Abstract
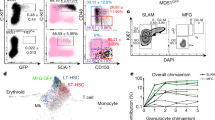
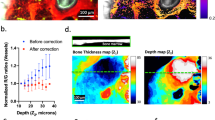
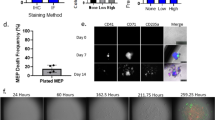
Stem cells reside in a specialized, regulatory environment termed the niche that dictates how they generate, maintain and repair tissues1,2. We have previously documented that transplanted haematopoietic stem and progenitor cell populations localize to subdomains of bone-marrow microvessels where the chemokine CXCL12 is particularly abundant3. Using a combination of high-resolution confocal microscopy and two-photon video imaging of individual haematopoietic cells in the calvarium bone marrow of living mice over time, we examine the relationship of haematopoietic stem and progenitor cells to blood vessels, osteoblasts and endosteal surface as they home and engraft in irradiated and c-Kit-receptor-deficient recipient mice. Osteoblasts were enmeshed in microvessels and relative positioning of stem/progenitor cells within this complex tissue was nonrandom and dynamic. Both cell autonomous and non-autonomous factors influenced primitive cell localization. Different haematopoietic cell subsets localized to distinct locations according to the stage of differentiation. When physiological challenges drove either engraftment or expansion, bone-marrow stem/progenitor cells assumed positions in close proximity to bone and osteoblasts. Our analysis permits observing in real time, at a single cell level, processes that previously have been studied only by their long-term outcome at the organismal level.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schofield, R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells 4, 7–25 (1978)
Xie, T. & Spradling, A. C. A niche maintaining germ line stem cells in the Drosophila ovary. Science 290, 328–330 (2000)
Sipkins, D. A. et al. In vivo imaging of specialized bone marrow endothelial microdomains for tumour engraftment. Nature 435, 969–973 (2005)
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003)
Nilsson, S. K. et al. Osteopontin, a key component of the hematopoietic stem cell niche and regulator of primitive hematopoietic progenitor cells. Blood 106, 1232–1239 (2005)
Stier, S. et al. Osteopontin is a hematopoietic stem cell niche component that negatively regulates stem cell pool size. J. Exp. Med. 201, 1781–1791 (2005)
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003)
Arai, F. et al. Tie2/angiopoietin-1 signaling regulates hematopoietic stem cell quiescence in the bone marrow niche. Cell 118, 149–161 (2004)
Kiel, M. J. et al. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005)
Nilsson, S. K., Johnston, H. M. & Coverdale, J. A. Spatial localization of transplanted hemopoietic stem cells: inferences for the localization of stem cell niches. Blood 97, 2293–2299 (2001)
Lord, B. I., Testa, N. G. & Hendry, J. H. The relative spatial distributions of CFUs and CFUc in the normal mouse femur. Blood 46, 65–72 (1975)
Taichman, R. S., Reilly, M. J. & Emerson, S. G. The Hematopoietic microenvironment: Osteoblasts and the hematopoietic microenvironment. Hematology 4, 421–426 (2000)
Mayack, S. R. & Wagers, A. J. Osteolineage niche cells initiate hematopoietic stem cell mobilization. Blood 112, 519–531 (2008)
Crock, H. V. The Blood Supply of the Lower Limb Bones in Man (E&S Livingstone LTD, 1967)
Kalajzic, Z. et al. Directing the expression of a green fluorescent protein transgene in differentiated osteoblasts: comparison between rat type I collagen and rat osteocalcin promoters. Bone 31, 654–660 (2002)
Bryder, D., Rossi, D. J. & Weissman, I. L. Hematopoietic stem cells: the paradigmatic tissue-specific stem cell. Am. J. Pathol. 169, 338–346 (2006)
Kiel, M. J. et al. Haematopoietic stem cells do not asymmetrically segregate chromosomes or retain BrdU. Nature 449, 238–242 (2007)
Abkowitz, J. L., Catlin, S. N., McCallie, M. T. & Guttorp, P. Evidence that the number of hematopoietic stem cells per animal is conserved in mammals. Blood 100, 2665–2667 (2002)
Lewin, M. et al. Tat peptide-derivatized magnetic nanoparticles allow in vivo tracking and recovery of progenitor cells. Nature Biotechnol. 18, 410–414 (2000)
