Abstract
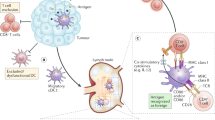
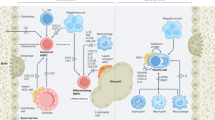
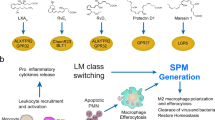
The mediators and cellular effectors of inflammation are important constituents of the local environment of tumours. In some types of cancer, inflammatory conditions are present before a malignant change occurs. Conversely, in other types of cancer, an oncogenic change induces an inflammatory microenvironment that promotes the development of tumours. Regardless of its origin, 'smouldering' inflammation in the tumour microenvironment has many tumour-promoting effects. It aids in the proliferation and survival of malignant cells, promotes angiogenesis and metastasis, subverts adaptive immune responses, and alters responses to hormones and chemotherapeutic agents. The molecular pathways of this cancer-related inflammation are now being unravelled, resulting in the identification of new target molecules that could lead to improved diagnosis and treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to Virchow? Lancet 357, 539–545 (2001). This paper revisits Virchow's legacy and highlights the connections between inflammation and cancer.
Balkwill, F., Charles, K. A. & Mantovani, A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell 7, 211–217 (2005).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Karin, M. Nuclear factor-κB in cancer development and progression. Nature 441, 431–436 (2006).
Koehne, C. H. & Dubois, R. N. COX-2 inhibition and colorectal cancer. Semin. Oncol. 31, 12–21 (2004).
Flossmann, E. & Rothwell, P. M. Effect of aspirin on long-term risk of colorectal cancer: consistent evidence from randomised and observational studies. Lancet 369, 1603–1613 (2007).
Chan, A. T., Ogino, S. & Fuchs, C. S. Aspirin and the risk of colorectal cancer in relation to the expression of COX-2. N. Engl. J. Med. 356, 2131–2142 (2007).
Borrello, M. G. et al. Induction of a proinflammatory program in normal human thyrocytes by the RET/PTC1 oncogene. Proc. Natl Acad. Sci. USA 102, 14825–14830 (2005). This is the first report that a frequent genetic event that causes cancer in humans (rearrangement of the chromosome on which RET is located, in human papillary thyroid carcinoma) activates an inflammatory transcriptional program in normal cells that is associated with metastatic behaviour.
De Falco, V. et al. Biological role and potential therapeutic targeting of the chemokine receptor CXCR4 in undifferentiated thyroid cancer. Cancer Res. 67, 11821–11829 (2007).
Xu, K. & Shu, H. K. EGFR activation results in enhanced cyclooxygenase-2 expression through p38 mitogen-activated protein kinase-dependent activation of the Sp1/Sp3 transcription factors in human gliomas. Cancer Res. 67, 6121–6129 (2007).
Guerra, C. et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell 11, 291–302 (2007).
Sparmann, A. & Bar-Sagi, D. Ras-induced interleukin-8 expression plays a critical role in tumor growth and angiogenesis. Cancer Cell 6, 447–458 (2004).
Sumimoto, H., Imabayashi, F., Iwata, T. & Kawakami, Y. The BRAF–MAPK signaling pathway is essential for cancer-immune evasion in human melanoma cells. J. Exp. Med. 203, 1651–1656 (2006).
Shchors, K. et al. The Myc-dependent angiogenic switch in tumors is mediated by interleukin 1β. Genes Dev. 20, 2527–2538 (2006).
Soucek, L. et al. Mast cells are required for angiogenesis and macroscopic expansion of Myc-induced pancreatic islet tumors. Nature Med. 13, 1211–1218 (2007).
Balkwill, F. Cancer and the chemokine network. Nature Rev. Cancer 4, 540–550 (2004).
Kobielak, A. & Fuchs, E. Links between α-catenin, NF-κB, and squamous cell carcinoma in skin. Proc. Natl Acad. Sci. USA 103, 2322–2327 (2006).
