Abstract
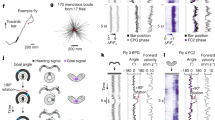
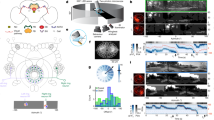
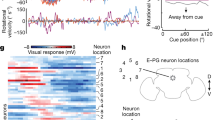
Flexible goal-driven orientation requires that the position of a target be stored, especially in case the target moves out of sight. The capability to retain, recall and integrate such positional information into guiding behaviour has been summarized under the term spatial working memory1. This kind of memory contains specific details of the presence that are not necessarily part of a long-term memory. Neurophysiological studies in primates2 indicate that sustained activity of neurons encodes the sensory information even though the object is no longer present. Furthermore they suggest that dopamine transmits the respective input to the prefrontal cortex, and simultaneous suppression by GABA spatially restricts this neuronal activity3. Here we show that Drosophila melanogaster possesses a similar spatial memory during locomotion. Using a new detour setup, we show that flies can remember the position of an object for several seconds after it has been removed from their environment. In this setup, flies are temporarily lured away from the direction towards their hidden target, yet they are thereafter able to aim for their former target. Furthermore, we find that the GABAergic (stainable with antibodies against GABA) ring neurons4 of the ellipsoid body in the central brain are necessary and their plasticity is sufficient for a functional spatial orientation memory in flies. We also find that the protein kinase S6KII (ignorant)5 is required in a distinct subset of ring neurons to display this memory. Conditional expression of S6KII in these neurons only in adults can restore the loss of the orientation memory of the ignorant mutant. The S6KII signalling pathway therefore seems to be acutely required in the ring neurons for spatial orientation memory in flies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Postle, B. R. Working memory as an emergent property of the mind and brain. Neuroscience 139, 23–38 (2006)
Chafee, M. V. & Goldman-Rakic, P. S. Matching patterns of activity in primate prefrontal area 8a and parietal area 7ip neurons during a spatial working memory task. J. Neurophysiol. 79, 2919–2940 (1998)
Williams, G. V. & Castner, S. A. Prefrontal cortex and working memory processes. Neuroscience 139, 251–261 (2006)
Hanesch, U., Fischbach, K.-F. & Heisenberg, M. Neuronal architecture of the central complex in Drosophila melanogaster . Cell Tissue Res. 257, 343–366 (1998)
Putz, G., Bertolucci, F., Raabe, T., Zars, T. & Heisenberg, M. The S6KII (rsk) gene of Drosophila melanogaster differentially affects an operant and a classical learning task. J. Neurosci. 24, 9745–9751 (2004)
Strauss, R. & Pichler, J. Persistence of orientation toward a temporarily invisible landmark in Drosophila melanogaster . J. Comp. Physiol. A 182, 411–423 (1998)
Strauss, R., Schuster, S. & Götz, K. G. Processing of artificial visual feedback in the walking fruit fly Drosophila melanogaster . J. Exp. Biol. 200, 1281–1296 (1997)
Bülthoff, H., Götz, K. G. & Herre, M. Recurrent inversion of visual orientation in the walking fly, Drosophila melanogaster . J. Comp. Physiol. A 148, 471–481 (1982)
Collett, T. S. & Collett, M. Memory use in insect visual navigation. Nature Rev. Neurosci. 3, 542–552 (2002)
Strauss, R. & Heisenberg, M. A higher control center of locomotor behavior in the Drosophila brain. J. Neurosci. 13, 1852–1861 (1993)
Strauss, R. The central complex and the genetic dissection of locomotor behaviour. Curr. Opin. Neurobiol. 12, 633–638 (2002)
De Belle, J. S. & Heisenberg, M. Associative odor learning in Drosophila abolished by chemical ablation of mushroom bodies. Science 263, 692–695 (1994)
Renn, S. C. et al. Genetic analysis of the Drosophila ellipsoid body neuropil: organization and development of the central complex. J. Neurobiol. 41, 189–207 (1999)
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993)
Sweeney, S. T., Broadie, K., Keane, J., Niemann, H. & ÒKane, C. J. Targeted expression of tetanus toxin light chain in Drosophila specifically eliminates synaptic transmission and causes behavioral defects. Neuron 14, 341–351 (1995)
McGuire, S. E., Le, P. T., Osborn, A. J., Matsumoto, K. & Davis, R. L. Spatiotemporal rescue of memory dysfunction in Drosophila . Science 302, 1765–1768 (2003)
McGuire, S. E., Deshazer, M. & Davis, R. L. Thirty years of olfactory learning and memory research in Drosophila melanogaster . Prog. Neurobiol. 76, 328–347 (2005)
