Abstract
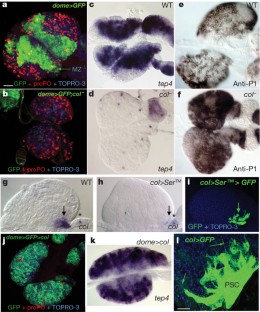
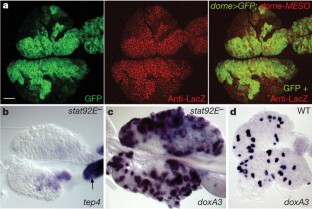
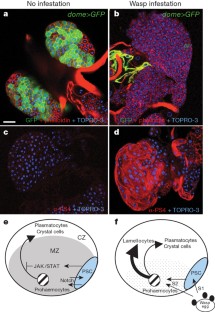
Drosophila haemocytes (blood cells) originate from a specialized haematopoietic organ—the lymph gland. Larval haematopoietic progenitors (prohaemocytes) give rise to three types of circulating haemocytes: plasmatocytes, crystal cells and lamellocytes. Lamellocytes, which are devoted to encapsulation of large foreign bodies, only differentiate in response to specific immune threats, such as parasitization by wasps. Here we show that a small cluster of signalling cells, termed the PSC (posterior signalling centre)1, controls the balance between multipotent prohaemocytes and differentiating haemocytes, and is necessary for the massive differentiation of lamellocytes that follows parasitization. Communication between the PSC and haematopoietic progenitors strictly depends on the PSC-restricted expression of Collier, the Drosophila orthologue of mammalian early B-cell factor. PSC cells act, in a non-cell-autonomous manner, to maintain JAK/STAT signalling activity in prohaemocytes, preventing their premature differentiation. Serrate-mediated Notch signalling from the PSC is required to maintain normal levels of col transcription. The key role of the PSC in controlling blood cell homeostasis is reminiscent of interactions between haematopoietic progenitors and their micro-environment in vertebrates2,3,4, thus further highlighting the interest of Drosophila as a model system for studying the evolution of haematopoiesis and cellular innate immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lebestky, T., Jung, S. H. & Banerjee, U. A Serrate-expressing signaling center controls Drosophila hematopoiesis. Genes Dev. 17, 348–353 (2003)
Nagasawa, T. Microenvironmental niches in the bone marrow required for B-cell development. Nature Rev. Immunol. 6, 107–116 (2006)
Wilson, A. & Trumpp, A. Bone-marrow haematopoietic-stem-cell niches. Nature Rev. Immunol. 6, 93–106 (2006)
Scadden, D. T. The stem-cell niche as an entity of action. Nature 441, 1075–1079 (2006)
Holz, A., Bossinger, B., Strasser, T., Janning, W. & Klapper, R. The two origins of haemocytes in Drosophila. Development 130, 4955–4962 (2003)
Bataille, L., Auge, B., Ferjoux, G., Haenlin, M. & Waltzer, L. Resolving embryonic blood cell fate choice in Drosophila: interplay of GCM and RUNX factors. Development 132, 4635–4644 (2005)
Rizki, R. M. & Rizki, T. M. Selective destruction of a host blood cell type by a parasitoid wasp. Proc. Natl Acad. Sci. USA 81, 6154–6158 (1984)
Lanot, R., Zachary, D., Holder, F. & Meister, M. Postembryonic hematopoiesis in Drosophila. Dev. Biol. 230, 243–257 (2001)
Evans, C. J., Hartenstein, V. & Banerjee, U. Thicker than blood: conserved mechanisms in Drosophila and vertebrate hematopoiesis. Dev. Cell 5, 673–690 (2003)
Meister, M. & Lagueux, M. Drosophila blood cells. Cell. Microbiol. 5, 573–580 (2003)
Jung, S. H., Evans, C. J., Uemura, C. & Banerjee, U. The Drosophila lymph gland as a developmental model of hematopoiesis. Development 132, 2521–2533 (2005)
Crozatier, M., Ubeda, J. M., Vincent, A. & Meister, M. Cellular immune response to parasitization in Drosophila requires the EBF orthologue Collier. PLoS Biol. 2, E196 (2004)
Irving, P. et al. New insights into Drosophila larval haemocyte functions through genome-wide analysis. Cell. Microbiol. 7, 335–350 (2005)
Sun, X. & Artavanis-Tsakonas, S. The intracellular deletions of Delta and Serrate define dominant negative forms of the Drosophila Notch ligands. Development 122, 2465–2474 (1996)
De Joussineau, C. et al. Delta-promoted filopodia mediate long-range lateral inhibition in Drosophila. Nature 426, 555–559 (2003)
Spradling, A., Drummond-Barbosa, D. & Kai, T. Stem cells find their niche. Nature 414, 98–104 (2001)