Phillips, R. J. et al. Epidermal growth factor and hypoxia-induced expression of CXC chemokine receptor 4 on non-small cell lung cancer cells is regulated by the phosphatidylinositol 3-kinase/PTEN/AKT/mammalian target of rapamycin signaling pathway and activation of hypoxia inducible factor-1α. J. Biol. Chem. 280, 22473–22481 (2005).
Schioppa, T. et al. Regulation of the chemokine receptor CXCR4 by hypoxia. J. Exp. Med. 198, 1391–1402 (2003).
Staller, P. et al. Chemokine receptor CXCR4 downregulated by von Hippel-Lindau tumour suppressor pVHL. Nature 425, 307–311 (2003).
Bierie, B. & Moses, H. L. TGF-β and cancer. Cytokine Growth Factor Rev. 17, 29–40 (2006).
Yu, H., Kortylewski, M. & Pardoll, D. Crosstalk between cancer and immune cells: role of STAT3 in the tumour microenvironment. Nature Rev. Immunol. 7, 41–51 (2007).
Voronov, E. et al. IL-1 is required for tumor invasiveness and angiogenesis. Proc. Natl Acad. Sci. USA 100, 2645–2650 (2003).
Grivennikov, S. & Karin, M. Autocrine IL-6 signaling: a key event in tumorigenesis? Cancer Cell 13, 7–9 (2008).
Szlosarek, P. W. & Balkwill, F. R. Tumour necrosis factor α: a potential target for the therapy of solid tumours. Lancet Oncol. 4, 565–573 (2003).
Langowski, J. L. et al. IL-23 promotes tumour incidence and growth. Nature 442, 461–465 (2006).
Courtois, G. & Gilmore, T. D. Mutations in the NF-κB signaling pathway: implications for human disease. Oncogene 25, 6831–6843 (2006).
Carbia-Nagashima, A. et al. RSUME, a small RWD-containing protein, enhances SUMO conjugation and stabilizes HIF-1α during hypoxia. Cell 131, 309–323 (2007).
Mizukami, Y. et al. Induction of interleukin-8 preserves the angiogenic response in HIF-1α-deficient colon cancer cells. Nature Med. 11, 992–997 (2005).
Rius, J. et al. NF-κB links innate immunity to the hypoxic response through transcriptional regulation of HIF-1α. Nature 453, 807–811 (2008).
Greten, F. R. et al. IKKβ links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118, 285–296 (2004).
Pikarsky, E. et al. NF-κB functions as a tumour promoter in inflammation-associated cancer. Nature 431, 461–466 (2004). References 31 and 32 provide evidence that NF-κB is an endogenous promoter of colon and liver carcinogenesis. Reference 31 also shows that NF-κB activation in myeloid cells is required for colitis-associated cancer.
Maeda, S., Kamata, H., Luo, J. L., Leffert, H. & Karin, M. IKKβ couples hepatocyte death to cytokine-driven compensatory proliferation that promotes chemical hepatocarcinogenesis. Cell 121, 977–990 (2005).
Garlanda, C. et al. Increased susceptibility to colitis-associated cancer of mice lacking TIR8, an inhibitory member of the interleukin-1 receptor family. Cancer Res. 67, 6017–6021 (2007).
Xiao, H. et al. The Toll–interleukin-1 receptor member SIGIRR regulates colonic epithelial homeostasis, inflammation, and tumorigenesis. Immunity 26, 461–475 (2007).
Biswas, S. K. et al. A distinct and unique transcriptional program expressed by tumor-associated macrophages: defective NF-κB and enhanced IRF-3/STAT1 activation. Blood 107, 2112–2122 (2006).
Saccani, A. et al. p50 nuclear factor-κB overexpression in tumor-associated macrophages inhibits M1 inflammatory responses and antitumor resistance. Cancer Res. 66, 11432–11440 (2006).
Bromberg, J. F. et al. Stat3 as an oncogene. Cell 98, 295–303 (1999).
Wang, T. et al. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nature Med. 10, 48–54 (2004).
