Abstract
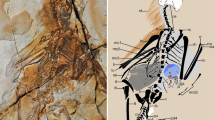
Bats (Chiroptera) represent one of the largest and most diverse radiations of mammals, accounting for one-fifth of extant species1. Although recent studies unambiguously support bat monophyly2,3,4 and consensus is rapidly emerging about evolutionary relationships among extant lineages5,6,7,8, the fossil record of bats extends over 50 million years, and early evolution of the group remains poorly understood5,7,8,9. Here we describe a new bat from the Early Eocene Green River Formation of Wyoming, USA, with features that are more primitive than seen in any previously known bat. The evolutionary pathways that led to flapping flight and echolocation in bats have been in dispute7,8,9,10,11,12,13,14,15,16,17,18, and until now fossils have been of limited use in documenting transitions involved in this marked change in lifestyle. Phylogenetically informed comparisons of the new taxon with other bats and non-flying mammals reveal that critical morphological and functional changes evolved incrementally. Forelimb anatomy indicates that the new bat was capable of powered flight like other Eocene bats, but ear morphology suggests that it lacked their echolocation abilities, supporting a ‘flight first’ hypothesis for chiropteran evolution. The shape of the wings suggests that an undulating gliding–fluttering flight style may be primitive for bats, and the presence of a long calcar indicates that a broad tail membrane evolved early in Chiroptera, probably functioning as an additional airfoil rather than as a prey-capture device. Limb proportions and retention of claws on all digits indicate that the new bat may have been an agile climber that employed quadrupedal locomotion and under-branch hanging behaviour.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Simmons, N. B. in Mammal Species of the World: a Taxonomic and Geographic Reference (eds Wilson, D. E. & Reeder, D. M.) 312–529 (Johns Hopkins Univ. Press, Baltimore, MD, 2005)
Simmons, N. B. The case for chiropteran monophyly. Am. Mus. Novitat. 3103, 1–54 (1994)
Murphy, W. J. et al. Molecular phylogenetics and the origin of placental mammals. Nature 409, 614–618 (2001)
Teeling, E. C. et al. Molecular evidence regarding the origin of echolocation and flight in bats. Nature 403, 188–192 (2000)
Springer, M. S., Teeling, E. C., Madsen, O., Stanhope, M. J. & de Jong, W. W. Integrated fossil and molecular data reconstruct bat echolocation. Proc. Natl Acad. Sci. USA 98, 6241–6246 (2001)
Van Den Bussche, R. A. & Hoofer, S. R. Phylogenetic relationships among recent chiropteran families and the importance of choosing appropriate out-group taxa. J. Mamm. 85, 321–330 (2004)
Eick, G. N., Jacobs, D. S. & Matthee, C. A. A nuclear DNA phylogenetic perspective on the evolution of echolocation and historical biogeography of extant bats (Chiroptera). Mol. Biol. Evol. 22, 1869–1886 (2005)
Teeling, E. C. et al. A molecular phylogeny for bats illuminates biogeography and the fossil record. Science 307, 580–584 (2005)
Simmons, N. B. & Geisler, J. H. Phylogenetic relationships of Icaronycteris, Archaeonycteris, Hassianycteris, and Palaeochiropteryx to extant bat lineages, with comments on the evolution of echolocation and foraging strategies in Microchiroptera. Bull. Am. Mus. Nat. Hist. 235, 1–182 (1998)
Norberg, U. M. & Rayner, J. M. V. Ecological morphology and flight in bats (Mammalia: Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Phil. Trans. R. Soc. B 316, 335–427 (1987)
Habersetzer, J. & Storch, G. Klassifikation und funktionelle Flügelmorphologie paläogener Fledermäuse (Mammalia, Chiroptera). Cour. Forsch.-Inst. Senckenberg 91, 11–150 (1987)
Norberg, U. M. in European Bat Research (eds Hanák, V., Horácek, I. & Gaisler, J.) 197–211 (Charles Univ. Press, Praha, 1989)
Speakman, J. R. & Racey, P. A. No cost of echolocation for bats in flight. Nature 350, 421–423 (1991)
Speakman, J. R. The evolution of flight and echolocation in bats: another leap in the dark. Mammal Rev. 31, 111–130 (2001)
Simmons, N. B. in Ecology, Evolution, and Behavior of Bats (eds Racey, P. A. & Swift, S. M.) 27–43 (Oxford Univ. Press, Oxford, 1995)
Arita, H. T. & Fenton, M. B. Flight and echolocation in the ecology and evolution of bats. Trends Ecol. Evol. 12, 53–58 (1997)
Jones, G. & Teeling, E. C. The evolution of echolocation in bats. Trends Ecol. Evol. 21, 149–156 (2006)
Schutt, W. A. & Simmons, N. B. Morphology and homology of the chiropteran calcar, with comments on the phylogenetic relationships of Archaeopteropus . J. Mamm. Evol. 5, 1–32 (1998)
Jepsen, G. L. Early Eocene bat from Wyoming. Science 154, 1333–1339 (1966)
Clyde, W. C., Sheldon, N. D., Koch, P. L., Gunnell, G. F. & Bartels, W. S. Linking the Wasatchian/Bridgerian boundary to the Cenozoic Global Climate Optimum: new magnetostratigraphic and isotopic results from South Pass, Wyoming. Palaeogeogr. Palaeoclimatol. Palaeoecol. 167, 175–199 (2001)
