Abstract
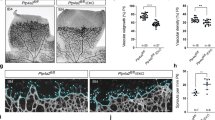
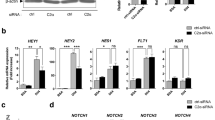
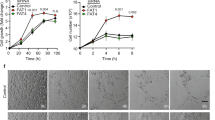
Phosphoinositide 3-kinases (PI3Ks) signal downstream of multiple cell-surface receptor types. Class IA PI3K isoforms1 couple to tyrosine kinases and consist of a p110 catalytic subunit (p110α, p110β or p110δ), constitutively bound to one of five distinct p85 regulatory subunits. PI3Ks have been implicated in angiogenesis2,3,4,5, but little is known about potential selectivity among the PI3K isoforms and their mechanism of action in endothelial cells during angiogenesis in vivo. Here we show that only p110α activity is essential for vascular development. Ubiquitous or endothelial cell-specific inactivation of p110α led to embryonic lethality at mid-gestation because of severe defects in angiogenic sprouting and vascular remodelling. p110α exerts this critical endothelial cell-autonomous function by regulating endothelial cell migration through the small GTPase RhoA. p110α activity is particularly high in endothelial cells and preferentially induced by tyrosine kinase ligands (such as vascular endothelial growth factor (VEGF)-A). In contrast, p110β in endothelial cells signals downstream of G-protein-coupled receptor (GPCR) ligands such as SDF-1α, whereas p110δ is expressed at low level and contributes only minimally to PI3K activity in endothelial cells. These results provide the first in vivo evidence for p110-isoform selectivity in endothelial PI3K signalling during angiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Vanhaesebroeck, B., Leevers, S. J., Panayotou, G. & Waterfield, M. D. Phosphoinositide 3-kinases: a conserved family of signal transducers. Trends Biochem. Sci. 22, 267–272 (1997)
Bi, L. et al. Proliferative defect and embryonic lethality in mice homozygous for a deletion in the p110alpha subunit of phosphoinositide 3-kinase. J. Biol. Chem. 274, 10963–10968 (1999)
Jiang, B. H., Zheng, J. Z., Aoki, M. & Vogt, P. K. Phosphatidylinositol 3-kinase signaling mediates angiogenesis and expression of vascular endothelial growth factor in endothelial cells. Proc. Natl Acad. Sci. USA 97, 1749–1753 (2000)
Geng, L. et al. A specific antagonist of the p110δ catalytic component of phosphatidylinositol 3′-kinase, IC486068, enhances radiation-induced tumor vascular destruction. Cancer Res. 64, 4893–4899 (2004)
Puri, K. D. et al. Mechanisms and implications of phosphoinositide 3-kinaseδ in promoting neutrophil trafficking into inflamed tissue. Blood 103, 3448–3456 (2004)
Vanhaesebroeck, B. et al. P110delta, a novel phosphoinositide 3-kinase in leukocytes. Proc. Natl Acad. Sci. USA 94, 4330–4335 (1997)
Chantry, D. et al. p110delta, a novel phosphatidylinositol 3-kinase catalytic subunit that associates with p85 and is expressed predominantly in leukocytes. J. Biol. Chem. 272, 19236–19241 (1997)
Sawyer, C. et al. Regulation of breast cancer cell chemotaxis by the phosphoinositide 3-kinase p110δ. Cancer Res. 63, 1667–1675 (2003)
Lelievre, E. et al. Deficiency in the p110alpha subunit of PI3K results in diminished Tie2 expression and Tie2(-/-)-like vascular defects in mice. Blood 105, 3935–3938 (2005)
Ueki, K., Algenstaedt, P., Mauvais-Jarvis, F. & Kahn, C. R. Positive and negative regulation of phosphoinositide 3-kinase-dependent signaling pathways by three different gene products of the p85α regulatory subunit. Mol. Cell. Biol. 20, 8035–8046 (2000)
Foukas, L. C. et al. Critical role for the p110α phosphoinositide-3-OH kinase in growth and metabolic regulation. Nature 441, 366–370 (2006)
Lucitti, J. L. et al. Vascular remodeling of the mouse yolk sac requires hemodynamic force. Development 134, 3317–3326 (2007)
Kisanuki, Y. Y. et al. Tie2-Cre transgenic mice: a new model for endothelial cell-lineage analysis in vivo. Dev. Biol. 230, 230–242 (2001)
Bi, L., Okabe, I., Bernard, D. J. & Nussbaum, R. L. Early embryonic lethality in mice deficient in the p110β catalytic subunit of PI 3-kinase. Mamm. Genome 13, 169–172 (2002)
Guillermet-Guibert, J. et al. The p110β isoform of phosphoinositide 3-kinase signals downstream of G protein-coupled receptors and is functionally redundant with p110γ. Proc. Natl Acad. Sci. USA (in the press)
Okkenhaug, K. et al. Impaired B and T cell antigen receptor signaling in p110δ PI 3-kinase mutant mice. Science 297, 1031–1034 (2002)
Dayanir, V., Meyer, R. D., Lashkari, K. & Rahimi, N. Identification of tyrosine residues in vascular endothelial growth factor receptor-2/FLK-1 involved in activation of phosphatidylinositol 3-kinase and cell proliferation. J. Biol. Chem. 276, 17686–17692 (2001)
Gille, H. et al. A repressor sequence in the juxtamembrane domain of Flt-1 (VEGFR-1) constitutively inhibits vascular endothelial growth factor-dependent phosphatidylinositol 3′-kinase activation and endothelial cell migration. EMBO J. 19, 4064–4073 (2000)
Gerber, H. P., Dixit, V. & Ferrara, N. Vascular endothelial growth factor induces expression of the antiapoptotic proteins Bcl-2 and A1 in vascular endothelial cells. J. Biol. Chem. 273, 13313–13316 (1998)