Kortylewski, M. et al. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nature Med. 11, 1314–1321 (2005).
Mantovani, A., Bottazzi, B., Colotta, F., Sozzani, S. & Ruco, L. The origin and function of tumor-associated macrophages. Immunol. Today 13, 265–270 (1992).
Coussens, L. M., Tinkle, C. L., Hanahan, D. & Werb, Z. MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 103, 481–490 (2000).
Bunt, S. K. et al. Reduced inflammation in the tumor microenvironment delays the accumulation of myeloid-derived suppressor cells and limits tumor progression. Cancer Res. 67, 10019–10026 (2007).
Lin, E. Y., Nguyen, A. V., Russell, R. G. & Pollard, J. W. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J. Exp. Med. 193, 727–740 (2001). This paper describes the first genetic evidence that TAMs promote cancer, in a study of a primary breast carcinoma model.
De Palma, M. et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8, 211–226 (2005).
Sica, A. & Bronte, V. Altered macrophage differentiation and immune dysfunction in tumor development. J. Clin. Invest. 117, 1155–1166 (2007).
Mantovani, A., Sozzani, S., Locati, M., Allavena, P. & Sica, A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 23, 549–555 (2002).
Hagemann, T. et al. Ovarian cancer cells polarize macrophages toward a tumor-associated phenotype. J. Immunol. 176, 5023–5032 (2006).
Fischer, C. et al. Anti-PlGF inhibits growth of VEGFR-inhibitor-resistant tumors without affecting healthy vessels. Cell 131, 463–475 (2007).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Shojaei, F. et al. Bv8 regulates myeloid-cell-dependent tumour angiogenesis. Nature 450, 825–831 (2007).
Coussens, L. M. et al. Inflammatory mast cells up-regulate angiogenesis during squamous epithelial carcinogenesis. Genes Dev. 13, 1382–1397 (1999).
Lewis, C. E., De Palma, M. & Naldini, L. Tie2-expressing monocytes and tumor angiogenesis: regulation by hypoxia and angiopoietin 2. Cancer Res. 67, 8429–8432 (2007).
Sozzani, S., Rusnati, M., Riboldi, E., Mitola, S. & Presta, M. Dendritic cell-endothelial cell cross-talk in angiogenesis. Trends Immunol. 28, 385–392 (2007).
Noonan, D. M., De Lerma Barbaro, A., Vannini, N., Mortara, L. & Albini, A. Inflammation, inflammatory cells and angiogenesis: decisions and indecisions. Cancer Metastasis Rev. 27, 31–40 (2008).
Dunn, G. P., Old, L. J. & Schreiber, R. D. The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21, 137–148 (2004).
Swann, J. B. et al. Demonstration of inflammation-induced cancer and cancer immunoediting during primary tumorigenesis. Proc. Natl Acad. Sci. USA 105, 652–656 (2008).
de Visser, K. E., Korets, L. V. & Coussens, L. M. De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer Cell 7, 411–423 (2005). This paper shows that in a model of human-papilloma-virus-driven carcinogenesis, adaptive immune responses mediated by B cells coordinate cancer-promoting inflammation.
Beatson, G. On the treatment of inoperable cases of carcinoma of the mamma: suggestions for a new method of treatment with illustrative cases. Lancet 2, 104–162, (1896).
Zhu, P. et al. Macrophage/cancer cell interactions mediate hormone resistance by a nuclear receptor derepression pathway. Cell 124, 615–629 (2006).
Naugler, W. E. et al. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science 317, 121–124 (2007). References 60 and 61 show that two classic pathways of cancer promotion, hormones and inflammation, are linked in both liver cancer and prostate cancer.
Muller, A. et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410, 50–56 (2001).
Burger, J. A. & Kipps, T. J. CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood 107, 1761–1767 (2006).
Kaifi, J. T. et al. Tumor-cell homing to lymph nodes and bone marrow and CXCR4 expression in esophageal cancer. J. Natl Cancer Inst. 97, 1840–1847 (2005).
