Abstract
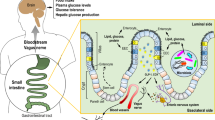
Energy and glucose homeostasis are regulated by food intake and liver glucose production, respectively. The upper intestine has a critical role in nutrient digestion and absorption. However, studies indicate that upper intestinal lipids inhibit food intake as well in rodents and humans by the activation of an intestine–brain axis1,2,3,4. In parallel, a brain–liver axis has recently been proposed to detect blood lipids to inhibit glucose production in rodents5. Thus, we tested the hypothesis that upper intestinal lipids activate an intestine–brain–liver neural axis to regulate glucose homeostasis. Here we demonstrate that direct administration of lipids into the upper intestine increased upper intestinal long-chain fatty acyl-coenzyme A (LCFA-CoA) levels and suppressed glucose production. Co-infusion of the acyl-CoA synthase inhibitor triacsin C or the anaesthetic tetracaine with duodenal lipids abolished the inhibition of glucose production, indicating that upper intestinal LCFA-CoAs regulate glucose production in the preabsorptive state. Subdiaphragmatic vagotomy or gut vagal deafferentation interrupts the neural connection between the gut and the brain, and blocks the ability of upper intestinal lipids to inhibit glucose production. Direct administration of the N-methyl-d-aspartate ion channel blocker MK-801 into the fourth ventricle or the nucleus of the solitary tract where gut sensory fibres terminate abolished the upper-intestinal-lipid-induced inhibition of glucose production. Finally, hepatic vagotomy negated the inhibitory effects of upper intestinal lipids on glucose production. These findings indicate that upper intestinal lipids activate an intestine–brain–liver neural axis to inhibit glucose production, and thereby reveal a previously unappreciated pathway that regulates glucose homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Badman, M. K. & Flier, J. S. The gut and energy balance: visceral allies in the obesity wars. Science 307, 1909–1914 (2005)
Cummings, D. E. & Overduin, J. Gastrointestinal regulation of food intake. J. Clin. Invest. 117, 13–23 (2007)
Moran, T. H. & Schwartz, G. J. Neurobiology of cholecystokinin. Crit. Rev. Neurobiol. 9, 1–28 (1994)
Murphy, K. G. & Bloom, S. R. Gut hormones and the regulation of energy homeostasis. Nature 444, 854–859 (2006)
Lam, T. K. et al. Hypothalamic sensing of circulating fatty acids is required for glucose homeostasis. Nature Med. 11, 320–327 (2005)
Greenberg, D., Smith, G. P. & Gibbs, J. Intraduodenal infusions of fats elicit satiety in sham-feeding rats. Am. J. Physiol. 259, R110–R118 (1990)
Matzinger, D. et al. The role of long chain fatty acids in regulating food intake and cholecystokinin release in humans. Gut 46, 689–693 (2000)
Monnikes, H. et al. Pathways of Fos expression in locus ceruleus, dorsal vagal complex, and PVN in response to intestinal lipid. Am. J. Physiol. 273, R2059–R2071 (1997)
Sclafani, A., Ackroff, K. & Schwartz, G. J. Selective effects of vagal deafferentation and celiac-superior mesenteric ganglionectomy on the reinforcing and satiating action of intestinal nutrients. Physiol. Behav. 78, 285–294 (2003)
Minokoshi, Y. et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428, 569–574 (2004)
Lam, T. K., Schwartz, G. J. & Rossetti, L. Hypothalamic sensing of fatty acids. Nature Neurosci. 8, 579–584 (2005)
Cota, D. et al. Hypothalamic mTOR signaling regulates food intake. Science 312, 927–930 (2006)
Schwartz, M. W., Woods, S. C., Porte, D., Seeley, R. J. & Baskin, D. G. Central nervous system control of food intake. Nature 404, 661–671 (2000)
Coll, A. P., Farooqi, I. S. & O’Rahilly, S. The hormonal control of food intake. Cell 129, 251–262 (2007)
Coppari, R. et al. The hypothalamic arcuate nucleus: A key site for mediating leptin’s effects on glucose homeostasis and locomotor activity. Cell Metab. 1, 63–72 (2005)
Bence, K. K. et al. Neuronal PTP1B regulates body weight, adiposity and leptin action. Nature Med. 12, 917–924 (2006)
Lam, T. K., Gutierrez-Juarez, R., Pocai, A. & Rossetti, L. Regulation of blood glucose by hypothalamic pyruvate metabolism. Science 309, 943–947 (2005)
