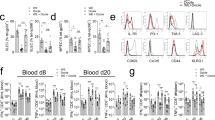
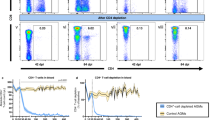
Abstract
For a wide variety of microbial pathogens, the outcome of the infection is indeterminate. In some individuals the microbe is cleared, but in others it establishes a chronic infection, and the factors that tip this balance are often unknown. In a widely used model of chronic viral infection, C57BL/6 mice clear the Armstrong strain of lymphocytic choriomeningitis virus (LCMV), but the clone 13 strain persists1,2. Here we show that the Armstrong strain induces a profound lymphopenia at days 1–3 after infection, but the clone 13 strain does not. If we transiently augment lymphopenia by treating the clone-13-infected mice with the drug FTY720 at days 0–2 after infection, the mice successfully clear the infection by day 30. Clearance does not occur when CD4 T cells are absent at the time of treatment, indicating that the drug is not exerting direct antiviral effects. Notably, FTY720 treatment of an already established persistent infection also leads to viral clearance. In both models, FTY720 treatment preserves or augments LCMV-specific CD4 and CD8 T-cell responses, a result that is counter-intuitive because FTY720 is generally regarded as a new immunosuppressive agent3. Because FTY720 targets host pathways that are completely evolutionarily conserved, our results may be translatable into new immunotherapies for the treatment of chronic microbial infections in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
08 April 2010
A Correction to this paper has been published: https://doi.org/10.1038/nature09030
References
Ahmed, R., Byrne, J. A. & Oldstone, M. B. Virus specificity of cytotoxic T lymphocytes generated during acute lymphocytic choriomeningitis virus infection: role of the H-2 region in determining cross-reactivity for different lymphocytic choriomeningitis virus strains. J. Virol. 51, 34–41 (1984)
Matloubian, M., Concepcion, R. J. & Ahmed, R. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J. Virol. 68, 8056–8063 (1994)
Kunzendorf, U., Ziegler, E. & Kabelitz, D. FTY720–the first compound of a new promising class of immunosuppressive drugs. Nephrol. Dial. Transplant. 19, 1677–1681 (2004)
Chisari, F. V. & Ferrari, C. Hepatitis B virus immunopathology. Springer Semin. Immunopathol. 17, 261–281 (1995)
Day, C. L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006)
Folgori, A. et al. Early impairment of hepatitis C virus specific T cell proliferation during acute infection leads to failure of viral clearance. Gut 55, 1012–1019 (2006)
Wherry, E. J., Blattman, J. N., Murali-Krishna, K., van der Most, R. & Ahmed, R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 77, 4911–4927 (2003)
Shiow, L. R. et al. CD69 acts downstream of interferon-α/β to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature 440, 540–544 (2006)
Mamalaki, C. et al. Positive and negative selection in transgenic mice expressing a T-cell receptor specific for influenza nucleoprotein and endogenous superantigen. Dev. Immunol. 3, 159–174 (1993)
Brinkmann, V. et al. The immune modulator FTY720 targets sphingosine 1-phosphate receptors. J. Biol. Chem. 277, 21453–21457 (2002)
Chiba, K. FTY720, a new class of immunomodulator, inhibits lymphocyte egress from secondary lymphoid tissues and thymus by agonistic activity at sphingosine 1-phosphate receptors. Pharmacol. Ther. 108, 308–319 (2005)
Kappos, L. et al. Oral fingolimod (FTY720) for relapsing multiple sclerosis. N. Engl. J. Med. 355, 1124–1140 (2006)
Budde, K. et al. First human trial of FTY720, a novel immunomodulator, in stable renal transplant patients. J. Am. Soc. Nephrol. 13, 1073–1083 (2002)
Pinschewer, D. D. et al. FTY720 immunosuppression impairs effector T cell peripheral homing without affecting induction, expansion, and memory. J. Immunol. 164, 5761–5770 (2000)
Moskophidis, D., Lechner, F., Pircher, H. & Zinkernagel, R. M. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature 362, 758–761 (1993)
Zajac, A. J. et al. Viral immune evasion due to persistence of activated T cells without effector function. J. Exp. Med. 188, 2205–2213 (1998)
Barber, D. L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006)
Brooks, D. G. et al. Interleukin-10 determines viral clearance or persistence in vivo . Nature Med. 12, 1301–1309 (2006)
Matter, M., Odermatt, B., Yagita, H., Nuoffer, J. M. & Ochsenbein, A. F. Elimination of chronic viral infection by blocking CD27 signaling. J. Exp. Med. 203, 2145–2155 (2006)
Brooks, D. G., Lee, A. M., Elsaesser, H., McGavern, D. B. & Oldstone, M. B. IL-10 blockade facilitates DNA vaccine-induced T cell responses and enhances clearance of persistent virus infection. J. Exp. Med. 205, 533–541 (2008)
Ha, S. J. et al. Enhancing therapeutic vaccination by blocking PD-1-mediated inhibitory signals during chronic infection. J. Exp. Med. 205, 543–555 (2008)
Muller, S. et al. Role of an intact splenic microarchitecture in early lymphocytic choriomeningitis virus production. J. Virol. 76, 2375–2383 (2002)
Altman, J. D. et al. Phenotypic analysis of antigen-specific T lymphocytes. Science 274, 94–96 (1996)
Cembrzynska-Nowak, M. Different antiviral activity and cell specificity of interferon preparations produced by mouse peritoneal cells at 37 degrees C and at 26 degrees C. Arch. Immunol. Ther. Exp. (Warsz.) 37, 499–502 (1989)
Merrill, A. H., Sullards, M. C., Allegood, J. C., Kelly, S. & Wang, E. Sphingolipidomics: high-throughput, structure-specific, and quantitative analysis of sphingolipids by liquid chromatography tandem mass spectrometry. Methods 36, 207–224 (2005)
Acknowledgements
We would like to thank R. Ahmed for his support and guidance during the project; D. Liotta for the use of his mass spectrometer; and the members of the Ahmed and the Altman laboratories for discussions. This work was supported by National Institute of Health grant numbers AI042373 (to J.D.A.) and 5F32AI062002 (M.P.-L.).
Author Contributions M.P.-L. originated and designed the project, analysed the results and co-wrote the manuscript with J.D.A. who also provided guidance and support throughout the project. N.B.M. performed viral load assays. S.T.P. measured S1P levels. P.A.R. was involved in preliminary experiments.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary information
The file contains Supplementary Figures 1-7 with Legends. (PDF 2588 kb)
Rights and permissions
About this article
Cite this article
Premenko-Lanier, M., Moseley, N., Pruett, S. et al. Transient FTY720 treatment promotes immune-mediated clearance of a chronic viral infection. Nature 454, 894–898 (2008). https://doi.org/10.1038/nature07199
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature07199
This article is cited by
-
FTY720 mediates activation suppression and G0/G1 cell cycle arrest in a concanavalin A-induced mouse lymphocyte pan-activation model
Inflammation Research (2012)
-
Closing in on an oral treatment
Nature (2010)
-
Retraction Note: Transient FTY720 treatment promotes immune-mediated clearance of a chronic viral infection
Nature (2010)
-
Lysophosphatidic acid and autotaxin: emerging roles in innate and adaptive immunity
Immunologic Research (2009)
-
Surprising side effects
Nature (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.