Abstract
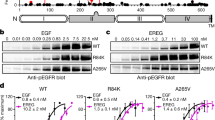
Members of the epidermal growth factor receptor family (EGFR/ERBB1, ERBB2/HER2, ERBB3/HER3 and ERBB4/HER4) are key targets for inhibition in cancer therapy1. Critical for activation is the formation of an asymmetric dimer by the intracellular kinase domains, in which the carboxy-terminal lobe (C lobe) of one kinase domain induces an active conformation in the other2. The cytoplasmic protein MIG6 (mitogen-induced gene 6; also known as ERRFI1) interacts with and inhibits the kinase domains of EGFR and ERBB2 (refs 3–5). Crystal structures of complexes between the EGFR kinase domain and a fragment of MIG6 show that a ∼25-residue epitope (segment 1) from MIG6 binds to the distal surface of the C lobe of the kinase domain. Biochemical and cell-based analyses confirm that this interaction contributes to EGFR inhibition by blocking the formation of the activating dimer interface. A longer MIG6 peptide that is extended C terminal to segment 1 has increased potency as an inhibitor of the activated EGFR kinase domain, while retaining a critical dependence on segment 1. We show that signalling by EGFR molecules that contain constitutively active kinase domains still requires formation of the asymmetric dimer, underscoring the importance of dimer interface blockage in MIG6-mediated inhibition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hynes, N. E. & Lane, H. A. ERBB receptors and cancer: the complexity of targeted inhibitors. Nature Rev. Cancer 5, 341–354 (2005)
Zhang, X. et al. An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor. Cell 125, 1137–1149 (2006)
Hackel, P. O., Gishizky, M. & Ullrich, A. Mig-6 is a negative regulator of the epidermal growth factor receptor signal. Biol. Chem. 382, 1649–1662 (2001)
Xu, D., Makkinje, A. & Kyriakis, J. M. Gene 33 is an endogenous inhibitor of epidermal growth factor (EGF) receptor signaling and mediates dexamethasone-induced suppression of EGF function. J. Biol. Chem. 280, 2924–2933 (2005)
Fiorentino, L. et al. Inhibition of ErbB-2 mitogenic and transforming activity by RALT, a mitogen-induced signal transducer which binds to the ErbB-2 kinase domain. Mol. Cell. Biol. 20, 7735–7750 (2000)
Wood, E. R. et al. A unique structure for epidermal growth factor receptor bound to GW572016 (Lapatinib): relationships among protein conformation, inhibitor off-rate, and receptor activity in tumor cells. Cancer Res. 64, 6652–6659 (2004)
Jeffrey, P. D. et al. Mechanism of CDK activation revealed by the structure of a cyclinA–CDK2 complex. Nature 376, 313–320 (1995)
Ballaro, C. et al. Targeted expression of RALT in mouse skin inhibits epidermal growth factor receptor signalling and generates a Waved-like phenotype. EMBO Rep. 6, 755–761 (2005)
Ferby, I. et al. Mig6 is a negative regulator of EGF receptor-mediated skin morphogenesis and tumor formation. Nature Med. 12, 568–573 (2006)
Zhang, Y. W. et al. Evidence that MIG-6 is a tumor-suppressor gene. Oncogene 26, 269–276 (2007)
Shen, F. et al. Activated Cdc42-associated kinase 1 is a component of EGF receptor signaling complex and regulates EGF receptor degradation. Mol. Biol. Cell 18, 732–742 (2007)
Anastasi, S. et al. Feedback inhibition by RALT controls signal output by the ErbB network. Oncogene 22, 4221–4234 (2003)
Anastasi, S. et al. The evolutionarily conserved EBR module of RALT/MIG6 mediates suppression of the EGFR catalytic activity. Oncogene advanced online publication. doi: 10.1038/sj.onc.1210590 (18 June 2007)
Stamos, J., Sliwkowski, M. X. & Eigenbrot, C. Structure of the epidermal growth factor receptor kinase domain alone and in complex with a 4-anilinoquinazoline inhibitor. J. Biol. Chem. 277, 46265–46272 (2002)
Depetris, R. S. et al. Structural basis for inhibition of the insulin receptor by the adaptor protein Grb14. Mol. Cell 20, 325–333 (2005)
Lei, M., Robinson, M. A. & Harrison, S. C. The active conformation of the PAK1 kinase domain. Structure 13, 769–778 (2005)
Reynolds, A. R. et al. EGFR activation coupled to inhibition of tyrosine phosphatases causes lateral signal propagation. Nature Cell Biol. 5, 447–453 (2003)
Ichinose, J., Murata, M., Yanagida, T. & Sako, Y. EGF signalling amplification induced by dynamic clustering of EGFR. Biochem. Biophys. Res. Commun. 324, 1143–1149 (2004)
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–2139 (2004)
Sordella, R., Bell, D. W., Haber, D. A. & Settleman, J. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 305, 1163–1167 (2004)
Conti, E. & Kuriyan, J. Crystallographic analysis of the specific yet versatile recognition of distinct nuclear localization signals by karyopherin α. Structure 8, 329–338 (2000)
Wedegaertner, P. B. & Gill, G. N. Activation of the purified protein tyrosine kinase domain of the epidermal growth factor receptor. J. Biol. Chem. 264, 11346–11353 (1989)
Acknowledgements
We thank X. Cao and A. Fisher for cell culture support; D. King for mass spectrometry; the staff at beamlines 8.2.1 and 12.3.1 of the Advanced Light Source for technical support; and M. Seeliger, S. Deindl, P. Pellicena, J. Gureasko, S. Jacques and other members in the Kuriyan and Cole groups for technical help and discussions. We thank T. Miller for discussions on ACK1. This work is supported in part by grants from the NCI to J.K. and from NIH to P.A.C. R.B. is supported by the Susan G. Komen Breast Cancer Foundation.
Author Contributions J.K., X.Z. and P.A.C. designed the project. X.Z. performed biochemical and cell-based assays and crystallography work. K.A.P. and R.B. synthesized peptides. N.J. performed some biochemical and cell-based assays. J.K. and X.Z. wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
The file contains Supplementary Notes, Supplementary Tables 1-2, Supplementary Figures 1-7 with Legends and additional references. (PDF 2393 kb)
Rights and permissions
About this article
Cite this article
Zhang, X., Pickin, K., Bose, R. et al. Inhibition of the EGF receptor by binding of MIG6 to an activating kinase domain interface. Nature 450, 741–744 (2007). https://doi.org/10.1038/nature05998
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05998
This article is cited by
-
Systematic Profiling of Mitogen-Inducible Gene 6 and Its Derived Peptides Binding to Receptor Tyrosine Kinases in Bone Cancers at Molecular and Cellular Levels
International Journal of Peptide Research and Therapeutics (2024)
-
Feedback activation of EGFR/wild-type RAS signaling axis limits KRASG12D inhibitor efficacy in KRASG12D-mutated colorectal cancer
Oncogene (2023)
-
Coupled folding-upon-binding of human tumor suppressor MIG6 to lung cancer EGFR kinase domain and molecular trimming/stapling of MIG6-derived β-hairpins to target the coupling event
European Biophysics Journal (2023)
-
Critical role of mitogen-inducible gene 6 in restraining endothelial cell permeability to maintain vascular homeostasis
Journal of Cell Communication and Signaling (2023)
-
Role of ErbB and IL-1 signaling pathways in the dermonecrotic lesion induced by Loxosceles sphingomyelinases D
Archives of Toxicology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.