Abstract
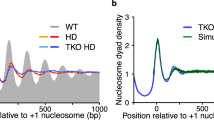
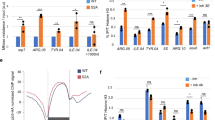
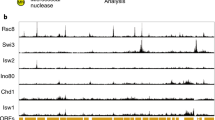
Chromatin allows the eukaryotic cell to package its DNA efficiently. To understand how chromatin structure is controlled across the Saccharomyces cerevisiae genome, we have investigated the role of the ATP-dependent chromatin remodelling complex Isw2 in positioning nucleosomes. We find that Isw2 functions adjacent to promoter regions where it repositions nucleosomes at the interface between genic and intergenic sequences. Nucleosome repositioning by Isw2 is directional and results in increased nucleosome occupancy of the intergenic region. Loss of Isw2 activity leads to inappropriate transcription, resulting in the generation of both coding and noncoding transcripts. Here we show that Isw2 repositions nucleosomes to enforce directionality on transcription by preventing transcription initiation from cryptic sites. Our analyses reveal how chromatin is organized on a global scale and advance our understanding of how transcription is regulated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Luger, K., Mader, A. W., Richmond, R. K., Sargent, D. F. & Richmond, T. J. Crystal structure of the nucleosome core particle at 2.8 Å resolution. Nature 389, 251–260 (1997)
Ehrenhofer-Murray, A. E. Chromatin dynamics at DNA replication, transcription and repair. Eur. J. Biochem. 271, 2335–2349 (2004)
Satchwell, S. C., Drew, H. R. & Travers, A. A. Sequence periodicities in chicken nucleosome core DNA. J. Mol. Biol. 191, 659–675 (1986)
Lowary, P. T. & Widom, J. New DNA sequence rules for high affinity binding to histone octamer and sequence-directed nucleosome positioning. J. Mol. Biol. 276, 19–42 (1998)
Anderson, J. D. & Widom, J. Poly(dA-dT) promoter elements increase the equilibrium accessibility of nucleosomal DNA target sites. Mol. Cell. Biol. 21, 3830–3839 (2001)
Segal, E. et al. A genomic code for nucleosome positioning. Nature 442, 772–778 (2006)
Ioshikhes, I. P., Albert, I., Zanton, S. J. & Pugh, B. F. Nucleosome positions predicted through comparative genomics. Nature Genet. 38, 1210–1215 (2006)
Narlikar, G. J., Fan, H. Y. & Kingston, R. E. Cooperation between complexes that regulate chromatin structure and transcription. Cell 108, 475–487 (2002)
Rando, O. J. & Ahmad, K. Rules and regulation in the primary structure of chromatin. Curr. Opin. Cell Biol. 19, 250–256 (2007)
Eisen, J. A., Sweder, K. S. & Hanawalt, P. C. Evolution of the SNF2 family of proteins: subfamilies with distinct sequences and functions. Nucleic Acids Res. 23, 2715–2723 (1995)
Flaus, A., Martin, D. M., Barton, G. J. & Owen-Hughes, T. Identification of multiple distinct Snf2 subfamilies with conserved structural motifs. Nucleic Acids Res. 34, 2887–2905 (2006)
Badenhorst, P., Voas, M., Rebay, I. & Wu, C. Biological functions of the ISWI chromatin remodeling complex NURF. Genes Dev. 16, 3186–3198 (2002)
Yasui, D., Miyano, M., Cai, S., Varga-Weisz, P. & Kohwi-Shigematsu, T. SATB1 targets chromatin remodelling to regulate genes over long distances. Nature 419, 641–645 (2002)
Deuring, R. et al. The ISWI chromatin-remodeling protein is required for gene expression and the maintenance of higher order chromatin structure in vivo . Mol. Cell 5, 355–365 (2000)
Collins, N. et al. An ACF1–ISWI chromatin-remodeling complex is required for DNA replication through heterochromatin. Nature Genet. 32, 627–632 (2002)
Poot, R. A. et al. The Williams syndrome transcription factor interacts with PCNA to target chromatin remodelling by ISWI to replication foci. Nature Cell Biol. 6, 1236–1244 (2004)
