Abstract
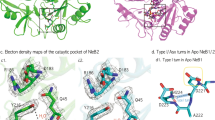
AB5 toxins comprise an A subunit that corrupts essential eukaryotic cell functions, and pentameric B subunits that direct target-cell uptake after binding surface glycans. Subtilase cytotoxin (SubAB) is an AB5 toxin secreted by Shiga toxigenic Escherichia coli (STEC)1, which causes serious gastrointestinal disease in humans2. SubAB causes haemolytic uraemic syndrome-like pathology in mice3 through SubA-mediated cleavage of BiP/GRP78, an essential endoplasmic reticulum chaperone4. Here we show that SubB has a strong preference for glycans terminating in the sialic acid N-glycolylneuraminic acid (Neu5Gc), a monosaccharide not synthesized in humans. Structures of SubB-Neu5Gc complexes revealed the basis for this specificity, and mutagenesis of key SubB residues abrogated in vitro glycan recognition, cell binding and cytotoxicity. SubAB specificity for Neu5Gc was confirmed using mouse tissues with a human-like deficiency of Neu5Gc and human cell lines fed with Neu5Gc. Despite lack of Neu5Gc biosynthesis in humans, assimilation of dietary Neu5Gc creates high-affinity receptors on human gut epithelia and kidney vasculature. This, and the lack of Neu5Gc-containing body fluid competitors in humans, confers susceptibility to the gastrointestinal and systemic toxicities of SubAB. Ironically, foods rich in Neu5Gc are the most common source of STEC contamination. Thus a bacterial toxin's receptor is generated by metabolic incorporation of an exogenous factor derived from food.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Primary accessions
Protein Data Bank
Data deposits
The coordinates and structure factors for the SubB structures are deposited in Protein Data Bank under accession numbers 3DWA, 3DWP and 3DWQ. Raw glycan array data are available at http://www.functionalglycomics.org/glycomics/publicdata/selectedScreens.jsp.
References
Paton, A. W., Srimanote, P., Talbot, U. M., Wang, H. & Paton, J. C. A new family of potent AB5 cytotoxins produced by Shiga toxigenic Escherichia coli . J. Exp. Med. 200, 35-46 (2004)
Paton, J. C. & Paton, A. W. Pathogenesis and diagnosis of Shiga toxin-producing Escherichia coli infections. Clin. Microbiol. Rev. 11, 450-479 (1998)
Wang, H., Paton, J. C. & Paton, A. W. Pathologic changes in mice induced by subtilase cytotoxin, a potent new Escherichia coli AB5 toxin that targets the endoplasmic reticulum. J. Infect. Dis. 196, 1093-1101 (2007)
Paton, A. W. & Beddoe, T. Thorpe, C.M., Whisstock, J.C., Wilce, M.C.J., Rossjohn, J., Talbot, U.M. and Paton J.C. AB5 subtilase cytotoxin inactivates the endoplasmic reticulum chaperone BiP. Nature 443, 548-552 (2006)
Sandvig, K. & van Deurs, B. Membrane traffic exploited by protein toxins. Annu. Rev. Cell Dev. Biol. 18, 1-24 (2002)
Lencer, W. I. & Tsai, B. The intracellular voyage of cholera toxin: going retro. Trends Biochem. Sci. 28, 639-645 (2003)
Crocker, P. R., Paulson, J. C. & Varki, A. Siglecs and their roles in the immune system. Nature Rev. Immunol. 7, 55-66 (2007)
Neu, U., Woellner, K., Gauglitz, G. & Stehle, T. Structural basis of GM1 ganglioside recognition by simian virus 40. Proc. Natl Acad. Sci. USA 105, 5219-5224 (2008)
Murzin, A. G. OB (oligonucleotide/oligosaccharide binding)-fold: common structural and functional solution for non-homologous sequences. EMBO J. 12, 861-867 (1993)
Stein, P. E. et al. Structure of a pertussis toxin-sugar complex as a model for receptor binding. Nature Struct. Biol. 1, 591-596 (1994)
Merritt, E. A., Sixma, T. K., Kalk, K. H., van Zanten, B. A. & Hol, W. G. Galactose-binding site in Escherichia coli heat-labile enterotoxin (LT) and cholera toxin (CT). Mol. Microbiol. 13, 745-753 (1994)
Merritt, E. A. et al. Structural studies of receptor binding by cholera toxin mutants. Protein Sci. 6, 1516-1528 (1997)
Ling, H., Bast, D. & Brunton, J. L. &. R. e. a. d. R. J. Structure of the Shiga-like toxin I B-pentamer complexed with an analogue of its receptor Gb3. Biochemistry 37, 1777-1788 (1998)
Yahiro, K. et al. Identification and characterization of receptors for vacuolating activity of subtilase cytotoxin. Mol. Microbiol. 62, 480-490 (2006)
Hedlund, M. et al. N-glycolylneuraminic acid deficiency in mice: implications for human biology and evolution. Mol. Cell. Biol. 27, 4340-4346 (2007)
Varki, A. Multiple changes in sialic acid biology during human evolution. Glycoconjugate J. 10.1007/s10719-008-9183-z (7 September 2008)
Tangvoranuntakul, P. et al. Human uptake and incorporation of an immunogenic nonhuman dietary sialic acid. Proc. Natl Acad. Sci. USA 100, 12045-12050 (2003)
Gagneux, P. et al. Proteomic comparison of human and great ape blood plasma reveals conserved glycosylation and differences in thyroid hormone metabolism. Am. J. Phys. Anthropol. 115, 99-109 (2001)