Salvucci, O. et al. The role of CXCR4 receptor expression in breast cancer: a large tissue microarray study. Breast Cancer Res. Treat. 97, 275–283 (2006).
Kim, J. et al. Chemokine receptor CXCR4 expression in colorectal cancer patients increases the risk for recurrence and for poor survival. J. Clin. Oncol. 23, 2744–2753 (2005).
Shields, J. D. et al. Autologous chemotaxis as a mechanism of tumor cell homing to lymphatics via interstitial flow and autocrine CCR7 signaling. Cancer Cell 11, 526–538 (2007).
Kawada, K. et al. Pivotal role of CXCR3 in melanoma cell metastasis to lymph nodes. Cancer Res. 64, 4010–4017 (2004).
Shulby, S. A., Dolloff, N. G., Stearns, M. E., Meucci, O. & Fatatis, A. CX3CR1-fractalkine expression regulates cellular mechanisms involved in adhesion, migration, and survival of human prostate cancer cells. Cancer Res. 64, 4693–4698 (2004).
Burns, J. M. et al. A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J. Exp. Med. 203, 2201–2213 (2006).
Zipin-Roitman, A. et al. CXCL10 promotes invasion-related properties in human colorectal carcinoma cells. Cancer Res. 67, 3396–3405 (2007).
Ghadjar, P. et al. Chemokine receptor CCR6 expression level and liver metastases in colorectal cancer. J. Clin. Oncol. 24, 1910–1916 (2006).
Kulbe, H. et al. The inflammatory cytokine tumor necrosis factor-α generates an autocrine tumor-promoting network in epithelial ovarian cancer cells. Cancer Res. 67, 585–592 (2007).
Kulbe, H., Hagemann, T., Szlosarek, P. W., Balkwill, F. R. & Wilson, J. L. The inflammatory cytokine tumor necrosis factor-α regulates chemokine receptor expression on ovarian cancer cells. Cancer Res. 65, 10355–10362 (2005).
Bates, R. C. & Mercurio, A. M. Tumor necrosis factor-α stimulates the epithelial-to-mesenchymal transition of human colonic organoids. Mol. Biol. Cell 14, 1790–1800 (2003).
Luo, J. L. et al. Nuclear cytokine-activated IKKα controls prostate cancer metastasis by repressing maspin. Nature 446, 690–694 (2007).
Condeelis, J. & Pollard, J. W. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell 124, 263–266 (2006).
Wyckoff, J. B. et al. Direct visualization of macrophage-assisted tumor cell intravasation in mammary tumors. Cancer Res. 67, 2649–2656 (2007).
Robinson-Smith, T. M. et al. Macrophages mediate inflammation-enhanced metastasis of ovarian tumors in mice. Cancer Res. 67, 5708–5716 (2007).
Hagemann, T. et al. Macrophages induce invasiveness of epithelial cancer cells via NF-κB and JNK. J. Immunol. 175, 1197–1205 (2005).
Marchesi, F. et al. Increased survival, proliferation, and migration in metastatic human pancreatic tumor cells expressing functional CXCR4. Cancer Res. 64, 8420–8427 (2004).
Nickoloff, B. J., Ben-Neriah, Y. & Pikarsky, E. Inflammation and cancer: is the link as simple as we think? J. Invest. Dermatol. 124, x–xiv (2005).
Hagemann, T. et al. Re-educating tumor-associated macrophages by targeting NF-κB. J. Exp. Med. 205, 1261–1268 (2008). This paper shows that NF-κB activated through the IL-1 receptor and MyD88 signalling pathway maintains the phenotype of TAMs, suggesting that tumour-promoting macrophages might be re-educated by the targeting of NF-κB.
Dajee, M. et al. NF-κB blockade and oncogenic Ras trigger invasive human epidermal neoplasia. Nature 421, 639–643 (2003).
