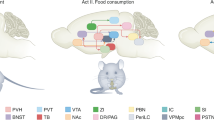
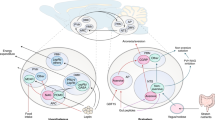
Abstract
Food intake, energy expenditure and body adiposity are homeostatically regulated. Central and peripheral signals communicate information about the current state of energy balance to key brain regions, including the hypothalamus and brainstem. Hunger and satiety represent coordinated responses to these signals, which include neural and hormonal messages from the gut. In recent years our understanding of how neural and hormonal brain–gut signalling regulates energy homeostasis has advanced considerably. Gut hormones have various physiological functions that include specifically targeting the brain to regulate appetite. New research suggests that gut hormones can be used to specifically regulate energy homeostasis in humans, and offer a target for anti-obesity drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Farooqi, I. S. & O'Rahilly, S. Monogenic obesity in humans. Annu. Rev. Med. 56, 443–458 (2005).
Halford, J. C. Pharmacotherapy for obesity. Appetite 46, 6–10 (2006).
James, W. P. et al. Effect of sibutramine on weight maintenance after weight loss: a randomised trial. STORM Study Group. Sibutramine Trial of Obesity Reduction and Maintenance. Lancet 356, 2119–2125 (2000).
Gelfand, E. V. & Cannon, C. P. Rimonabant: a cannabinoid receptor type 1 blocker for management of multiple cardiometabolic risk factors. J. Am. Coll. Cardiol. 47, 1919–1926 (2006).
Wang, H., Dey, S. K. & Maccarrone, M. Jekyll and Hyde: two faces of cannabinoid signaling in male and female fertility. Endocr. Rev. 27, 427–448 (2006).
Schwartz, M. W., Woods, S. C., Porte, D., Seeley, R. J. & Baskin, D. G. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Badman, M. K. & Flier, J. S. The gut and energy balance: visceral allies in the obesity wars. Science 307, 1909–1914 (2005).
Kojima, M. & Kangawa, K. Ghrelin: structure and function. Physiol. Rev. 85, 495–522 (2005).
Tschop, M., Smiley, D. L. & Heiman, M. L. Ghrelin induces adiposity in rodents. Nature 407, 908–913 (2000).
Theander-Carrillo, C. et al. Ghrelin action in the brain controls adipocyte metabolism. J. Clin. Invest. 116, 1983–1993 (2006).
Cummings, D. E. Ghrelin and the short- and long-term regulation of appetite and body weight. Physiol. Behav. 89, 71–84 (2006).
Dong, J. et al. Role of endogenous ghrelin in the hyperphagia of mice with streptozotocin-induced diabetes. Endocrinology 147, 2634–2642 (2006).
Sun, Y., Asnicar, M., Saha, P. K., Chan, L. & Smith, R. G. Ablation of ghrelin improves the diabetic but not obese phenotype of ob/ob mice. Cell Metab. 3, 379–386 (2006).
Zorrilla, E. P. et al. Vaccination against weight gain. Proc. Natl Acad. Sci. USA (2006).
Neary, N. M. et al. Ghrelin increases energy intake in cancer patients with impaired appetite: acute, randomized, placebo-controlled trial. J. Clin. Endocrinol. Metab. 89, 2832–2836 (2004).
Wynne, K. et al. Subcutaneous ghrelin enhances acute food intake in malnourished patients who receive maintenance peritoneal dialysis: a randomized, placebo-controlled trial. J. Am. Soc. Nephrol. 16, 2111–2118 (2005).
Murray, C. D. et al. Ghrelin enhances gastric emptying in diabetic gastroparesis: a double blind, placebo controlled, crossover study. Gut 54, 1693–1698 (2005).
Nogueiras, R., Perez-Tilve, D., Wortley, K. E. & Tschop, M. Growth hormone secretagogue (ghrelin-) receptors — a complex drug target for the regulation of body weight. CNS Neurol. Disord. Drug Targets 5, 335–343 (2006).