Adini, I. et al. RhoB controls Akt trafficking and stage-specific survival of endothelial cells during vascular development. Genes Dev. 17, 2721–2732 (2003)
Gerhardt, H. et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell Biol. 161, 1163–1177 (2003)
Ruhrberg, C. et al. Spatially restricted patterning cues provided by heparin-binding VEGF-A control blood vessel branching morphogenesis. Genes Dev. 16, 2684–2698 (2002)
Gerhardt, H. & Betsholtz, C. How do endothelial cells orientate? EXS 94, 3–15 (2005)
Worthylake, R. A., Lemoine, S., Watson, J. M. & Burridge, K. RhoA is required for monocyte tail retraction during transendothelial migration. J. Cell Biol. 154, 147–160 (2001)
Hughes, S. & Chan-Ling, T. Roles of endothelial cell migration and apoptosis in vascular remodeling during development of the central nervous system. Microcirculation 7, 317–333 (2000)
Bilancio, A. et al. Key role of the p110delta isoform of PI3K in B-cell antigen and IL-4 receptor signaling: comparative analysis of genetic and pharmacologic interference with p110δ function in B cells. Blood 107, 642–650 (2006)
Lidington, E. A. et al. Conditional immortalization of growth factor-responsive cardiac endothelial cells from H-2K(b)-tsA58 mice. Am. J. Physiol. Cell Physiol. 282, C67–C74 (2002)
Weis, S. et al. Src blockade stabilizes a Flk/cadherin complex, reducing edema and tissue injury following myocardial infarction. J. Clin. Invest. 113, 885–894 (2004)
Cutillas, P. R. et al. Ultrasensitive and absolute quantification of the phosphoinositide 3-kinase/Akt signal transduction pathway by mass spectrometry. Proc. Natl Acad. Sci. USA 103, 8959–8964 (2006)
Wells, C. M. & Ridley, A. J. Analysis of cell migration using the Dunn chemotaxis chamber and time-lapse microscopy. Methods Mol. Biol. 294, 31–41 (2005)
Ren, X. D. & Schwartz, M. A. Determination of GTP loading on Rho. Methods Enzymol. 325, 264–272 (2000)
Acknowledgements
We thank F. Ramadani and K. Okkenhaug (Babraham Institute, Cambridge), E. Cernuda (Hospital Universitario Central de Asturias), T. Makinen (Cancer Research UK London Research Institute), K. Hodivala-Dilke, A. Reynolds and G. D’Amico (Institute of Cancer, Queen Mary, University of London), P. Villalonga (Universitat de les Illes Balears, Spain) and members of the Vanhaesebroeck laboratory (especially N. Osborne, C. See and M. Whitehead) for help and advice, E. Wagner (Research Institute of Molecular Pathology, Vienna), E. Dejana (Institute of Molecular Oncology, Milan), G. Balconi (Mario Negri Institute for Pharmacological Research, Milan), M. Yanagisawa (University of Texas Southwestern Medical Center, Dallas), D. Vestweber (Max-Planck Institute, Muenster), C. Rommel, M. Camps and T. Ruckle (Merck-Serono, Geneva) and Piramed (Slough, UK) for mice and reagents. Personal support was from EMBO (M.G., J.G.-G.), Cancer Research UK (M.G.) and the Fondation pour la Recherche Médicale and the European Union Marie Curie (J.G.-G.). Work in the Vanhaesebroeck laboratory was supported by the Ludwig Institute for Cancer Research Institute, the Biotechnology and Biological Sciences Research Council (BB/C505659/1), the Association for International Cancer Research, European Union (FP6-502935), Cancer Research UK and Barts and the London Charity. R.J.C. is supported by an Association for International Cancer Research grant to A.J.R. (07-0173). L.-K.P. and H.G. are supported by Cancer Research UK.
Author Contributions All authors designed research and analysed data. M.G., J.G.-G., L.C.F., L.-K.P., R.J.C., A.S., W.P., S.M. and P.R.C. performed research. M.G., H.G. and B.V. wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
B.V. is a consultant for PIramed Pharma.
Supplementary information
Supplementary information
The file contains Supplementary Methods with additional references and Supplementary Figures S1-S29 with Legends. (PDF 30710 kb)
Rights and permissions
About this article
Cite this article
Graupera, M., Guillermet-Guibert, J., Foukas, L. et al. Angiogenesis selectively requires the p110α isoform of PI3K to control endothelial cell migration. Nature 453, 662–666 (2008). https://doi.org/10.1038/nature06892
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06892
This article is cited by
-
Analysis of the Molecular Mechanism of Xueshuantong in the Treatment of Wet Age-Related Macular Degeneration (AMD) Using GEO Datasets, Network Pharmacology, and Molecular Docking
Biochemical Genetics (2024)
-
Targeted next-generation sequencing for detection of PIK3CA mutations in archival tissues from patients with Klippel–Trenaunay syndrome in an Asian population
Orphanet Journal of Rare Diseases (2023)
-
Pharmacokinetics and Pharmacodynamic of Alpelisib
Clinical Pharmacokinetics (2023)
-
Phosphoinositides as membrane organizers
Nature Reviews Molecular Cell Biology (2022)
-
When, where and which PIK3CA mutations are pathogenic in congenital disorders
Nature Cardiovascular Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.