Storch, G., Sigé, B. & Habersetzer, J. Tachypteron franzeni, n. gen., n. sp., earliest emballonurid bat from the middle Eocene of Messel (Mammalia, Chiroptera). Paläontol. Z. 76, 189–199 (2002)
Novacek, M. J. Evidence for echolocation in the oldest known bats. Nature 315, 140–141 (1985)
Novacek, M. J. Auditory features and affinities of the Eocene bats Icaronycteris and Palaeochiropteryx (Microchiroptera, incertae sedis). Am. Mus. Novitat. 2877, 1–18 (1987)
Habersetzer, J. & Storch, G. Cochlea size in extant Chiroptera and Middle Eocene Microchiroptera from Messel. Naturwissenchaften 79, 462–466 (1992)
Gunnell, G. F. et al. Oldest placental mammal from sub-Saharan Africa: Eocene microbat from Tanzania—evidence for early evolution of sophisticated echolocation. Palaeontol. Elect. 5, 1–10 (2003)
Vaughan, T. A. in About Bats (eds Slaughter, B. H. & Walton, W. D.) 127–143 (Southern Methodist Univ. Press, Dallas, TX, 1970)
Norberg, U. M. in Bat Flight—Fledermausflug, Bionia Report 5 (ed. Nachtigall, W.) 13–26 (Gustav Fischer, Stuttgart, 1986)
Rayner, J. M. V. in Bat Flight—Fledermausflug, Bionia Report 5 (ed. Nachtigall, W.) 27–74 (Gustav Fischer, Stuttgart, 1986)
Rayner, J. M. V. Bounding and undulating flight in birds. J. Theor. Biol. 117, 47–77 (1985)
Schutt, W. A. & Simmons, N. B. in Functional and Evolutionary Ecology of Bats (eds Zubaid, A., McCracken, G. & Kunz, T.) 15–159 (Oxford Univ. Press, Oxford, 2006)
Buchheim, H. P. Paleoenvironments, lithofacies and varves of the Fossil Butte Member of the Eocene Green River Formation, southwestern Wyoming. Contrib. Geol. Univ. Wyoming 30, 3–14 (1994)
Grande, L. & Buchheim, H. P. Paleontological and sedimentological variation in early Eocene Fossil Lake. Contrib. Geol. Univ. Wyoming 30, 33–56 (1994)
Woodburne, M. O. in Late Cretaceous and Cenozoic Mammals of North America—Biostratigraphy and Geochronology (ed. Woodburne, M. O.) 315–343 (Columbia Univ. Press, New York, 2004)
Gunnell, G. F. & Simmons, N. B. Fossil evidence and the origin of bats. J. Mamm. Evol. 12, 209–246 (2005)
Swofford, D. L. PAUP*. Phylogenetic Analysis Using Parsimony (*and Other Methods) Version 4 (Sinauer Associates, Sunderland, MA, 2001)
Bremer, K. The limits of amino acid sequence data in angiosperm phylogenetic reconstructions. Evolution Int. J. Org. Evolution 42, 795–803 (1988)
Maddison, W. P. & Maddison, D. R. MacClade: Analysis of Phylogeny and Character Evolution Version 3.0 (Sinauer Associates, Sunderland, MA, 1992)
Miller-Butterworth, C. M. et al. A family matter: conclusive resolution of the taxonomic position of the long-fingered bats, Miniopterus . Mol. Biol. Evol. 24, 1553–1561 (2007)
Acknowledgements
We thank I. Morrison for preparing the holotype specimen of Onychonycteris finneyi; A. Aase and H. Galliano for help in obtaining photographs and casts of the paratype; B. Miljour and P. Wynne for their assistance with figures; B. Boyle for photographs; and P. Myers, L. Grande, W. Simpson, M. Engstrom, J. Eger, S. Woodward, S. Schaal, G. Storch, N. Micklich, D. Lunde, E. Westwig and C. Norris for access to specimens. Students sponsored by the Undergraduate Research Opportunities Program (UROP) at the University of Michigan were involved in data acquisition and compilation. Funding was provided by the United States National Science Foundation and Deutsche Forschungsgemeinschaft DFG.
Author information
Authors and Affiliations
Corresponding author
Additional information
Morphological data used in phylogenetic analyses are deposited in MorphoBank and can be obtained at http://morphobank.geongrid.org/permalink/?P104.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-3, Supplementary Figures 1-10 with Legends and additional references. (PDF 1884 kb)
Rights and permissions
About this article
Cite this article
Simmons, N., Seymour, K., Habersetzer, J. et al. Primitive Early Eocene bat from Wyoming and the evolution of flight and echolocation. Nature 451, 818–821 (2008). https://doi.org/10.1038/nature06549
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06549
This article is cited by
-
Development of the hyolaryngeal architecture in horseshoe bats: insights into the evolution of the pulse generation for laryngeal echolocation
EvoDevo (2024)
-
Insights into the formation and diversification of a novel chiropteran wing membrane from embryonic development
BMC Biology (2023)
-
Divergent vertebral formulae shape the evolution of axial complexity in mammals
Nature Ecology & Evolution (2023)
-
The evolutionary history and ancestral biogeographic range estimation of old-world Rhinolophidae and Hipposideridae (Chiroptera)
BMC Ecology and Evolution (2022)
-
The origin of placental mammal life histories
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.