Pinto, S. et al. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 304, 110–115 (2004).
Diano, S. et al. Ghrelin controls hippocampal spine synapse density and memory performance. Nature Neurosci. 9, 381–388 (2006).
Zhang, J. V. et al. Obestatin, a peptide encoded by the ghrelin gene, opposes ghrelin's effects on food intake. Science 310, 996–999 (2005).
Gourcerol, G. et al. Lack of interaction between peripheral injection of CCK and obestatin in the regulation of gastric satiety signaling in rodents. Peptides 27, 2811–2819 (2006).
Holst, B. et al. GPR39 signaling is stimulated by zinc ions but not by obestatin. Endocrinology advance online publication, 7 September 2006 (doi:10.1210/en.2006-0933).
Nogueiras, R. et al. Effects of obestatin on energy balance and growth hormone secretion in rodents. Endocrinology advance online publication, 28 September 2006 (doi:10.1210/en.2006-0915).
Batterham, R. L. et al. Gut hormone PYY3–36 physiologically inhibits food intake. Nature 418, 650–654 (2002).
Tschop, M. et al. Physiology: does gut hormone PYY3–36 decrease food intake in rodents? Nature 430, 1 (2004).
Halatchev, I. G., Ellacott, K. L., Fan, W. & Cone, R. D. Peptide YY3–36 inhibits food intake in mice through a melanocortin-4 receptor-independent mechanism. Endocrinology 145, 2585–2590 (2004).
Abbott, C. R. et al. The importance of acclimatisation and habituation to experimental conditions when investigating the anorectic effects of gastrointestinal hormones in the rat. Int. J. Obes. (Lond.) 30, 288–292 (2006).
Adams, S. H., Won, W. B., Schonhoff, S. E., Leiter, A. B. & Paterniti, J. R. Effects of peptide YY3–36 on short-term food intake in mice are not affected by prevailing plasma ghrelin levels. Endocrinology 145, 4967–4975 (2004).
Challis, B. G. et al. Mice lacking pro-opiomelanocortin are sensitive to high-fat feeding but respond normally to the acute anorectic effects of peptide-YY3–36 . Proc. Natl Acad. Sci. USA 101, 4695–4700 (2004).
Chelikani, P. K., Haver, A. C. & Reidelberger, R. D. Intravenous infusion of peptide YY3–36 potently inhibits food intake in rats. Endocrinology 146, 879–888 (2005).
Cox, J. E. & Randich, A. Enhancement of feeding suppression by PYY3–36 in rats with area postrema ablations. Peptides 25, 985–989 (2004).
Pittner, R. A. et al. Effects of PYY3–36 in rodent models of diabetes and obesity. Int. J. Obes. Relat. Metab. Disord. 28, 963–971 (2004).
Moran, T. H. et al. Peptide YY3–36 inhibits gastric emptying and produces acute reductions in food intake in rhesus monkeys. Am. J. Physiol. Regul. Integr. Comp. Physiol. 288, R384–R388 (2005).
Abbott, C. R. et al. Blockade of the neuropeptide Y Y2 receptor with the specific antagonist BIIE0246 attenuates the effect of endogenous and exogenous peptide YY3–36 on food intake. Brain Res. 1043, 139–144 (2005).
Boey, D. et al. Peptide YY ablation in mice leads to the development of hyperinsulinaemia and obesity. Diabetologia 49, 1360–1370 (2006).
Batterham, R. L. et al. Critical role for peptide YY in protein-mediated satiation and body-weight regulation. Cell Metab. 4, 223–233 (2006).
Batterham, R. L. et al. Inhibition of food intake in obese subjects by peptide YY3–36 . N. Engl. J. Med. 349, 941–948 (2003).
Korner, J. et al. Effects of Roux-en-Y gastric bypass surgery on fasting and postprandial concentrations of plasma ghrelin, peptide YY, and insulin. J. Clin. Endocrinol. Metab. 90, 359–365 (2005).
le Roux, C. W. et al. Attenuated peptide YY release in obese subjects is associated with reduced satiety. Endocrinology 147, 3–8 (2006).
