Abstract
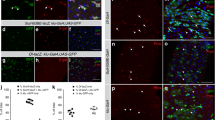
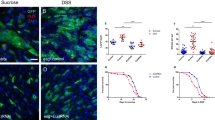
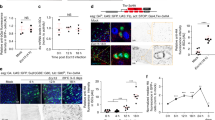
In the Drosophila midgut, multipotent intestinal stem cells (ISCs) that are scattered along the epithelial basement membrane maintain tissue homeostasis by their ability to steadily produce daughters that differentiate into either enterocytes or enteroendocrine cells, depending on the levels of Notch activity1,2,3. However, the mechanisms controlling ISC self-renewal remain elusive. Here we show that a canonical Wnt signalling pathway controls ISC self-renewal. The ligand Wingless (Wg) is specifically expressed in the circular muscles next to ISCs, separated by a thin layer of basement membrane. Reduced function of wg causes ISC quiescence and differentiation, whereas wg overexpression produces excessive ISC-like cells that express high levels of the Notch ligand, Delta. Clonal analysis shows that the main downstream components of the Wg pathway, including Frizzled, Dishevelled and Armadillo, are autonomously required for ISC self-renewal. Furthermore, epistatic analysis suggests that Notch acts downstream of the Wg pathway and a hierarchy of Wg/Notch signalling pathways controls the balance between self-renewal and differentiation of ISCs. These data suggest that the underlying circular muscle constitutes the ISC niche, which produce Wg signals that act directly on ISCs to promote ISC self-renewal. This study demonstrates markedly conserved mechanisms regulating ISCs from Drosophila to mammals. The identification of the Drosophila ISC niche and the principal self-renewal signal will facilitate further understanding of intestinal homeostasis control and tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Micchelli, C. A. & Perrimon, N. Evidence that stem cells reside in the adult Drosophila midgut epithelium. Nature 439, 475–479 (2006)
Ohlstein, B. & Spradling, A. The adult Drosophila posterior midgut is maintained by pluripotent stem cells. Nature 439, 470–474 (2006)
Ohlstein, B. & Spradling, A. Multipotent Drosophila intestinal stem cells specify daughter cell fates by differential notch signaling. Science 315, 988–992 (2007)
Crosnier, C., Stamataki, D. & Lewis, J. Organizing cell renewal in the intestine: stem cells, signals and combinatorial control. Nature Rev. Genet. 7, 349–359 (2006)
Kassis, J. A., Noll, E., VanSickle, E. P., Odenwald, W. F. & Perrimon, N. Altering the insertional specificity of a Drosophila transposable element. Proc. Natl Acad. Sci. USA 89, 1919–1923 (1992)
Bejsovec, A. & Martinez Arias, A. Roles of wingless in patterning the larval epidermis of Drosophila . Development 113, 471–485 (1991)
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993)
Moon, R. T., Bowerman, B., Boutros, M. & Perrimon, N. The promise and perils of Wnt signaling through β-catenin. Science 296, 1644–1646 (2002)
Lee, T., Winter, C., Marticke, S. S., Lee, A. & Luo, L. Essential roles of Drosophila RhoA in the regulation of neuroblast proliferation and dendritic but not axonal morphogenesis. Neuron 25, 307–316 (2000)
Chen, C. M. & Struhl, G. Wingless transduction by the Frizzled and Frizzled2 proteins of Drosophila . Development 126, 5441–5452 (1999)
Bhanot, P. et al. Frizzled and Dfrizzled-2 function as redundant receptors for Wingless during Drosophila embryonic development. Development 126, 4175–4186 (1999)
Heslip, T. R., Theisen, H., Walker, H. & Marsh, J. L. Shaggy and dishevelled exert opposite effects on Wingless and Decapentaplegic expression and on positional identity in imaginal discs. Development 124, 1069–1078 (1997)
Peifer, M. & Wieschaus, E. The segment polarity gene armadillo encodes a functionally modular protein that is the Drosophila homolog of human plakoglobin. Cell 63, 1167–1176 (1990)
Song, X. & Xie, T. wingless signaling regulates the maintenance of ovarian somatic stem cells in Drosophila . Development 130, 3259–3268 (2003)
Pai, L. M., Orsulic, S., Bejsovec, A. & Peifer, M. Negative regulation of Armadillo, a Wingless effector in Drosophila . Development 124, 2255–2266 (1997)
Struhl, G. & Basler, K. Organizing activity of wingless protein in Drosophila . Cell 72, 527–540 (1993)
van de Wetering, M. et al. Armadillo coactivates transcription driven by the product of the Drosophila segment polarity gene dTCF . Cell 88, 789–799 (1997)
Bourouis, M. Targeted increase in shaggy activity levels blocks wingless signaling. Genesis 34, 99–102 (2002)
Ohlstein, B., Kai, T., Decotto, E. & Spradling, A. The stem cell niche: theme and variations. Curr. Opin. Cell Biol. 16, 693–699 (2004)
Nystul, T. & Spradling, A. An epithelial niche in the Drosophila ovary undergoes long-range stem cell replacement. Cell Stem Cell 1, 277–285 (2007)
Nikolova, G., Strilic, B. & Lammert, E. The vascular niche and its basement membrane. Trends Cell Biol. 17, 19–25 (2007)
Gregorieff, A. et al. Expression pattern of Wnt signaling components in the adult intestine. Gastroenterology 129, 626–638 (2005)
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5 . Nature 449, 1003–1007 (2007)
Radtke, F. & Clevers, H. Self-renewal and cancer of the gut: two sides of a coin. Science 307, 1904–1909 (2005)
Acknowledgements
We thank G. Struhl, L. Cooley, S. Hayashi, T. Xie, the Bloomington Stock Center for fly stocks, and members of the Xi laboratory for comments and discussions. This work was supported by an ‘863’ grant 2007AA02Z1A2 to R.X. from the Chinese Ministry of Science and Technology.
Author Contributions G.L. and R.X. designed the research; N.X. performed the in situ hybridization experiments and G.L. performed all other experiments; G.L., N.X. and R.X. analysed the data and R.X. wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, G., Xu, N. & Xi, R. Paracrine Wingless signalling controls self-renewal of Drosophila intestinal stem cells. Nature 455, 1119–1123 (2008). https://doi.org/10.1038/nature07329
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07329
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.