Coley, W. B. The treatment of malignant tumors by repeated inoculations of erysipelas: with a report of ten original cases. Am. J. Med. Sci. 105, 487–511 (1893).
Apetoh, L. et al. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nature Med. 13, 1050–1059 (2007).
Sato, Y. et al. IL-10 deficiency leads to somatic mutations in a model of IBD. Carcinogenesis 27, 1068–1073 (2006).
Bielas, J. H., Loeb, K. R., Rubin, B. P., True, L. D. & Loeb, L. A. Human cancers express a mutator phenotype. Proc. Natl Acad. Sci. USA 103, 18238–18242 (2006).
Gungor, N., Godschalk, R. W. L., Pachen, D. M., Van Schooten, F. J. & Knaapen, A. M. Activated neutrophils inhibit nucleotide excision repair in human pulmonary epithelial cells: role of myeloperoxidase. FASEB J. 21, 2359–2367 (2007).
Dally, H. et al. Myeloperoxidase (MPO) genotype and lung cancer histologic types: the MPO −463 A allele is associated with reduced risk for small cell lung cancer in smokers. Int. J. Cancer 102, 530–535 (2002).
Rao, V. P. et al. Innate immune inflammatory response against enteric bacteria Helicobacter hepaticus induces mammary adenocarcinoma in mice. Cancer Res. 66, 7395–7400 (2006).
Mantovani, A. Cancer: an infernal triangle. Nature 448, 547–548 (2007).
Madhusudan, S. et al. Study of etanercept, a tumor necrosis factor-α inhibitor, in recurrent ovarian cancer. J. Clin. Oncol. 23, 5950–5959 (2005).
Brown, E. R. et al. A clinical study assessing the tolerability and biological effects of infliximab, a TNF-α inhibitor, in patients with advanced cancer. Ann. Oncol. 19, 1340–1346 (2008).
Harrison, M. L. et al. Tumor necrosis factor α as a new target for renal cell carcinoma: two sequential phase II trials of infliximab at standard and high dose. J. Clin. Oncol. 25, 4542–4549 (2007). The paper reports the first clinical evidence that TNF-α could be a target for treating renal-cell carcinoma.
Weber, D. M. et al. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N. Engl. J. Med. 357, 2133–2142 (2007).
Bertagnolli, M. M. et al. Celecoxib for the prevention of sporadic colorectal adenomas. N. Engl. J. Med. 355, 873–884 (2006).
Steinbach, G. et al. The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N. Engl. J. Med. 342, 1946–1952 (2000).
Acknowledgements
A.M., P.A. and A.S. are supported by the Italian Association for Cancer Research, the Italian Ministry of Health, the Italian Ministry of Universities and Research, and the European Commission. F.B. is supported by Cancer Research UK, the Medical Research Council, the Association for International Cancer Research and the Higher Education Funding Council for England.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reprints and permissions information is available at http://www.nature.com/reprints.
Correspondence should be addressed to A.M. (alberto.mantovani@humanitas.it).
Rights and permissions
About this article
Cite this article
Mantovani, A., Allavena, P., Sica, A. et al. Cancer-related inflammation. Nature 454, 436–444 (2008). https://doi.org/10.1038/nature07205
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07205
This article is cited by
-
The relationship between tumor-infiltrating neutrophils and clinical outcomes in patients with resectable hepatocellular carcinoma
BMC Cancer (2024)
-
Prognostic and clinicopathological significance of C-reactive protein in patients with ovarian cancer: a meta-analysis
World Journal of Surgical Oncology (2024)
-
Combined score based on plasma fibrinogen and platelet-lymphocyte ratio as a prognostic biomarker in esophageal squamous cell carcinoma
BMC Cancer (2024)
-
Predicting the unpredictable: a robust nomogram for predicting recurrence in patients with ampullary carcinoma
BMC Cancer (2024)
-
Utility and predictive value of the CRAFITY score in advanced hepatocellular carcinoma treated with transarterial chemoembolization plus tyrosine kinase inhibitors and PD-1 inhibitor
BMC Cancer (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.