Abstract
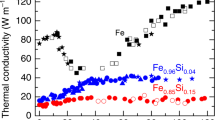
Iron in crustal and mantle minerals adopts several possible oxidation states: this has implications for biogeochemical processes1, oxygenation of the atmosphere2 and the oxidation state of the mantle3,4. In the deep Earth, iron in silicate perovskite, (Mg0.9Fe0.1)SiO3, and ferropericlase, (Mg0.85Fe0.15)O, influences the thermal conductivity of the lower mantle and therefore heat flux from the core. Little is known, however, about the effect of iron oxidation states on transport properties. Here we show that the radiative component of thermal conductivity in the dominant silicate perovskite material of Earth’s lower mantle is controlled by the amount of ferric iron, Fe3+. We obtained the optical absorption spectra of silicate perovskite and ferropericlase at pressures up to 133 GPa, corresponding to pressures at the core–mantle boundary. Absorption spectra of ferropericlase up to 800 K and 60 GPa exhibit minimal temperature dependence. The results on silicate perovskite show that optical absorption in the visible and near-infrared spectral range is dominated by O–Fe3+ charge transfer and Fe3+–Fe2+ intervalence transitions, whereas a contribution from the Fe2+ crystal-field transitions is substantially smaller. The estimated pressure-dependent radiative conductivity, krad, from these data is 2–5 times lower than previously inferred from model extrapolations, with implications for the evolution of the mantle, such as generation and stability of thermo-chemical plumes in the lower mantle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Johnson, C. M. & Beard, B. L. Biogeochemical cycling of iron isotopes. Science 309, 1025–1027 (2005)
Rouxel, O. J., Bekker, A. & Edwards, K. J. Iron isotope constraints on the Archean and Paleoproterozoic ocean redox state. Science 307, 1088–1091 (2005)
McCammon, C. Perovskite as a possible sink for ferric iron in the lower mantle. Nature 387, 694–696 (1997)
Frost, D. J. et al. Experimental evidence for the existence of iron-rich metal in the Earth’s lower mantle. Nature 428, 409–412 (2004)
Zhong, S. Constraints on thermochemical convection of the mantle from plume heat flux, plume excess temperature, and upper mantle temperature. J. Geophys. Res. 111 B04409 doi: 10.1029/2005JB003972 (2006)
Yanagawa, T. K. B., Nakada, M. & Yuen, D. A. Influence of lattice thermal conductivity on thermal convection with strongly temperature-dependent viscosity. Earth Planets Space 57, 15–28 (2005)
Hofmeister, A. M. Mantle values of thermal conductivity and the geotherm from phonon lifetimes. Science 283, 1699–1706 (1999)
Hofmeister, A. M. & Yuen, D. A. Critical phenomena in thermal conductivity: Implications for lower mantle dynamics. J. Geodyn. 44, 186–199 (2007)
Dubuffet, F., Yuen, D. A. & Rainey, E. S. G. Controlling thermal chaos in the mantle by positive feedback from radiative thermal conductivity. Nonlin. Process. Geophys. 9, 1–13 (2002)
van den Berg, A. P., Rainey, E. S. G. & Yuen, D. A. The combined influences of variable thermal conductivity, temperature- and pressure-dependent viscosity and core–mantle coupling on thermal evolution. Phys. Earth Planet. Inter. 149, 259–278 (2005)
Clark, S. P. Radiative transfer in the Earth’s mantle. Trans. Am. Geophys. Union 38, 931–938 (1957)
Shankland, T. J., Nitsan, U. & Duba, A. G. Optical absorption and radiative heat transport in olivine at high temperature. J. Geophys. Res. 84, 1603–1610 (1979)
Burns, R. G. Mineralogical Applications of Crystal Field Theory 2nd edn (Cambridge Univ. Press, 1993)
Sherman, D. M. The high-pressure electronic structure of magnesiowüstite (Mg,Fe)O: Applications to the physics and chemistry of the lower mantle. J. Geophys. Res. 96, 14299–14312 (1991)
Badro, J. et al. Transitions in perovskite: Possible nonconvecting layers in the lower mantle. Science 305, 383–386 (2004)
Ross, R. G., Andersson, P., Sundqvist, B. & Bäckström, G. Thermal conductivity of solids and liquids under pressure. Rep. Prog. Phys. 47, 1347–1402 (1984)
Mao, H. K. & Bell, P. M. Electrical conductivity and the red shift of absorption in olivine and spinel at high pressure. Science 176, 403–406 (1972)
