Abstract
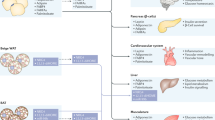
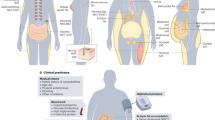
Adipocytes have been studied with increasing intensity as a result of the emergence of obesity as a serious public health problem and the realization that adipose tissue serves as an integrator of various physiological pathways. In particular, their role in calorie storage makes adipocytes well suited to the regulation of energy balance. Adipose tissue also serves as a crucial integrator of glucose homeostasis. Knowledge of adipocyte biology is therefore crucial for understanding the pathophysiological basis of obesity and metabolic diseases such as type 2 diabetes. Furthermore, the rational manipulation of adipose physiology is a promising avenue for therapy of these conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mokdad, A. H. et al. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. J. Am. Med. Assoc. 289, 76–79 (2003).
Bray, G. A. & Bellanger, T. Epidemiology, trends, and morbidities of obesity and the metabolic syndrome. Endocrine 29, 109–117 (2006).
Trayhurn, P. Endocrine and signalling role of adipose tissue: new perspectives on fat. Acta Physiol. Scand. 184, 285–293 (2005).
Pond, C. M. The Fats of Life (Cambridge Univ. Press, Cambridge, 1998).
Giorgino, F., Laviola, L. & Eriksson, J. W. Regional differences of insulin action in adipose tissue: insights from in vivo and in vitro studies. Acta Physiol. Scand. 183, 13–30 (2005).
Green, H. & Kehinde, O. An established preadipose cell line and its differentiation in culture. II. Factors affecting the adipose conversion. Cell 5, 19–27 (1975).
Farmer, S.R. Transcriptional control of adipocyte formation. Cell Metab. 4, 263–273 (2006).
Hansen, J. B. & Kristiansen, K. Regulatory circuits controlling white versus brown adipocyte differentiation. Biochem. J. 398, 153–168 (2006).
Rosen, E. D. et al. C/EBPα induces adipogenesis through PPARγ: a unified pathway. Genes Dev. 16, 22–26 (2002).
Rosen, E. D. et al. PPARγ is required for the differentiation of adipose tissue in vivo and in vitro. Mol. Cell 4, 611–617 (1999).
Tontonoz, P., Hu, E. & Spiegelman, B. M. Stimulation of adipogenesis in fibroblasts by PPARγ2, a lipid-activated transcription factor. Cell 79, 1147–1156 (1994).
Oishi, Y. et al. Kruppel-like transcription factor KLF5 is a key regulator of adipocyte differentiation. Cell Metab. 1, 27–39 (2005).
Mori, T. et al. Role of Kruppel-like factor 15 (KLF15) in transcriptional regulation of adipogenesis. J. Biol. Chem. 280, 12867–12875 (2005).
Banerjee, S. S. et al. The Kruppel-like factor KLF2 inhibits peroxisome proliferator-activated receptor-γ expression and adipogenesis. J. Biol. Chem. 278, 2581–2584 (2003).
Chen, Z., Torrens, J. I., Anand, A., Spiegelman, B. M. & Friedman, J. M. Krox20 stimulates adipogenesis via C/EBPβ-dependent and -independent mechanisms. Cell Metab. 1, 93–106 (2005).
Akerblad, P., Lind, U., Liberg, D., Bamberg, K. & Sigvardsson, M. Early B-cell factor (O/E-1) is a promoter of adipogenesis and involved in control of genes important for terminal adipocyte differentiation. Mol. Cell Biol. 22, 8015–8025 (2002).
Hansen, J. B. et al. Retinoblastoma protein functions as a molecular switch determining white versus brown adipocyte differentiation. Proc. Natl Acad. Sci. USA 101, 4112–4117 (2004).
Scime, A. et al. Rb and p107 regulate preadipocyte differentiation into white versus brown fat through repression of PGC-1α. Cell Metab. 2, 283–295 (2005).
Picard, F. et al. SRC-1 and TIF2 control energy balance between white and brown adipose tissues. Cell 111, 931–941 (2002).
Puigserver, P. et al. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 92, 829–839 (1998).
