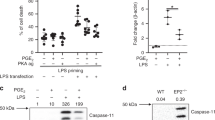
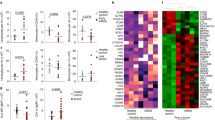
Abstract
It is becoming increasingly clear that the autonomic nervous system and the immune system demonstrate cross-talk during inflammation by means of sympathetic and parasympathetic pathways1,2. We investigated whether phagocytes are capable of de novo production of catecholamines, suggesting an autocrine/paracrine self-regulatory mechanism by catecholamines during inflammation, as has been described for lymphocytes3. Here we show that exposure of phagocytes to lipopolysaccharide led to a release of catecholamines and an induction of catecholamine-generating and degrading enzymes, indicating the presence of the complete intracellular machinery for the generation, release and inactivation of catecholamines. To assess the importance of these findings in vivo, we chose two models of acute lung injury. Blockade of α2-adrenoreceptors or catecholamine-generating enzymes greatly suppressed lung inflammation, whereas the opposite was the case either for an α2-adrenoreceptor agonist or for inhibition of catecholamine-degrading enzymes. We were able to exclude T cells or sympathetic nerve endings as sources of the injury-modulating catecholamines. Our studies identify phagocytes as a new source of catecholamines, which enhance the inflammatory response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tracey, K. J. The inflammatory reflex. Nature 420, 853–859 (2002)
Sternberg, E. M. Neural regulation of innate immunity: a coordinated nonspecific host response to pathogens. Nature Rev. Immunol. 6, 318–328 (2006)
Bergquist, J., Tarkowski, A., Ekman, R. & Ewing, A. Discovery of endogenous catecholamines in lymphocytes and evidence for catecholamine regulation of lymphocyte function via an autocrine loop. Proc. Natl Acad. Sci. USA 91, 12912–12916 (1994)
Cook, D. N., Pisetsky, D. S. & Schwartz, D. A. Toll-like receptors in the pathogenesis of human disease. Nature Immunol. 5, 975–979 (2004)
Borovikova, L. V. et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405, 458–462 (2000)
Wang, H. et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature 421, 384–388 (2003)
Musso, N. R., Brenci, S., Setti, M., Indiveri, F. & Lotti, G. Catecholamine content and in vitro catecholamine synthesis in peripheral human lymphocytes. J. Clin. Endocrinol. Metab. 81, 3553–3557 (1996)
Swanson, M. A., Lee, W. T. & Sanders, V. M. IFN-γ production by Th1 cells generated from naive CD4+ T cells exposed to norepinephrine. J. Immunol. 166, 232–240 (2001)
Sanders, V. M. et al. Differential expression of the β2-adrenergic receptor by Th1 and Th2 clones: implications for cytokine production and B cell help. J. Immunol. 158, 4200–4210 (1997)
Kohm, A. P., Mozaffarian, A. & Sanders, V. M. B cell receptor- and β2-adrenergic receptor-induced regulation of B7-2 (CD86) expression in B cells. J. Immunol. 168, 6314–6322 (2002)
Bergquist, J., Ohlsson, B. & Tarkowski, A. Nuclear factor-κB is involved in the catecholaminergic suppression of immunocompetent cells. Ann. NY Acad. Sci. 917, 281–289 (2000)
Torres, K. C. et al. Norepinephrine, dopamine and dexamethasone modulate discrete leukocyte subpopulations and cytokine profiles from human PBMC. J. Neuroimmunol. 166, 144–157 (2005)
Brown, S. W. et al. Catecholamines in a macrophage cell line. J. Neuroimmunol. 135, 47–55 (2003)
Cosentino, M. et al. Endogenous catecholamine synthesis, metabolism, storage and uptake in human neutrophils. Life Sci. 64, 975–981 (1999)
Elenkov, I. J., Wilder, R. L., Chrousos, G. P. & Vizi, E. S. The sympathetic nerve–an integrative interface between two supersystems: the brain and the immune system. Pharmacol. Rev. 52, 595–638 (2000)
Ueland, P. M. Pharmacological and biochemical aspects of S-adenosylhomocysteine and S-adenosylhomocysteine hydrolase. Pharmacol. Rev. 34, 223–253 (1982)
Zhu, B. T. Catechol-O-methyltransferase (COMT)-mediated methylation metabolism of endogenous bioactive catechols and modulation by endobiotics and xenobiotics: importance in pathophysiology and pathogenesis. Curr. Drug Metab. 3, 321–349 (2002)
Bone, R. C., Francis, P. B. & Pierce, A. K. Intravascular coagulation associated with the adult respiratory distress syndrome. Am. J. Med. 61, 585–589 (1976)
Bylund, D. B. et al. International Union of Pharmacology nomenclature of adrenoceptors. Pharmacol. Rev. 46, 121–136 (1994)
Hoffman, B. B. in Goodman & Gilman’s The Pharmacological Basics of Therapeutics (eds Brunton, L. L., Lazo, J. S. & Parker, K. L.) 845–868 (McGraw-Hill, New York, 2006)
Ward, P. A. Rous–Whipple Award Lecture. Role of complement in lung inflammatory injury. Am. J. Pathol. 149, 1081–1086 (1996)
Spengler, R. N., Allen, R. M., Remick, D. G., Strieter, R. M. & Kunkel, S. L. Stimulation of α-adrenergic receptor augments the production of macrophage-derived tumor necrosis factor. J. Immunol. 145, 1430–1434 (1990)
Czermak, B. J. et al. Synergistic enhancement of chemokine generation and lung injury by C5a or the membrane attack complex of complement. Am. J. Pathol. 154, 1513–1524 (1999)
Huber-Lang, M. et al. Generation of C5a in the absence of C3: a new complement activation pathway. Nature Med. 12, 682–687 (2006)
Gao, H. et al. Stat3 activation in acute lung injury. J. Immunol. 172, 7703–7712 (2004)
Hoesel, L. M. et al. Harmful and protective roles of neutrophils in sepsis. Shock 24, 40–47 (2005)
Acknowledgements
We thank B. Schumann and S. Scott for assistance in the preparation of this manuscript. This study was supported by NIH grants to P.A.W. and R.R.N., and by Deutsche Forschungsgemeinschaft grants to M.S.H.-L.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints.
Supplementary information
Supplementary Information
The file contains Supplementary Figure S1 and Supplementary Table S1 with Legends. (PDF 389 kb)
Rights and permissions
About this article
Cite this article
Flierl, M., Rittirsch, D., Nadeau, B. et al. Phagocyte-derived catecholamines enhance acute inflammatory injury. Nature 449, 721–725 (2007). https://doi.org/10.1038/nature06185
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature06185
This article is cited by
-
Pathophysiology of acute lung injury in patients with acute brain injury: the triple-hit hypothesis
Critical Care (2024)
-
Lung microbiome: new insights into the pathogenesis of respiratory diseases
Signal Transduction and Targeted Therapy (2024)
-
Metabolomics-directed nanotechnology in viral diseases management: COVID-19 a case study
Pharmacological Reports (2023)
-
Intrinsic cardiac adrenergic cells contribute to LPS-induced myocardial dysfunction
Communications Biology (2022)
-
Increased Renal Dysfunction, Apoptosis, and Fibrogenesis Through Sympathetic Hyperactivity After Focal Cerebral Infarction
Translational Stroke Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.