (2008)
Mebius, R. E. Organogenesis of lymphoid tissues. Nature Rev. Immunol. 3, 292–303 (2003)
Kanamori, Y. et al. Identification of novel lymphoid tissues in murine intestinal mucosa where clusters of c-kit+ IL-7R+ Thy1+ lympho-hemopoietic progenitors develop. J. Exp. Med. 184, 1449–1459 (1996)
Pabst, O. et al. Cryptopatches and isolated lymphoid follicles: Dynamic lymphoid tissues dispensable for the generation of intraepithelial lymphocytes. Eur. J. Immunol. 35, 98–107 (2005)
Eberl, G. & Littman, D. R. Thymic origin of intestinal αβ T cells revealed by fate mapping of RORγt+ cells. Science 305, 248–251 (2004)
Eberl, G. Inducible lymphoid tissues in the adult gut: Recapitulation of a fetal developmental pathway? Nature Rev. Immunol. 5, 413–420 (2005)
Pabst, O. et al. Adaptation of solitary intestinal lymphoid tissue in response to microbiota and chemokine receptor CCR7 signaling. J. Immunol. 177, 6824–6832 (2006)
Fagarasan, S. et al. Critical roles of activation-induced cytidine deaminase in the homeostasis of gut flora. Science 298, 1424–1427 (2002)
Eberl, G. et al. An essential function for the nuclear receptor RORγt in the generation of fetal lymphoid tissue inducer cells. Nature Immunol. 5, 64–73 (2004)
Lorenz, R. G., Chaplin, D. D., McDonald, K. G., McDonough, J. S. & Newberry, R. D. Isolated lymphoid follicle formation is inducible and dependent upon lymphotoxin-sufficient B lymphocytes, lymphotoxin β receptor, and TNF receptor I function. J. Immunol. 170, 5475–5482 (2003)
Rennert, P. D., Browning, J. L., Mebius, R., Mackay, F. & Hochman, P. S. Surface lymphotoxin α/β complex is required for the development of peripheral lymphoid organs. J. Exp. Med. 184, 1999–2006 (1996)
Dewhirst, F. E. et al. Phylogeny of the defined murine microbiota: Altered Schaedler flora. Appl. Environ. Microbiol. 65, 3287–3292 (1999)
Fritz, J. H., Ferrero, R. L., Philpott, D. J. & Girardin, S. E. Nod-like proteins in immunity, inflammation and disease. Nature Immunol. 7, 1250–1257 (2006)
Magalhaes, J. G. et al. Murine Nod1 but not its human orthologue mediates innate immune detection of tracheal cytotoxin. EMBO Rep. 6, 1201–1207 (2005)
Koropatnick, T. A. et al. Microbial factor-mediated development in a host-bacterial mutualism. Science 306, 1186–1188 (2004)
Lugering, A. & Kucharzik, T. Induction of intestinal lymphoid tissue: The role of cryptopatches. Ann. NY Acad. Sci. 1072, 210–217 (2006)
McDonald, K. G. et al. CC chemokine receptor 6 expression by B lymphocytes is essential for the development of isolated lymphoid follicles. Am. J. Pathol. 170, 1229–1240 (2007)
Tanaka, Y. et al. Selective expression of liver and activation-regulated chemokine (LARC) in intestinal epithelium in mice and humans. Eur. J. Immunol. 29, 633–642 (1999)
Yang, D. et al. Beta-defensins: Linking innate and adaptive immunity through dendritic and T cell CCR6. Science 286, 525–528 (1999)
Bals, R. et al. Mouse beta-defensin 3 is an inducible antimicrobial peptide expressed in the epithelia of multiple organs. Infect. Immun. 67, 3542–3547 (1999)
Voss, E. et al. NOD2/CARD15 mediates induction of the antimicrobial peptide human beta-defensin-2. J. Biol. Chem. 281, 2005–2011 (2006)
Uehara, A., Fujimoto, Y., Fukase, K. & Takada, H. Various human epithelial cells express functional Toll-like receptors, NOD1 and NOD2 to produce anti-microbial peptides, but not proinflammatory cytokines. Mol. Immunol. 44, 3100–3111 (2007)
Boughan, P. K. et al. Nucleotide-binding oligomerization domain-1 and epidermal growth factor receptor: Critical regulators of beta-defensins during Helicobacter pylori infection. J. Biol. Chem. 281, 11637–11648 (2006)
Wehkamp, J. et al. Inducible and constitutive beta-defensins are differentially expressed in Crohn's disease and ulcerative colitis. Inflamm. Bowel Dis. 9, 215–223 (2003)
Talham, G. L., Jiang, H. Q., Bos, N. A. & Cebra, J. J. Segmented filamentous bacteria are potent stimuli of a physiologically normal state of the murine gut mucosal immune system. Infect. Immun. 67, 1992–2000 (1999)
Schroder, J. M. & Harder, J. Human β-defensin-2. Int. J. Biochem. Cell Biol. 31, 645–651 (1999)
Fritz, J. H. et al. Nod1-mediated innate immune recognition of peptidoglycan contributes to the onset of adaptive immunity. Immunity 26, 445–459 (2007)
Wehkamp, J. et al. NF-kappaB- and AP-1-mediated induction of human beta defensin-2 in intestinal epithelial cells by Escherichia coli Nissle 1917: A novel effect of a probiotic bacterium. Infect. Immun. 72, 5750–5758 (2004)