Puigserver, P. Tissue-specific regulation of metabolic pathways through the transcriptional coactivator PGC1-α. Int. J. Obes. (Lond.) 29 (Suppl. 1), S5–S9 (2005).
Uldry, M. et al. Complementary action of the PGC-1 coactivators in mitochondrial biogenesis and brown fat differentiation. Cell Metab. 3, 333–341 (2006).
Schwartz, M. W. et al. Is the energy homeostasis system inherently biased toward weight gain? Diabetes 52, 232–238 (2003).
Abizaid, A., Gao, Q. & Horvath, T. L. Thoughts for food: brain mechanisms and peripheral energy balance. Neuron 51, 691–702 (2006).
Mauer, M. M., Harris, R. B. & Bartness, T. J. The regulation of total body fat: lessons learned from lipectomy studies. Neurosci. Biobehav. Rev. 25, 15–28 (2001).
Friedman, J. M. Leptin and the regulation of body weight. Harvey Lect. 95, 107–136 (1999).
Friedman, J. M. The function of leptin in nutrition, weight, and physiology. Nutr. Rev. 60, S1–S14 (2002).
Halaas, J. L. et al. Weight-reducing effects of the plasma protein encoded by the obese gene. Science 269, 543–546 (1995).
Pelleymounter, M. A. et al. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269, 540–543 (1995).
Schwartz, M. W., Seeley, R. J., Campfield, L. A., Burn, P. & Baskin, D. G. Identification of targets of leptin action in rat hypothalamus. J. Clin. Invest. 98, 1101–1106 (1996).
Bjorbaek, C. & Kahn, B. B. Leptin signaling in the central nervous system and the periphery. Recent Prog. Horm. Res. 59, 305–331 (2004).
Fei, H. et al. Anatomic localization of alternatively spliced leptin receptors (Ob-R) in mouse brain and other tissues. Proc. Natl Acad. Sci. USA 94, 7001–7005 (1997).
Balthasar, N. et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 123, 493–505 (2005).
Grill, H. J. Distributed neural control of energy balance: contributions from hindbrain and hypothalamus. Obesity (Silver Spring) 14 (Suppl. 5), 216S–221S (2006).
Bartness, T. J. & Bamshad, M. Innervation of mammalian white adipose tissue: implications for the regulation of total body fat. Am. J. Physiol. 275, R1399–R1411 (1998).
Bartness, T. J., Kay Song, C., Shi, H., Bowers, R. R. & Foster, M. T. Brain–adipose tissue cross talk. Proc. Nutr. Soc. 64, 53–64 (2005).
Yamada, T. et al. Signals from intra-abdominal fat modulate insulin and leptin sensitivity through different mechanisms: neuronal involvement in food-intake regulation. Cell Metab. 3, 223–229 (2006).
Bachman, E. S. et al. βAR signaling required for diet-induced thermogenesis and obesity resistance. Science 297, 843–845 (2002).
Lowell, B. B. et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature 366, 740–742 (1993).
Enerback, S. et al. Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Nature 387, 90–94 (1997).
Abu-Elheiga, L., Matzuk, M. M., Abo-Hashema, K. A. & Wakil, S. J. Continuous fatty acid oxidation and reduced fat storage in mice lacking acetyl-CoA carboxylase 2. Science 291, 2613–2616 (2001).
Parker, M. G., Christian, M. & White, R. The nuclear receptor co-repressor RIP140 controls the expression of metabolic gene networks. Biochem. Soc. Trans. 34, 1103–1106 (2006).
Qi, L. et al. TRB3 links the E3 ubiquitin ligase COP1 to lipid metabolism. Science 312, 1763–1766 (2006).
Herman, M. A. & Kahn, B. B. Glucose transport and sensing in the maintenance of glucose homeostasis and metabolic harmony. J. Clin. Invest. 116, 1767–1775 (2006).
Tirone, T. A. & Brunicardi, F. C. Overview of glucose regulation. World J. Surg. 25, 461–467 (2001).
Kahn, B. B. Lilly lecture 1995. Glucose transport: pivotal step in insulin action. Diabetes 45, 1644–1654 (1996).