Kim, B. J. et al. Peptide YY is secreted after oral glucose administration in a gender-specific manner. J. Clin. Endocrinol. Metab. 90, 6665–6671 (2005).
Stock, S. et al. Ghrelin, peptide YY, glucose-dependent insulinotropic polypeptide, and hunger responses to a mixed meal in anorexic, obese, and control female adolescents. J. Clin. Endocrinol. Metab. 90, 2161–2168 (2005).
Halatchev, I. G. & Cone, R. D. Peripheral administration of PYY3–36 produces conditioned taste aversion in mice. Cell Metab. 1, 159–168 (2005).
Chelikani, P. K., Haver, A. C. & Reidelberger, R. D. Dose-dependent effects of peptide YY3–36 on conditioned taste aversion in rats. Peptides advance online publication 6 September 2006 (doi:10.1016/j.peptides.2006.08.001).
Degen, L. et al. Effect of peptide YY3–36 on food intake in humans. Gastroenterology 129, 1430–1436 (2005).
Scott, V., Kimura, N., Stark, J. A. & Luckman, S. M. Intravenous peptide YY3–36 and Y2 receptor antagonism in the rat: effects on feeding behaviour. J. Neuroendocrinol. 17, 452–457 (2005).
Chelikani, P. K., Haver, A. C., Reeve, J. R., Keire, D. A. & Reidelberger, R. D. Daily, intermittent intravenous infusion of peptide YY3–36 reduces daily food intake and adiposity in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R298–R305 (2006).
Bi, S. & Moran, T. H. Actions of CCK in the controls of food intake and body weight: lessons from the CCK-A receptor deficient OLETF rat. Neuropeptides 36, 171–181 (2002).
Crawley, J. N. & Beinfeld, M. C. Rapid development of tolerance to the behavioural actions of cholecystokinin. Nature 302, 703–706 (1983).
West, D. B., Fey, D. & Woods, S. C. Cholecystokinin persistently suppresses meal size but not food intake in free-feeding rats. Am. J. Physiol. 246, R776–R787 (1984).
West, D. B. et al. Infusion of cholecystokinin between meals into free-feeding rats fails to prolong the intermeal interval. Physiol. Behav. 39, 111–115 (1987).
Moran, T. H. Cholecystokinin and satiety: current perspectives. Nutrition 16, 858–865 (2000).
Greenough, A., Cole, G., Lewis, J., Lockton, A. & Blundell, J. Untangling the effects of hunger, anxiety, and nausea on energy intake during intravenous cholecystokinin octapeptide (CCK-8) infusion. Physiol. Behav. 65, 303–310 (1998).
Drucker, D. J. The biology of incretin hormones. Cell Metab. 3, 153–165 (2006).
Wynne, K. et al. Subcutaneous oxyntomodulin reduces body weight in overweight and obese subjects: a double-blind, randomized, controlled trial. Diabetes 54, 2390–2395 (2005).
Katsuura, G., Asakawa, A. & Inui, A. Roles of pancreatic polypeptide in regulation of food intake. Peptides 23, 323–329 (2002).
Batterham, R. L. et al. Pancreatic polypeptide reduces appetite and food intake in humans. J. Clin. Endocrinol. Metab. 88, 3989–3992 (2003).
Asakawa, A. et al. Characterization of the effects of pancreatic polypeptide in the regulation of energy balance. Gastroenterology 124, 1325–1336 (2003).
Schmidt, P. T. et al. A role for pancreatic polypeptide in the regulation of gastric emptying and short-term metabolic control. J. Clin. Endocrinol. Metab. 90, 5241–5246 (2005).
Whitehouse, F. et al. A randomized study and open-label extension evaluating the long-term efficacy of pramlintide as an adjunct to insulin therapy in type 1 diabetes. Diabetes Care 25, 724–730 (2002).