Fyodorov, D. V., Blower, M. D., Karpen, G. H. & Kadonaga, J. T. Acf1 confers unique activities to ACF/CHRAC and promotes the formation rather than disruption of chromatin in vivo . Genes Dev. 18, 170–183 (2004)
Zhou, Y., Santoro, R. & Grummt, I. The chromatin remodeling complex NoRC targets HDAC1 to the ribosomal gene promoter and represses RNA polymerase I transcription. EMBO J. 21, 4632–4640 (2002)
Li, J., Langst, G. & Grummt, I. NoRC-dependent nucleosome positioning silences rRNA genes. EMBO J. 25, 5735–5741 (2006)
Hakimi, M. A. et al. A chromatin remodelling complex that loads cohesin onto human chromosomes. Nature 418, 994–998 (2002)
Goldmark, J. P., Fazzio, T. G., Estep, P. W., Church, G. M. & Tsukiyama, T. The Isw2 chromatin remodeling complex represses early meiotic genes upon recruitment by Ume6p. Cell 103, 423–433 (2000)
Fazzio, T. G., Gelbart, M. E. & Tsukiyama, T. Two distinct mechanisms of chromatin interaction by the isw2 chromatin remodeling complex in vivo . Mol. Cell. Biol. 25, 9165–9174 (2005)
Kent, N. A., Karabetsou, N., Politis, P. K. & Mellor, J. In vivo chromatin remodeling by yeast ISWI homologs Isw1p and Isw2p. Genes Dev. 15, 619–626 (2001)
Whitehouse, I. & Tsukiyama, T. Antagonistic forces that position nucleosomes in vivo . Nature Struct. Mol. Biol. 13, 633–640 (2006)
Yuan, G. C. et al. Genome-scale identification of nucleosome positions in S. cerevisiae . Science 309, 626–630 (2005)
Gelbart, M. E., Bachman, N., Delrow, J., Boeke, J. D. & Tsukiyama, T. Genome-wide identification of Isw2 chromatin-remodeling targets by localization of a catalytically inactive mutant. Genes Dev. 19, 942–954 (2005)
Fazzio, T. G. et al. Widespread collaboration of Isw2 and Sin3–Rpd3 chromatin remodeling complexes in transcriptional repression. Mol. Cell. Biol. 21, 6450–6460 (2001)
Albert, I. et al. Translational and rotational settings of H2A.Z nucleosomes across the Saccharomyces cerevisiae genome. Nature 446, 572–576 (2007)
Houseley, J., LaCava, J. & Tollervey, D. RNA-quality control by the exosome. Nature Rev. Mol. Cell Biol. 7, 529–539 (2006)
Wyers, F. et al. Cryptic pol II transcripts are degraded by a nuclear quality control pathway involving a new poly(A) polymerase. Cell 121, 725–737 (2005)
LaCava, J. et al. RNA degradation by the exosome is promoted by a nuclear polyadenylation complex. Cell 121, 713–724 (2005)
Vanacova, S. et al. A new yeast poly(A) polymerase complex involved in RNA quality control. PLoS Biol. 3, e189 (2005)
Davis, C. A. & Ares, M. Accumulation of unstable promoter-associated transcripts upon loss of the nuclear exosome subunit Rrp6p in Saccharomyces cerevisiae . Proc. Natl Acad. Sci. USA 103, 3262–3267 (2006)
Egecioglu, D. E., Henras, A. K. & Chanfreau, G. F. Contributions of Trf4p- and Trf5p-dependent polyadenylation to the processing and degradative functions of the yeast nuclear exosome. RNA 12, 26–32 (2006)
Pan, X. et al. A DNA integrity network in the yeast Saccharomyces cerevisiae . Cell 124, 1069–1081 (2006)
Behe, M. J. An overabundance of long oligopurine tracts occurs in the genome of simple and complex eukaryotes. Nucleic Acids Res. 23, 689–695 (1995)
Kunkel, G. R. & Martinson, H. G. Nucleosomes will not form on double-stranded RNa or over poly(dA).poly(dT) tracts in recombinant DNA. Nucleic Acids Res. 9, 6869–6888 (1981)
Iyer, V. & Struhl, K. Poly(dA:dT), a ubiquitous promoter element that stimulates transcription via its intrinsic DNA structure. EMBO J. 14, 2570–2579 (1995)