Talbot, U. M., Paton, J. C. & Paton, A. W. Protective immunization of mice with an active-site mutant of subtilase cytotoxin of Shiga toxin-producing Escherichia coli . Infect. Immun. 73, 4432-4436 (2005)
Chong, D. C., Paton, J. C., Thorpe, C. M. & Paton, A. W. Clathrin-dependent trafficking of subtilase cytotoxin, a novel AB5 toxin that targets the ER chaperone BiP. Cell. Microbiol. 10, 795-806 (2008)
Leslie, A. G. W. in Joint CCP4 and ESF-EACMB Newsletter on Protein Crystallography 26 (SERC, 1992)
Evans, P. R. in Proc. CCP4 Study Weekend on Recent Advances in Phasing 97-102 (CCLRC, 1997)
Collaborative Computational Project. Number 4. Acta Crystallogr. D 50, 760-763 (1994)
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948-1954 (2002)
Terwilliger, T. C. Automated main-chain model building by template matching and iterative fragment extension. Acta Crystallogr. D 59, 38-44 (2003)
Perrakis, A., Sixma, T. K., Wilson, K. S. & Lamzin, V. S. Warp: improvement and extension of crystallographic phases by weighted averaging of multiple refined dummy models. Acta Crystallogr. D 30, 551-554 (1997)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126-2132 (2004)
Murshodov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum likelihood method. Acta Crystallogr. D 53, 240-255 (1997)
McCoy, A. J., Grosse-Kunstleve, R. W., Storoni, L. C. & Read, R. J. Likelihood-enhanced fast translation functions. Acta Crystallogr. D 61, 458-464 (2005)
Schuettelkopf, A. W. & van Aalten, D. M. F. PRODRG - a tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr. D 60, 1355-1363 (2004)
Acknowledgements
We thank the staff at the General Medicine and Cancer Institutes Collaborative Access Team Advanced Photon Source, Chicago, for assistance with data collection. This research was supported by a Program Grant from the National Health and Medical Research Council of Australia (NHMRC; to A.W.P. and J.C.P.), an NHMRC Project Grant (to T.B. and A.W.P.), a grant from the National Institute of General Medical Sciences to the Consortium for Functional Glycomics, RO1 grants from the National Institutes of Health (to A.W.P., J.C.P., J.R., X.C. and A.V.) and from the ARC Centre of Excellence in Structural and Functional Microbial Genomics (to J.R.). J.R. is supported by an Australian Research Council Federation Fellowship; T.B. by an NHMRC Career Development Award; J.C.P. by an NHMRC Australia Fellowship. We also thank C. J. Gregg for assistance with in vivo experiments and with collection of data, and L. Wiggleton for technical assistance with tissue sectioning and staining.
Author Contributions E.B. and A.W.P. contributed equally. E.B, T.B. and M.C.J.W. crystallized SubB, solved the structure and contributed to manuscript preparation. A.W.P. constructed mutants and contributed to design and interpretation of experiments, project management and writing of the manuscript. J.C.P. and J.R. contributed to design and interpretation of experiments, project management and writing of the manuscript. D.C.C. and U.M.T. performed experiments. D.F.S. performed and interpreted glycan array experiments. J.C.L., N.M.V. and A.V. designed, performed and interpreted experiments relating to Neu5Gc on cells and tissues and to cytotoxicity in vivo, and contributed to manuscript preparation. H.Y., S.H. and X.C. synthesized oligosaccharides. A.V., T.B. and J.R. are joint senior and corresponding authors.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-5 and Supplementary Figures 1-2 with Legends (PDF 418 kb)
Rights and permissions
About this article
Cite this article
Byres, E., Paton, A., Paton, J. et al. Incorporation of a non-human glycan mediates human susceptibility to a bacterial toxin. Nature 456, 648–652 (2008). https://doi.org/10.1038/nature07428
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07428
This article is cited by
-
Sialic acid exacerbates gut dysbiosis-associated mastitis through the microbiota-gut-mammary axis by fueling gut microbiota disruption
Microbiome (2023)
-
N-glycolylneuraminic acid serum biomarker levels are elevated in breast cancer patients at all stages of disease
BMC Cancer (2022)
-
A novel endoplasmic stress mediator, Kelch domain containing 7B (KLHDC7B), increased Harakiri (HRK) in the SubAB-induced apoptosis signaling pathway
Cell Death Discovery (2021)
-
The enzyme subunit SubA of Shiga toxin-producing E. coli strains demonstrates comparable intracellular transport and cytotoxic activity as the holotoxin SubAB in HeLa and HCT116 cells in vitro
Archives of Toxicology (2021)
-
Subtilase cytotoxin induces a novel form of Lipocalin 2, which promotes Shiga-toxigenic Escherichia coli survival
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.