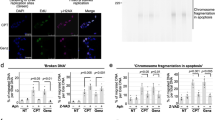
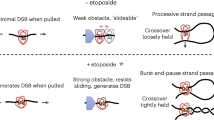
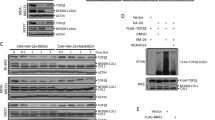
Abstract
Increasing the ability of chemotherapeutic drugs to kill cancer cells is often hampered by a limited understanding of their mechanism of action. Camptothecins, such as topotecan, induce cell death by poisoning DNA topoisomerase I, an enzyme capable of removing DNA supercoils1,2,3,4. Topotecan is thought to stabilize a covalent topoisomerase–DNA complex5,6,7, rendering it an obstacle to DNA replication forks2,3,8,9. Here we use single-molecule nanomanipulation to monitor the dynamics of human topoisomerase I in the presence of topotecan. This allowed us to detect the binding and unbinding of an individual topotecan molecule in real time and to quantify the drug-induced trapping of topoisomerase on DNA. Unexpectedly, our findings also show that topotecan significantly hinders topoisomerase-mediated DNA uncoiling, with a more pronounced effect on the removal of positive (overwound) versus negative supercoils. In vivo experiments in the budding yeast verified the resulting prediction that positive supercoils would accumulate during transcription and replication as a consequence of camptothecin poisoning of topoisomerase I. Positive supercoils, however, were not induced by drug treatment of cells expressing a catalytically active, camptothecin-resistant topoisomerase I mutant. This combination of single-molecule and in vivo data suggests a cytotoxic mechanism for camptothecins, in which the accumulation of positive supercoils ahead of the replication machinery induces potentially lethal DNA lesions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Champoux, J. J. DNA topoisomerases: structure, function, and mechanism. Annu. Rev. Biochem. 70, 369–413 (2001)
Li, T. K. & Liu, L. F. Tumor cell death induced by topoisomerase-targeting drugs. Annu. Rev. Pharmacol. Toxicol. 41, 53–77 (2001)
Pommier, Y. Topoisomerase I inhibitors: camptothecins and beyond. Nature Rev. Cancer 6, 789–802 (2006)
Wang, J. C. Cellular roles of DNA topoisomerases: a molecular perspective. Nature Rev. Mol. Cell Biol. 3, 430–440 (2002)
Hsiang, Y. H., Hertzberg, R., Hecht, S. & Liu, L. F. Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J. Biol. Chem. 260, 14873–14878 (1985)
Porter, S. E. & Champoux, J. J. The basis for camptothecin enhancement of DNA breakage by eukaryotic topoisomerase I. Nucleic Acids Res. 17, 8521–8532 (1989)
Staker, B. L. et al. The mechanism of topoisomerase I poisoning by a camptothecin analog. Proc. Natl Acad. Sci. USA 99, 15387–15392 (2002)
Holm, C., Covey, J. M., Kerrigan, D. & Pommier, Y. Differential requirement of DNA replication for the cytotoxicity of DNA topoisomerase I and II inhibitors in Chinese hamster DC3F cells. Cancer Res. 49, 6365–6368 (1989)
Hsiang, Y. H., Lihou, M. G. & Liu, L. F. Arrest of replication forks by drug-stabilized topoisomerase I–DNA cleavable complexes as a mechanism of cell killing by camptothecin. Cancer Res. 49, 5077–5082 (1989)
Corbett, K. D. & Berger, J. M. Structure, molecular mechanisms, and evolutionary relationships in DNA topoisomerases. Annu. Rev. Biophys. Biomol. Struct. 33, 95–118 (2004)
Bjornsti, M. A. Cancer therapeutics in yeast. Cancer Cell 2, 267–273 (2002)
Minsky, B. D. Combined-modality therapy of rectal cancer with irinotecan-based regimens. Oncology (Huntingt.) 18, 49–55 (2004)
Rodriguez-Galindo, C. et al. Clinical use of topoisomerase I inhibitors in anticancer treatment. Med. Pediatr. Oncol. 35, 385–402 (2000)
Stewart, D. J. Topotecan in the first-line treatment of small cell lung cancer. Oncologist 9 (Suppl 6). 33–42 (2004)
Sekiguchi, J. & Shuman, S. Vaccinia topoisomerase binds circumferentially to DNA. J. Biol. Chem. 269, 31731–31734 (1994)