Hombria, J. C., Brown, S., Hader, S. & Zeidler, M. P. Characterisation of Upd2, a Drosophila JAK/STAT pathway ligand. Dev. Biol. 288, 420–433 (2005)
Arbouzova, N. I. & Zeidler, M. P. JAK/STAT signalling in Drosophila: insights into conserved regulatory and cellular functions. Development 133, 2605–2616 (2006)
Kraaijeveld, A. R. & Godfray, H. C. Trade-off between parasitoid resistance and larval competitive ability in Drosophila melanogaster. Nature 389, 278–280 (1997)
Rolff, J. & Siva-Jothy, M. T. Invertebrate ecological immunology. Science 301, 472–475 (2003)
Decotto, E. & Spradling, A. C. The Drosophila ovarian and testis stem cell niches: similar somatic stem cells and signals. Dev. Cell 9, 501–510 (2005)
Lin, H. The stem-cell niche theory: lessons from flies. Nature Rev. Genet. 3, 931–940 (2002)
Kimble, J. & Crittenden, S. L. Germline proliferation and its control. In WormBook (eds The C. elegans Research Community), doi/10.1895/worm-book.1.13.1 (15 August, 2005)
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003)
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003)
Sepp, K. J. & Auld, V. J. Conversion of lacZ enhancer trap lines to GAL4 lines using targeted transposition in Drosophila melanogaster. Genetics 151, 1093–1101 (1999)
Bourbon, H. M. et al. A P-insertion screen identifying novel X-linked essential genes in Drosophila. Mech. Dev. 110, 71–83 (2002)
Crozatier, M. & Vincent, A. Requirement for the Drosophila COE transcription factor Collier in formation of an embryonic muscle: transcriptional response to Notch signalling. Development 126, 1495–1504 (1999)
Acknowledgements
We thank J. Castelli-Gair Hombria, O. Devergne, M. Milan, S. Noselli, M. Zeidler and the Bloomington Stock Center for fly stocks, I. Ando and H. M. Müller for antibodies, and M. Haenlin, J. Smith, L. Waltzer for critical reading of the manuscript and discussion. We are grateful to platforme Toulouse RIO imaging and B. Ronsin for assistance with confocal microscopy. This work was supported by CNRS, a European Marie Curie PhD Training programme grant and Ministère de la Recherche et de la technologie (ACI Biologie Cellulaire, Moléculaire et Structurale) et Ministère des affaires étrangères (Programme Egide).
Author Contributions J.K., M.M., A.V. and M.C. conceived the experiments, which were essentially performed by J.K. and M.C. L.D. and M.M. provided crucial reagents. R.M. assisted with experiments involving wasp infestation. A.V. and M.C. wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures S1- S3 with Legends. Fig. S1 illustrates PSC-specific expression driven by Pcol85-Gal4. Forced expression of the apoptosis inducing gene reaper in the PSC leads to death of the PSC cells. Fig. S2 illustrates PSC cells extend filopodia. Fig.S3 illustrates loss of col activity in the PSC does not affect the formation of the medullary zone in the second instar larvae. (PDF 258 kb)
Rights and permissions
About this article
Cite this article
Krzemień, J., Dubois, L., Makki, R. et al. Control of blood cell homeostasis in Drosophila larvae by the posterior signalling centre. Nature 446, 325–328 (2007). https://doi.org/10.1038/nature05650
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature05650
This article is cited by
-
Drosophila Hox genes induce melanized pseudo-tumors when misexpressed in hemocytes
Scientific Reports (2021)
-
Single-cell transcriptome maps of myeloid blood cell lineages in Drosophila
Nature Communications (2020)
-
Molecular control of the female germline stem cell niche size in Drosophila
Cellular and Molecular Life Sciences (2019)
-
Regulation of phagocyte triglyceride by a STAT-ATG2 pathway controls mycobacterial infection
Nature Communications (2017)
-
Vascular control of the Drosophila haematopoietic microenvironment by Slit/Robo signalling
Nature Communications (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.