Mazmanian, S. K., Round, J. L. & Kasper, D. L. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature 453, 620–625 (2008)
Lochner, M. et al. In vivo equilibrium of proinflammatory IL-17+ and regulatory IL-10+ Foxp3+ RORγt+ T cells. J. Exp. Med. 205, 1381–1393 (2008)
Girardin, S. E. et al. Nod1 detects a unique muropeptide from gram-negative bacterial peptidoglycan. Science 300, 1584–1587 (2003)
Antignac, A. et al. Detailed structural analysis of the peptidoglycan of the human pathogen Neisseria meningitidis . J. Biol. Chem. 278, 31521–31528 (2003)
Schaedler, R. W. & Dubos, R. J. The fecal flora of various strains of mice. Its bearing on their susceptibility to endotoxin. J. Exp. Med. 115, 1149–1160 (1962)
Heidrich, C., Ursinus, A., Berger, J., Schwarz, H. & Holtje, J. V. Effects of multiple deletions of murein hydrolases on viability, septum cleavage, and sensitivity to large toxic molecules in Escherichia coli . J. Bacteriol. 184, 6093–6099 (2002)
Godon, J. J., Zumstein, E., Dabert, P., Habouzit, F. & Moletta, R. Molecular microbial diversity of an anaerobic digestor as determined by small-subunit rDNA sequence analysis. Appl. Environ. Microbiol. 63, 2802–2813 (1997)
Barman, M. et al. Enteric salmonellosis disrupts the microbial ecology of the murine gastrointestinal tract. Infect. Immun. 76, 907–915 (2008)
Sarma-Rupavtarm, R. B., Ge, Z., Schauer, D. B., Fox, J. G. & Polz, M. F. Spatial distribution and stability of the eight microbial species of the altered Schaedler flora in the mouse gastrointestinal tract. Appl. Environ. Microbiol. 70, 2791–2800 (2004)
Ge, Z. et al. Colonization dynamics of altered Schaedler flora is influenced by gender, aging, and Helicobacter hepaticus infection in the intestines of Swiss Webster mice. Appl. Environ. Microbiol. 72, 5100–5103 (2006)
Rivas-Santiago, B. et al. β-Defensin gene expression during the course of experimental tuberculosis infection. J. Infect. Dis. 194, 697–701 (2006)
Acknowledgements
We thank G. Milon, N. Cerf-Bensussan, P. Sansonetti, P. Cossart, A. Phalipon, D. Philpott, G. Marchal and D. Guy-Grand for discussions and critical reading of the manuscript; N. Huntington for intrahepatic injections; M. Cherrier for biochemistry; J. Perez, E. Maranghi and M. Manich for technical assistance; S. Akira, M. Chignard and V. Balloy for Myd88-/- , Trif-/- , Tlr2-/- or Tlr4-/- mice, J.P. Hugot for card15-/- mice, Millennium Pharmaceuticals for card4-/- mice and D. Philpott for card4-/- mice backcrossed to C57BL/6; J. Browning for LTβR-Ig and Z. Ge for the 16S rDNA plasmids. This work was supported by Institut Pasteur, CNRS, INSERM, ANR, Fondation de la Recherche Médicale, Mairie de Paris, a Marie Curie Excellence grant, La Fondation de France (D.B.) and la Ligue Nationale contre le Cancer (D.B.).
Author Contributions All authors, except R.V., contributed to the design of experiments, analysis of the data and writing of the manuscript. G.E. supervised experiments and wrote the manuscript, D.B. performed most experiments, C.B. and M.B. reconstituted germ-free mice with bacterial cocktails or strains, C.B. developed strain-specific qPCR on 16S rDNA, R.V. provided CCR6-deficient mice, I.G.B. purified PGNs and prepared E. coli mutants, and C.W. tested the functionality of PGNs and E. coli mutants.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-7 with Legends, Supplementary Table 1 and Supplementary References (PDF 5408 kb)
Rights and permissions
About this article
Cite this article
Bouskra, D., Brézillon, C., Bérard, M. et al. Lymphoid tissue genesis induced by commensals through NOD1 regulates intestinal homeostasis. Nature 456, 507–510 (2008). https://doi.org/10.1038/nature07450
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07450
This article is cited by
-
The emerging family of RORγt+ antigen-presenting cells
Nature Reviews Immunology (2024)
-
Microbial ligand-independent regulation of lymphopoiesis by NOD1
Nature Immunology (2023)
-
Role of the gut microbiota in anticancer therapy: from molecular mechanisms to clinical applications
Signal Transduction and Targeted Therapy (2023)
-
Towards modulating the gut microbiota to enhance the efficacy of immune-checkpoint inhibitors
Nature Reviews Clinical Oncology (2023)
-
Optical imaging of the small intestine immune compartment across scales
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.