Suzuki, N. et al. Combinatorial Gata2 and Sca1 expression defines hematopoietic stem cells in the bone marrow niche. Proc. Natl Acad. Sci. USA 103, 2202–2207 (2006)
Adams, G. B. et al. Stem cell engraftment at the endosteal niche is specified by the calcium-sensing receptor. Nature 439, 599–603 (2006)
Zhong, J. F., Zhan, Y., Anderson, W. F. & Zhao, Y. Murine hematopoietic stem cell distribution and proliferation in ablated and nonablated bone marrow transplantation. Blood 100, 3521–3526 (2002)
Migliaccio, A. R., Carta, C. & Migliaccio, G. In vivo expansion of purified hematopoietic stem cells transplanted in nonablated W/Wv mice. Exp. Hematol. 27, 1655–1666 (1999)
Nilsson, S. K., Dooner, M. S. & Quesenberry, P. J. Synchronized cell-cycle induction of engrafting long-term repopulating stem cells. Blood 90, 4646–4650 (1997)
Forsberg, E. C., Serwold, T., Kogan, S., Weissman, I. L. & Passegue, E. New evidence supporting megakaryocyte-erythrocyte potential of flk2/flt3+ multipotent hematopoietic progenitors. Cell 126, 415–426 (2006)
Dykstra, B. et al. Long-term propagation of distinct hematopoietic differentiation programs in vivo . Cell Stem Cell 1, 218–229 (2007)
Adolfsson, J. et al. Identification of Flt3+ lympho-myeloid stem cells lacking erythro-megakaryocytic potential: a revised road map for adult blood lineage commitment. Cell 121, 295–306 (2005)
Osawa, M., Hanada, K., Hamada, H. & Nakauchi, H. Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science 273, 242–245 (1996)
Calvi, L. M. et al. Activated parathyroid hormone/parathyroid hormone-related protein receptor in osteoblastic cells differentially affects cortical and trabecular bone. J. Clin. Invest. 107, 277–286 (2001)
Adams, G. B. et al. Therapeutic targeting of a stem cell niche. Nature Biotechnol. 25, 238–243 (2007)
Zipfel, W. R., Williams, R. M. & Webb, W. W. Nonlinear magic: multiphoton microscopy in the biosciences. Nature Biotechnol. 21, 1369–1377 (2003)
Veilleux, I., Spencer, J. A., Biss, D. P., Cote, D. & Lin, C. P. In vivo cell tracking with video rate multimodality laser scanning microscopy. IEEE JSTQE 14, 10–18 (2008)
Brennand, K., Huangfu, D. & Melton, D. All beta cells contribute equally to islet growth and maintenance. PLoS Biol. 5, e163 (2007)
Acknowledgements
We thank E. Schipani for providing the PPR mice. We are grateful for help and advice from A. Catic, L. Purton, V. Janzen, G. Adams, J. Spencer, J. Runnels and P. O’Donovan. We thank Y. Tang for the mice husbandry care; D. Dombkowski, L. Prickett and K. Folz-Donahue for cell sorting expertise; R. Klein and K. Chomsky-Higgins for technical assistance; and C. Pasker, V. Shannon, M. Indico Miklosik and D. Machon for administrative assistance. C.L.C. was funded by EMBO and HFSP. The project was funded by the National Institutes of Health (to D.T.S. and C.P.L.), the Harvard Stem Cell Institute (to C.P.L.) and philanthropic sources (to D.T.S. and C.L.C.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
D.T.S. is a consultant and stockholder for Fate Therapeusis.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-9 with Legends and Supplementary Movie 1 Legend. (PDF 13921 kb)
Supplementary Movie 1
This file contains Supplementary Movie 1. (MOV 7538 kb)
Rights and permissions
About this article
Cite this article
Lo Celso, C., Fleming, H., Wu, J. et al. Live-animal tracking of individual haematopoietic stem/progenitor cells in their niche. Nature 457, 92–96 (2009). https://doi.org/10.1038/nature07434
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07434
This article is cited by
-
Resilient anatomy and local plasticity of naive and stress haematopoiesis
Nature (2024)
-
Minimally invasive longitudinal intravital imaging of cellular dynamics in intact long bone
Nature Protocols (2023)
-
Distinct and targetable role of calcium-sensing receptor in leukaemia
Nature Communications (2023)
-
Different niches for stem cells carrying the same oncogenic driver affect pathogenesis and therapy response in myeloproliferative neoplasms
Nature Cancer (2023)
-
Bone marrow imaging reveals the migration dynamics of neonatal hematopoietic stem cells
Communications Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.