Keppler, H. & Smyth, J. R. Optical and near infrared spectra of ringwoodite to 21.5 GPa: Implications for radiative heat transport in the mantle. Am. Mineral. 90, 1209–1212 (2005)
Pertermann, M. & Hofmeister, A. M. Thermal diffusivity of olivine-group minerals at high temperature. Am. Mineral. 91, 1747–1760 (2006)
Goncharov, A. F., Struzhkin, V. V. & Jacobsen, S. D. Reduced radiative conductivity of low-spin (Mg,Fe)O in the lower mantle. Science 312, 1205–1208 (2006)
Keppler, H., Kantor, I. & Dubrovinsky, L. S. Optical absorption spectra of ferropericlase to 84 GPa. Am. Mineral. 92, 433–436 (2007)
Keppler, H., McCammon, C. A. & Rubie, D. C. Crystal-field and charge-transfer spectra of (Mg,Fe)SiO3 perovskite. Am. Mineral. 79, 1215–1218 (1994)
Shen, G., Fei, Y., Hålenius, U. & Wang, Y. Optical absorption spectra of (Mg,Fe)SiO3 silicate perovskites. Phys. Chem. Miner. 20, 478–482 (1994)
Mattson, S. M. & Rossman, G. R. Identifying characteristics of charge transfer transitions in minerals. Phys. Chem. Miner. 14, 94–99 (1987)
Bocquet, A. E. et al. Electronic structure of early 3d-transition-metal oxides by analysis of the 2p core-level photoemission spectra. Phys. Rev. B 53, 1161–1170 (1996)
Badro, J. et al. Iron partitioning in Earth’s mantle: Toward a deep lower mantle discontinuity. Science 300, 789–791 (2003)
Li, J. et al. Pressure effect on the electronic structure of iron in (Mg,Fe)(Si,Al)O3 perovskite: A combined synchrotron Mössbauer and X-ray emission spectroscopy study up to 100 GPa. Phys. Chem. Miner. 33, 575–585 (2006)
Turcotte, D. L. & Schubert, G. Geodynamics (Cambridge Univ. Press, 2002)
Montague, N. L., Kellogg, L. H. & Manga, M. High-Rayleigh number thermo-chemical models of a dense boundary layer in D. Geophys. Res. Lett. 25, 2345–2348 (1998)
Naliboff, J. B. & Kellogg, L. H. Can large increases in viscosity and thermal conductivity preserve large-scale heterogeneity in the mantle? Phys. Earth Planet. Inter. 161, 86–102 (2007)
Acknowledgements
We acknowledge support from NSF/EAR, DOE/BES, DOE/NNSA (CDAC) and the W. M. Keck Foundation. S.D.J. thanks D. J. Frost, S. J. Mackwell and D. P. Dobson for help with sample synthesis, C. A. McCammon for Mössbauer spectroscopy, J. R. Smyth for single-crystal X-ray diffraction, H. Watson for electron microprobe analysis of the silicate perovskite material and the NSF and Bayerishes Geoinstitut Visitor’s Program for support. B.D.H. was supported by the NSF Research Experience for Undergraduates (REU) Program at the Carnegie Institution of Washington. P.B. was partially supported by the Balzan Foundation.
Author Contributions A.F.G., V.V.S. and S.D.J. designed the research programme; S.D.J. synthesized and polished the single crystals; A.F.G. and B.D.H. performed high-pressure experiments at room temperature; A.F.G., V.V.S. and P.B. performed high-temperature experiments; A.F.G., V.V.S., B.D.H. and P.B. analysed the data; P.B. developed a thermal conductivity model; V.V.S, A.F.G. and S.D.J interpreted the results; A.F.G, S.D.J., P.B. and V.V.S. wrote the paper. All authors discussed the results and commented on the manuscripts.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Figures S1-S3 with Legends and Supplementary References. (PDF 330 kb)
PowerPoint slides
Rights and permissions
About this article
Cite this article
Goncharov, A., Haugen, B., Struzhkin, V. et al. Radiative conductivity in the Earth’s lower mantle. Nature 456, 231–234 (2008). https://doi.org/10.1038/nature07412
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature07412
This article is cited by
-
Full-waveform tomography reveals iron spin crossover in Earth’s lower mantle
Nature Communications (2024)
-
Thermal conductivity of materials under pressure
Nature Reviews Physics (2022)
-
Spectroscopic and ab initio studies of the pressure-induced Fe2+ high-spin-to-low-spin electronic transition in natural triphylite–lithiophilite
Physics and Chemistry of Minerals (2019)
-
High-pressure optical spectroscopy study of natural siderite
Physics and Chemistry of Minerals (2017)
-
High-pressure radiative conductivity of dense silicate glasses with potential implications for dark magmas
Nature Communications (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.