Abstract
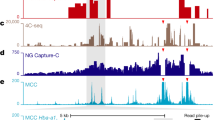
The architecture of human chromosomes in interphase nuclei is still largely unknown. Microscopy studies have indicated that specific regions of chromosomes are located in close proximity to the nuclear lamina (NL)1,2,3. This has led to the idea that certain genomic elements may be attached to the NL, which may contribute to the spatial organization of chromosomes inside the nucleus. However, sequences in the human genome that interact with the NL in vivo have not been identified. Here we construct a high-resolution map of the interaction sites of the entire genome with NL components in human fibroblasts. This map shows that genome–lamina interactions occur through more than 1,300 sharply defined large domains 0.1–10 megabases in size. These lamina-associated domains (LADs) are typified by low gene-expression levels, indicating that LADs represent a repressive chromatin environment. The borders of LADs are demarcated by the insulator protein CTCF, by promoters that are oriented away from LADs, or by CpG islands, suggesting possible mechanisms of LAD confinement. Taken together, these results demonstrate that the human genome is divided into large, discrete domains that are units of chromosome organization within the nucleus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
Data deposits
Microarray probe coordinates and data have been submitted to the Gene Expression Omnibus (http://www.ncbi.nlm.nih.gov/geo/) under accession number GSE8854.
Change history
12 June 2013
Nature 453, 948–951 (2008); doi: 10.1038/nature06947 In this Letter, Fig. 4g and Supplementary Fig. 5f depict an analysis of H3K9me3 (not H3K9me2) ChIP data. In addition, three sentences in the third paragraph of the ‘Microarray data analysis’ section of the Methods should read: “Gene expression data and H3K27me3 ChIP data are from Tig3 lung fibroblasts18.
References
Misteli, T. Beyond the sequence: cellular organization of genome function. Cell 128, 787–800 (2007)
Akhtar, A. & Gasser, S. M. The nuclear envelope and transcriptional control. Nature Rev. Genet. 8, 507–517 (2007)
Taddei, A., Hediger, F., Neumann, F. R. & Gasser, S. M. The function of nuclear architecture: a genetic approach. Annu. Rev. Genet. 38, 305–345 (2004)
Pickersgill, H. et al. Characterization of the Drosophila melanogaster genome at the nuclear lamina. Nature Genet. 38, 1005–1014 (2006)
Vogel, M. J., Peric-Hupkes, D. & van Steensel, B. Detection of in vivo protein–DNA interactions using DamID in mammalian cells. Nature Protocols 2, 1467–1478 (2007)
Manilal, S., Nguyen, T. M., Sewry, C. A. & Morris, G. E. The Emery–Dreifuss muscular dystrophy protein, emerin, is a nuclear membrane protein. Hum. Mol. Genet. 5, 801–808 (1996)
Bengtsson, L. & Wilson, K. L. Multiple and surprising new functions for emerin, a nuclear membrane protein. Curr. Opin. Cell Biol. 16, 73–79 (2004)
Bolzer, A. et al. Three-dimensional maps of all chromosomes in human male fibroblast nuclei and prometaphase rosettes. PLoS Biol. 3, e157 (2005)
Croft, J. A. et al. Differences in the localization and morphology of chromosomes in the human nucleus. J. Cell Biol. 145, 1119–1131 (1999)
Goetze, S. et al. The three-dimensional structure of human interphase chromosomes is related to the transcriptome map. Mol. Cell. Biol. 27, 4475–4487 (2007)
Gilchrist, S., Gilbert, N., Perry, P. & Bickmore, W. A. Nuclear organization of centromeric domains is not perturbed by inhibition of histone deacetylases. Chromosome Res. 12, 505–516 (2004)
Wiblin, A. E., Cui, W., Clark, A. J. & Bickmore, W. A. Distinctive nuclear organisation of centromeres and regions involved in pluripotency in human embryonic stem cells. J. Cell Sci. 118, 3861–3868 (2005)
Boyle, S. et al. The spatial organization of human chromosomes within the nuclei of normal and emerin-mutant cells. Hum. Mol. Genet. 10, 211–219 (2001)
Murmann, A. E. et al. Local gene density predicts the spatial position of genetic loci in the interphase nucleus. Exp. Cell Res. 311, 14–26 (2005)
Ovcharenko, I. et al. Evolution and functional classification of vertebrate gene deserts. Genome Res. 15, 137–145 (2005)
Lanctot, C. et al. Dynamic genome architecture in the nuclear space: regulation of gene expression in three dimensions. Nature Rev. Genet. 8, 104–115 (2007)
Berger, S. L. The complex language of chromatin regulation during transcription. Nature 447, 407–412 (2007)
Bracken, A. P. et al. Genome-wide mapping of Polycomb target genes unravels their roles in cell fate transitions. Genes Dev. 20, 1123–1136 (2006)
Kim, T. H. et al. Analysis of the vertebrate insulator protein CTCF-binding sites in the human genome. Cell 128, 1231–1245 (2007)
Gaszner, M. & Felsenfeld, G. Insulators: exploiting transcriptional and epigenetic mechanisms. Nature Rev. Genet. 7, 703–713 (2006)
Yusufzai, T. M., Tagami, H., Nakatani, Y. & Felsenfeld, G. CTCF tethers an insulator to subnuclear sites, suggesting shared insulator mechanisms across species. Mol. Cell 13, 291–298 (2004)