Schwartz, M. W. & Porte, D. Diabetes, obesity, and the brain. Science 307, 375–379 (2005)
Lam, T. K. et al. Brain glucose metabolism controls the hepatic secretion of triglyceride-rich lipoproteins. Nature Med. 13, 171–180 (2007)
Obici, S., Feng, Z., Arduini, A., Conti, R. & Rossetti, L. Inhibition of hypothalamic carnitine palmitoyltransferase-1 decreases food intake and glucose production. Nature Med. 9, 756–761 (2003)
Greenberg, D., Kava, R. A., Lewis, D. R., Greenwood, M. R. & Smith, G. P. Time course for entry of intestinally infused lipids into blood of rats. Am. J. Physiol. 269, R432–R436 (1995)
Aicher, S. A., Sharma, S. & Pickel, V. M. N-methyl-d-aspartate receptors are present in vagal afferents and their dendritic targets in the nucleus tractus solitarius. Neuroscience 91, 119–132 (1999)
Berthoud, H. R., Earle, T., Zheng, H., Patterson, L. M. & Phifer, C. Food-related gastrointestinal signals activate caudal brainstem neurons expressing both NMDA and AMPA receptors. Brain Res. 915, 143–154 (2001)
Covasa, M., Hung, C. Y., Ritter, R. C. & Burns, G. A. Intracerebroventricular administration of MK-801 increases food intake through mechanisms independent of gastric emptying. Am. J. Physiol. Regul. Integr. Comp. Physiol. 287, R1462–R1467 (2004)
Treece, B. R., Covasa, M., Ritter, R. C. & Burns, G. A. Delay in meal termination follows blockade of N-methyl-d-aspartate receptors in the dorsal hindbrain. Brain Res. 810, 34–40 (1998)
Pocai, A. et al. Restoration of hypothalamic lipid sensing normalizes energy and glucose homeostasis in overfed rats. J. Clin. Invest. 116, 1081–1091 (2006)
Caspi, L., Wang, P. Y. & Lam, T. K. A balance of lipid-sensing mechanisms in the brain and liver. Cell Metab. 6, 99–104 (2007)
Ley, R. E., Turnbaugh, P. J., Klein, S. & Gordon, J. I. Microbial ecology: human gut microbes associated with obesity. Nature 444, 1022–1023 (2006)
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006)
Cummings, D. E., Overduin, J. & Foster-Schubert, K. E. Gastric bypass for obesity: mechanisms of weight loss and diabetes resolution. J. Clin. Endocrinol. Metab. 89, 2608–2615 (2004)
Acknowledgements
We thank C. Baveghems for technical assistance. This work is supported by a research grant to T.K.T.L. from the Canadian Institute of Health Research (MOP-82701). R.G.-J. is supported by the National Institutes of Health (DK45024). G.J.S. is supported by the National Institutes of Health (DK47208) and the Skirball Institute. T.K.T.L. holds the John Kitson McIvor Endowed Chair in Diabetes Research at the University Health Network and the University of Toronto.
Author Contributions P.Y.T.W. conducted and designed experiments, performed data analyses and wrote the manuscript; L.C., C.K.L.L., M.C. and M.A. conducted experiments; X.L. assisted in surgical procedures; P.E.L. and R.G.-J. assisted in LCFA-CoA measurements; G.J.S. assisted in surgical procedures and designed experiments; and T.K.T.L. supervised the project, designed experiments and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Methods, Supplementary Tables S1-S2 and Supplementary Figures S1-S2 with Legends (PDF 286 kb)
Rights and permissions
About this article
Cite this article
Wang, P., Caspi, L., Lam, C. et al. Upper intestinal lipids trigger a gut–brain–liver axis to regulate glucose production. Nature 452, 1012–1016 (2008). https://doi.org/10.1038/nature06852
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06852
This article is cited by
-
Gut microbiota and type 2 diabetes mellitus: a focus on the gut-brain axis
Endocrine (2024)
-
Oligofructose improves small intestinal lipid-sensing mechanisms via alterations to the small intestinal microbiota
Microbiome (2023)
-
Gut liver brain axis in diseases: the implications for therapeutic interventions
Signal Transduction and Targeted Therapy (2023)
-
Effect of duodenal-jejunal bypass on diabetes in the early postoperative period
Scientific Reports (2023)
-
Are gut dysbiosis, barrier disruption, and endotoxemia related to adipose tissue dysfunction in metabolic disorders? Overview of the mechanisms involved
Internal and Emergency Medicine (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.