Raisner, R. M. et al. Histone variant H2A.Z marks the 5′ ends of both active and inactive genes in euchromatin. Cell 123, 233–248 (2005)
Guillemette, B. et al. Variant histone H2A.Z is globally localized to the promoters of inactive yeast genes and regulates nucleosome positioning. PLoS Biol. 3, e384 (2005)
Zhang, H., Roberts, D. N. & Cairns, B. R. Genome-wide dynamics of Htz1, a histone H2A variant that poises repressed/basal promoters for activation through histone loss. Cell 123, 219–231 (2005)
Brickner, D. G. et al. H2A.Z-mediated localization of genes at the nuclear periphery confers epigenetic memory of previous transcriptional state. PLoS Biol. 5, e81 (2007)
Dion, M. F. et al. Dynamics of replication-independent histone turnover in budding yeast. Science 315, 1405–1408 (2007)
Eisen, M. B., Spellman, P. T., Brown, P. O. & Botstein, D. Cluster analysis and display of genome-wide expression patterns. Proc. Natl Acad. Sci. USA 95, 14863–14868 (1998)
Liu, C. L. et al. Single-nucleosome mapping of histone modifications in S. cerevisiae . PLoS Biol. 3, e328 (2005)
Zhang, Z. & Dietrich, F. S. Mapping of transcription start sites in Saccharomyces cerevisiae using 5′ SAGE. Nucleic Acids Res. 33, 2838–2851 (2005)
MacIsaac, K. D. et al. An improved map of conserved regulatory sites for Saccharomyces cerevisiae . BMC Bioinformatics 7, 113 (2006)
Miura, F. et al. A large-scale full-length cDNA analysis to explore the budding yeast transcriptome. Proc. Natl Acad. Sci. USA 103, 17846–17851 (2006)
Acknowledgements
We thank members of the Tsukiyama and Henikoff laboratories for discussions, R. Basom for help with data analysis, and S. Henikoff, S. Biggins, S. Hahn and T. Owen-Hughes for critical reading of the manuscript. This work was supported by funds from NIGMS and the Leukemia and Lymphoma Society to T.T, from the Burroughs Wellcome Fund and Human Frontier Science Program to O.J.R, and from the NCI to J.D.
Author Contributions Experimental strategy was designed by I.W. and T.T., and experiments were performed by I.W. Preliminary nucleosome mapping was performed in collaboration with O.J.R. Data were analysed by I.W., with technical assistance from J.D. The paper was written by I.W., with assistance from T.T. All authors discussed the results and experiments, and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
This file contains Supplementary Table S1and Supplementary Figures S1-S18 with Legends. (PDF 4886 kb)
Supplementary Table
This file contains Supplementary Table with summary data of regions bound by Isw2, sites of chromatin remodelling and lists of nucleosome positions. (XLS 25913 kb)
Rights and permissions
About this article
Cite this article
Whitehouse, I., Rando, O., Delrow, J. et al. Chromatin remodelling at promoters suppresses antisense transcription. Nature 450, 1031–1035 (2007). https://doi.org/10.1038/nature06391
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature06391
This article is cited by
-
Energy-driven genome regulation by ATP-dependent chromatin remodellers
Nature Reviews Molecular Cell Biology (2024)
-
Structures of transcription preinitiation complex engaged with the +1 nucleosome
Nature Structural & Molecular Biology (2023)
-
Nucleosome density shapes kilobase-scale regulation by a mammalian chromatin remodeler
Nature Structural & Molecular Biology (2023)
-
Global histone protein surface accessibility in yeast indicates a uniformly loosely packed genome with canonical nucleosomes
Epigenetics & Chromatin (2021)
-
Ruler elements in chromatin remodelers set nucleosome array spacing and phasing
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.