Xu, X. et al. A comprehensive ChIP-chip analysis of E2F1, E2F4, and E2F6 in normal and tumor cells reveals interchangeable roles of E2F family members. Genome Res. 17, 1550–1561 (2007)
Imai, S. et al. Dissociation of Oct-1 from the nuclear peripheral structure induces the cellular aging-associated collagenase gene expression. Mol. Biol. Cell 8, 2407–2419 (1997)
Parada, L. A., Roix, J. J. & Misteli, T. An uncertainty principle in chromosome positioning. Trends Cell Biol. 13, 393–396 (2003)
Bridger, J. M., Boyle, S., Kill, I. R. & Bickmore, W. A. Re-modelling of nuclear architecture in quiescent and senescent human fibroblasts. Curr. Biol. 10, 149–152 (2000)
Masny, P. S. et al. Localization of 4q35.2 to the nuclear periphery: is FSHD a nuclear envelope disease? Hum. Mol. Genet. 13, 1857–1871 (2004)
Reddy, K. L., Zullo, J. M., Bertolino, E. & Singh, H. Transcriptional repression mediated by repositioning of genes to the nuclear lamina. Nature 452, 243–247 (2008)
Kumaran, R. I. & Spector, D. L. A genetic locus targeted to the nuclear periphery in living cells maintains its transcriptional competence. J. Cell Biol. 180, 51–65 (2008)
Weber, M. et al. Distribution, silencing potential and evolutionary impact of promoter DNA methylation in the human genome. Nature Genet. 39, 457–466 (2007)
O’Geen, H. et al. Genome-wide analysis of KAP1 binding suggests autoregulation of KRAB-ZNFs. PLoS Genet. 3, e89 (2007)
Lenhard, B. & Wasserman, W. W. TFBS: Computational framework for transcription factor binding site analysis. Bioinformatics 18, 1135–1136 (2002)
Wingender, E. et al. TRANSFAC: an integrated system for gene expression regulation. Nucleic Acids Res. 28, 316–319 (2000)
van Steensel, B. et al. Localization of the glucocorticoid receptor in discrete clusters in the cell nucleus. J. Cell Sci. 108, 3003–3011 (1995)
Solovei, I. et al. Spatial preservation of nuclear chromatin architecture during three-dimensional fluorescence in situ hybridization (3D-FISH). Exp. Cell Res. 276, 10–23 (2002)
Simonis, M. et al. Nuclear organization of active and inactive chromatin domains uncovered by chromosome conformation capture-on-chip (4C). Nature Genet. 38, 1348–1354 (2006)
Acknowledgements
We thank R. Versteeg, M. Indemans, R. van Driel and O. Giromus for help with FISH; L. Oomen for help with confocal microscopy; A. Bracken and K. Blahnik for data sets; the NKI microarray facility for technical support; E. de Wit for preliminary data analysis; and M. Fornerod, F. van Leeuwen, H.J. Bussemaker, M. van Lohuizen and members of the B.v.S. laboratory for helpful suggestions. This study was supported by the Netherlands Cancer Society, the Netherlands Genomics Initiative, and an European Young Investigator Award to B.v.S. E.B. was supported by NWO Rubicon.
Author Contributions L.G. performed DamID and related experiments; M.B.F. and L.G. performed immunofluorescence microscopy studies; E.B. conducted FISH experiments in the W.d.L. laboratory, with technical support from L.G., B.H.E., A.d.K. and W.T.; L.P. designed microarrays; L.P. and B.v.S. performed data analysis together with L.G.; W.M. and L.W. developed motif and repeat search algorithms. L.G. and B.v.S. conceived and designed the study and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures 1-8
The file contains Supplementary Figures 1-8 and Legends. This file contains a cartoon model summarizing the main results (Supplementary Figure 1) and seven figures showing supporting data and analyses (Supplementary Figures 2-8). (PDF 5445 kb)
Supplementary Data
The file contains Supplementary Data. This file is a flat text file in GFF format (http://www.sanger.ac.uk/Software/formats/GFF/) listing the positions of all 1,344 LADs (NCBI build 36). Score (column 6) indicates the fraction of array probes inside the LAD with a positive Lamin B1 DamID logratio, after applying a running median filter with window size 5. (TXT 119 kb)
Rights and permissions
About this article
Cite this article
Guelen, L., Pagie, L., Brasset, E. et al. Domain organization of human chromosomes revealed by mapping of nuclear lamina interactions. Nature 453, 948–951 (2008). https://doi.org/10.1038/nature06947
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06947
This article is cited by
-
Protein kinase C activity modulates nuclear Lamin A/C dynamics in HeLa cells
Scientific Reports (2024)
-
Computational methods for analysing multiscale 3D genome organization
Nature Reviews Genetics (2024)
-
Transposable elements as essential elements in the control of gene expression
Mobile DNA (2023)
-
Detection and characterization of constitutive replication origins defined by DNA polymerase epsilon
BMC Biology (2023)
-
Strong interactions between highly dynamic lamina-associated domains and the nuclear envelope stabilize the 3D architecture of Drosophila interphase chromatin
Epigenetics & Chromatin (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.