Redinbo, M. R., Stewart, L., Kuhn, P., Champoux, J. J. & Hol, W. G. Crystal structures of human topoisomerase I in covalent and noncovalent complexes with DNA. Science 279, 1504–1513 (1998)
Koster, D. A., Croquette, V., Dekker, C., Shuman, S. & Dekker, N. H. Friction and torque govern the relaxation of DNA supercoils by eukaryotic topoisomerase IB. Nature 434, 671–674 (2005)
Stivers, J. T., Harris, T. K. & Mildvan, A. S. Vaccinia DNA topoisomerase I: evidence supporting a free rotation mechanism for DNA supercoil relaxation. Biochemistry 36, 5212–5222 (1997)
Chrencik, J. E. et al. Mechanisms of camptothecin resistance by human topoisomerase I mutations. J. Mol. Biol. 339, 773–784 (2004)
Strick, T., Allemand, J., Croquette, V. & Bensimon, D. Twisting and stretching single DNA molecules. Prog. Biophys. Mol. Biol. 74, 115–140 (2000)
Champoux, J. J. & Aronoff, R. The effects of camptothecin on the reaction and the specificity of the wheat germ type I topoisomerase. J. Biol. Chem. 264, 1010–1015 (1989)
Stewart, L., Ireton, G. C. & Champoux, J. J. A functional linker in human topoisomerase I is required for maximum sensitivity to camptothecin in a DNA relaxation assay. J. Biol. Chem. 274, 32950–32960 (1999)
Woo, M. H., Vance, J. R., Marcos, A. R., Bailly, C. & Bjornsti, M. A. Active site mutations in DNA topoisomerase I distinguish the cytotoxic activities of camptothecin and the indolocarbazole, rebeccamycin. J. Biol. Chem. 277, 3813–3822 (2002)
Giaever, G. N. & Wang, J. C. Supercoiling of intracellular DNA can occur in eukaryotic cells. Cell 55, 849–856 (1988)
Liu, L. F. & Wang, J. C. Supercoiling of the DNA template during transcription. Proc. Natl Acad. Sci. USA 84, 7024–7027 (1987)
Goto, T. & Wang, J. C. Yeast DNA topoisomerase II is encoded by a single-copy, essential gene. Cell 36, 1073–1080 (1984)
Woo, M. H. et al. Locking the DNA topoisomerase I protein clamp inhibits DNA rotation and induces cell lethality. Proc. Natl Acad. Sci. USA 100, 13767–13772 (2003)
Strick, T. R., Allemand, J. F., Bensimon, D., Bensimon, A. & Croquette, V. The elasticity of a single supercoiled DNA molecule. Science 271, 1835–1837 (1996).
Hann, C. et al. Increased camptothecin toxicity induced in mammalian cells expressing Saccharomyces cerevisiae DNA topoisomerase I. J. Biol. Chem. 273, 8425–8433 (1998)
Megonigal, M. D., Fertala, J. & Bjornsti, M. A. Alterations in the catalytic activity of yeast DNA topoisomerase I result in cell cycle arrest and cell death. J. Biol. Chem. 272, 12801–12808 (1997)
Acknowledgements
We thank K. Besteman and U. Keyser for discussions, S. Hage and Y.-H. Chien for DNA preparation, R. van Waardenburg for help with PyMol, and C. Dekker for critical reading and general support. We thank FOM, NWO, NIH and ALSAC for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Notes, Supplementary Figures 1-11 with Legends and additional references. (PDF 1026 kb)
Rights and permissions
About this article
Cite this article
Koster, D., Palle, K., Bot, E. et al. Antitumour drugs impede DNA uncoiling by topoisomerase I. Nature 448, 213–217 (2007). https://doi.org/10.1038/nature05938
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05938
This article is cited by
-
A novel bis(intercalating) agent for DNA based on the bis(benzoquinolizinium) derivative
Russian Chemical Bulletin (2023)
-
Mechanism of action of non-camptothecin inhibitor Genz-644282 in topoisomerase I inhibition
Communications Biology (2022)
-
The benzylisoquinoline alkaloids, berberine and coptisine, act against camptothecin-resistant topoisomerase I mutants
Scientific Reports (2021)
-
Interaction dynamics and site-specific electronic recognition of DNA-nicks with 2D solid-state nanopores
npj 2D Materials and Applications (2020)
-
Abortive activity of Topoisomerase I: a challenge for genome integrity?
Current Genetics (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.