Ratner, R. et al. Adjunctive therapy with pramlintide lowers HbA1c without concomitant weight gain and increased risk of severe hypoglycemia in patients with type 1 diabetes approaching glycemic targets. Exp. Clin. Endocrinol. Diabetes 113, 199–204 (2005).
Miyawaki, K. et al. Inhibition of gastric inhibitory polypeptide signaling prevents obesity. Nature Med. 8, 738–742 (2002).
Miyawaki, K. et al. Glucose intolerance caused by a defect in the entero-insular axis: a study in gastric inhibitory polypeptide receptor knockout mice. Proc. Natl Acad. Sci. USA 96, 14843–14847 (1999).
Buse, J. B. et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 27, 2628–2635 (2004).
DeFronzo, R. A. et al. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 28, 1092–1100 (2005).
Kendall, D. M. et al. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 28, 1083–1091 (2005).
Drucker, D. J. Biologic actions and therapeutic potential of the proglucagon-derived peptides. Nature Clin. Pract. Endocrinol. Metab. 1, 22–31 (2005).
Dakin, C. L. et al. Peripheral oxyntomodulin reduces food intake and body weight gain in rats. Endocrinology 145, 2687–2695 (2004).
Cohen, M. A. et al. Oxyntomodulin suppresses appetite and reduces food intake in humans. J. Clin. Endocrinol. Metab. 88, 4696–4701 (2003).
Baggio, L. L., Huang, Q., Brown, T. J. & Drucker, D. J. Oxyntomodulin and glucagon-like peptide-1 differentially regulate murine food intake and energy expenditure. Gastroenterology 127, 546–558 (2004).
Dakin, C. L. et al. Oxyntomodulin inhibits food intake in the rat. Endocrinology 142, 4244–4250 (2001).
Dakin, C. L. et al. Repeated ICV administration of oxyntomodulin causes a greater reduction in body weight gain than in pair-fed rats. Am. J. Physiol. Endocrinol. Metab. 283, E1173–E1177 (2002).
Wynne, K. et al. Oxyntomodulin increases energy expenditure in addition to decreasing energy intake in overweight and obese humans: a randomised controlled trial. Int. J. Obes. (Lond.) advance online publication 18 April 2006 (doi:10.1038/sj.ijo.0803344).
Martin, N. M. et al. Pre-obese and obese agouti mice are sensitive to the anorectic effects of peptide YY3–36 but resistant to ghrelin. Int. J. Obes. Relat. Metab. Disord. 28, 886–893 (2004).
Acuna-Goycolea, C. & van den Pol, A. N. Peptide YY3–36 inhibits both anorexigenic proopiomelanocortin and orexigenic neuropeptide Y neurons: implications for hypothalamic regulation of energy homeostasis. J. Neurosci. 25, 10510–10519 (2005).
Cone, R. D. Anatomy and regulation of the central melanocortin system. Nature Neurosci. 8, 571–578 (2005).
Abbott, C. R. et al. The inhibitory effects of peripheral administration of peptide YY3–36 and glucagon-like peptide-1 on food intake are attenuated by ablation of the vagal-brainstem-hypothalamic pathway. Brain Res. 1044, 127–131 (2005).
Browning, K. N. & Travagli, R. A. Neuropeptide Y and peptide YY inhibit excitatory synaptic transmission in the rat dorsal motor nucleus of the vagus. J. Physiol. (Lond.) 549, 775–785 (2003).
Chen, C. H. & Rogers, R. C. Peptide YY and the Y2 agonist PYY-(13-36) inhibit neurons of the dorsal motor nucleus of the vagus. Am. J. Physiol. 273, R213–R218 (1997).
Koda, S. et al. The role of the vagal nerve in peripheral PYY3–36-induced feeding reduction in rats. Endocrinology 146, 2369–2375 (2005).