Liu, G. et al. Distinct memory traces for two visual features in the Drosophila brain. Nature 439, 551–556 (2006)
Davis, R. L. & Kiger, J. A. Dunce mutants of Drosophila melanogaster: mutants defective in the cyclic AMP phosphodiesterase enzyme system. J. Cell Biol. 90, 101–107 (1981)
Dudai, Y., Jan, Y. N., Byers, D., Quinn, W. G. & Benzer, S. dunce, a mutant of Drosophila deficient in learning. Proc. Natl Acad. Sci. USA 73, 1684–1688 (1976)
Wustmann, G., Rein, K., Wolf, R. & Heisenberg, M. A new paradigm for operant conditioning of Drosophila melanogaster . J. Comp. Physiol. A 179, 429–436 (1996)
Kim, M. et al. Inhibition of ERK-MAP kinase signaling by RSK during Drosophila development. EMBO J. 25, 3056–3067 (2006)
Myers, A. P., Corson, L. B., Rossant, J. & Baker, J. C. Characterization of mouse Rsk4 as an inhibitor of fibroblast growth factor-RAS-extracellular signal-regulated kinase signaling. Mol. Cell. Biol. 24, 4255–4266 (2004)
Dietzl, G. et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila . Nature 448, 151–156 (2007)
Torroja, L., Chu, H., Kotovsky, I. & White, K. Neuronal overexpression of APPL, the Drosophila homologue of the amyloid precursor protein (APP), disrupts axonal transport. Curr. Biol. 9, 489–492 (1999)
Luo, L., Liao, Y. J., Jan, L. Y. & Jan, Y. N. Distinct morphogenetic functions of similar small GTPases: Drosophila Drac1 is involved in axonal outgrowth and myoblast fusion. Genes Dev. 8, 1787–1802 (1994)
Kim, Y. C., Lee, H. G., Seong, C. S. & Han, K. A. Expression of a D1 dopamine receptor dDA1/DmDOP1 in the central nervous system of Drosophila melanogaster . Gene Expr. Patterns 3, 237–245 (2003)
Mronz, M. Die visuell motivierte Objektwahl laufender Taufliegen (Drosophila melanogaster)–Verhaltensphysiologie, Modellbildung und Implementierung in einem Roboter. PhD thesis, Univ. Würzburg. (2004)
Heisenberg, M. & Boehl, K. Isolation of anatomical brain mutants of Drosophila by histological means. Z. Naturforsch. C 34, 143–147 (1979)
Botella, J. A. et al. Deregulation of the Egfr/Ras signaling pathway induces age-related brain degeneration in the Drosophila mutant vap . Mol. Biol. Cell 14, 241–250 (2003)
Acknowledgements
We thank M. Heisenberg for discussions and continuous support, D. Kretzschmar for reading the manuscript, and E. Stepien-Bötsch for experimental contributions. This work was supported by the Deutsche Forschungsgemeinschaft (SFB 554-B7, GRK 1156).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
The file contains Supplementary Figures S1-S3 and Legends and Supplementary Table S1 on Statistics
The file shows additional data on the persistence of orientation in mutant lines with structural central complex defects (Figure S1), immunohistological preparations showing no thoracic expression of GAL4-lines c232, c481 and c105 (Figure S2), the locomotor and orientation behaviour of wild-type Canton-S and ign58/1 mutant flies in Buridan`s paradigm (Figure S3), and Supplementary Table S1 on statistics. (PDF 231 kb)
The file contains Supplementary Movie 1 which shows wild-type Canton-S male performing the detour paradigm in real-time
The fly (arrow) approaches the left stripe. The initial target automatically disappears, while laterally to the fly (in the upper half of the screen) another vertical stripe appears as distracter. As soon as the fly has turned toward this distracter with less than 15° deviation, the distracter will disappear as well within 1s. The fly reorients towards the position of its former target and eventually shows random search behaviour. (MOV 15310 kb)
Rights and permissions
About this article
Cite this article
Neuser, K., Triphan, T., Mronz, M. et al. Analysis of a spatial orientation memory in Drosophila. Nature 453, 1244–1247 (2008). https://doi.org/10.1038/nature07003
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature07003
This article is cited by
-
Localization of SNARE proteins in the brain and corpus allatum of Bombyx mori
Histochemistry and Cell Biology (2023)
-
Lineages to circuits: the developmental and evolutionary architecture of information channels into the central complex
Journal of Comparative Physiology A (2023)
-
Impact of central complex lesions on innate and learnt visual navigation in ants
Journal of Comparative Physiology A (2023)
-
Differential mechanisms underlie trace and delay conditioning in Drosophila
Nature (2022)
-
Thermoresponsive motor behavior is mediated by ring neuron circuits in the central complex of Drosophila
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.