Date, Y. et al. The role of the gastric afferent vagal nerve in ghrelin-induced feeding and growth hormone secretion in rats. Gastroenterology 123, 1120–1128 (2002).
le Roux, C. W. et al. Ghrelin does not stimulate food intake in patients with surgical procedures involving vagotomy. J. Clin. Endocrinol. Metab. 90, 4521–4524 (2005).
Kuvshinoff, B. W., Rudnicki, M. & McFadden, D. W. The effect of SMS 201-995 on meal and CCK-stimulated peptide YY release. J. Surg. Res. 50, 425–429 (1991).
Flanagan, D. E. et al. The influence of insulin on circulating ghrelin. Am. J. Physiol. Endocrinol. Metab. 284, E313–E316 (2003).
Ueno, M., Carvalheira, J. B., Oliveira, R. L., Velloso, L. A. & Saad, M. J. Circulating ghrelin concentrations are lowered by intracerebroventricular insulin. Diabetologia 49, 2449–2452 (2006).
Arafat, A. M. et al. Glucagon suppression of ghrelin secretion is exerted at hypothalamus–pituitary level. J. Clin. Endocrinol. Metab. 91, 3528–3533 (2006).
Neary, N. M. et al. Peptide YY3–36 and glucagon-like peptide-17–36 inhibit food intake additively. Endocrinology 146, 5120–5127 (2005).
Talsania, T., Anini, Y., Siu, S., Drucker, D. J. & Brubaker, P. L. Peripheral exendin-4 and peptide YY3–36 synergistically reduce food intake through different mechanisms in mice. Endocrinology 146, 3748–3756 (2005).
Besterman, H. S. et al. Gut hormones in tropical malabsorption. Br. Med. J. ii, 1252–1255 (1979).
Besterman, H. S. et al. Gut hormones in inflammatory bowel disease. Scand. J. Gastroenterol. 18, 845–852 (1983).
Adrian, T. E. et al. Peptide YY abnormalities in gastrointestinal diseases. Gastroenterology 90, 379–384 (1986).
Allen, J. M. et al. Effect of partial ileal bypass on the gut hormone responses to food in man. Digestion 28, 191–196 (1983).
Borg, C. M. et al. Progressive rise in gut hormone levels after Roux-en-Y gastric bypass suggests gut adaptation and explains altered satiety. Br. J. Surg. 93, 210–215 (2006).
Chan, J. L., Mun, E. C., Stoyneva, V., Mantzoros, C. S. & Goldfine, A. B. Peptide YY levels are elevated after gastric bypass surgery. Obesity (Silver Spring) 14, 194–198 (2006).
le Roux, C. W. et al. Gut hormone profiles following bariatric surgery favor an anorectic state, facilitate weight loss, and improve metabolic parameters. Ann. Surg. 243, 108–114 (2006).
Acknowledgements
We thank W. S. Dhillo and N. M. Martin for their assistance with the preparation of this manuscript. K.G.M. is supported by Biotechnology and Biological Sciences Research Council New Investigator Award.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
Stephen Bloom is Chief Scientific Officer, a director and a shareholder of Thiakis.
Rights and permissions
About this article
Cite this article
Murphy, K., Bloom, S. Gut hormones and the regulation of energy homeostasis. Nature 444, 854–859 (2006). https://doi.org/10.1038/nature05484
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05484
This article is cited by
-
Gut-Brain Axis Modulation of Metabolic Disorders: Exploring the Intertwined Neurohumoral Pathways and Therapeutic Prospects
Neurochemical Research (2024)
-
Functional advantages of building nanosystems using multiple molecular components
Nature Chemistry (2023)
-
Activation of gastrointestinal ileal brake response with dietary slowly digestible carbohydrates, with no observed effect on subjective appetite, in an acute randomized, double-blind, crossover trial
European Journal of Nutrition (2022)
-
The hypothalamus for whole-body physiology: from metabolism to aging
Protein & Cell (2022)
-
Gut-brain mechanisms underlying changes in disordered eating behaviour after bariatric surgery: a review
Reviews in Endocrine and Metabolic Disorders (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.