Schwartz, M. W. et al. Specificity of leptin action on elevated blood glucose levels and hypothalamic neuropeptide Y gene expression in ob/ob mice. Diabetes 45, 531–535 (1996).
Farooqi, I. S. et al. Beneficial effects of leptin on obesity, T cell hyporesponsiveness, and neuroendocrine/metabolic dysfunction of human congenital leptin deficiency. J. Clin. Invest. 110, 1093–1103 (2002).
Shimomura, I., Hammer, R. E., Ikemoto, S., Brown, M. S. & Goldstein, J. L. Leptin reverses insulin resistance and diabetes mellitus in mice with congenital lipodystrophy. Nature 401, 73–76 (1999).
Oral, E. A. et al. Leptin-replacement therapy for lipodystrophy. N. Engl. J. Med. 346, 570–578 (2002).
Heymsfield, S. B. et al. Recombinant leptin for weight loss in obese and lean adults: a randomized, controlled, dose-escalation trial. J. Am Med. Assoc. 282, 1568–1575 (1999).
Minokoshi, Y. et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature 415, 339–343 (2002).
Kamohara, S., Burcelin, R., Halaas, J. L., Friedman, J. M. & Charron, M. J. Acute stimulation of glucose metabolism in mice by leptin treatment. Nature 389, 374–377 (1997).
Kieffer, T. J. & Habener, J. F. The adipoinsular axis: effects of leptin on pancreatic β-cells. Am. J. Physiol. Endocrinol. Metab. 278, E1–E14 (2000).
Covey, S. D. et al. The pancreatic β cell is a key site for mediating the effects of leptin on glucose homeostasis. Cell Metab. 4, 291–302 (2006).
Kadowaki, T. et al. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J. Clin. Invest. 116, 1784–1792 (2006).
Waki, H. et al. Impaired multimerization of human adiponectin mutants associated with diabetes. Molecular structure and multimer formation of adiponectin. J. Biol. Chem. 278, 40352–40363 (2003).
Pajvani, U. B. et al. Structure–function studies of the adipocyte-secreted hormone Acrp30/adiponectin. Implications for metabolic regulation and bioactivity. J. Biol. Chem. 278, 9073–9085 (2003).
Yamauchi, T. et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 423, 762–769 (2003).
Hug, C. et al. T-cadherin is a receptor for hexameric and high-molecular-weight forms of Acrp30/adiponectin. Proc. Natl Acad. Sci. USA 101, 10308–10313 (2004).
Arita, Y. et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem. Biophys. Res. Commun. 257, 79–83 (1999).
Yatagai, T. et al. Hypoadiponectinemia is associated with visceral fat accumulation and insulin resistance in Japanese men with type 2 diabetes mellitus. Metabolism 52, 1274–1278 (2003).
Hotta, K. et al. Circulating concentrations of the adipocyte protein adiponectin are decreased in parallel with reduced insulin sensitivity during the progression to type 2 diabetes in rhesus monkeys. Diabetes 50, 1126–1133 (2001).
Kubota, N. et al. Disruption of adiponectin causes insulin resistance and neointimal formation. J. Biol. Chem. 277, 25863–25866 (2002).
Ma, K. et al. Increased β-oxidation but no insulin resistance or glucose intolerance in mice lacking adiponectin. J. Biol. Chem. 277, 34658–34661 (2002).
Maeda, N. et al. Diet-induced insulin resistance in mice lacking adiponectin/ACRP30. Nature Med. 8, 731–737 (2002).
Nawrocki, A. R. et al. Mice lacking adiponectin show decreased hepatic insulin sensitivity and reduced responsiveness to peroxisome proliferator-activated receptor γ agonists. J. Biol. Chem. 281, 2654–2660 (2006).
Kubota, N. et al. Pioglitazone ameliorates insulin resistance and diabetes by both adiponectin-dependent and -independent pathways. J. Biol. Chem. 281, 8748–8755 (2006).
Maeda, N. et al. PPARγ ligands increase expression and plasma concentrations of adiponectin, an adipose-derived protein. Diabetes 50, 2094–2099 (2001).
Winzell, M. S., Nogueiras, R., Dieguez, C. & Ahren, B. Dual action of adiponectin on insulin secretion in insulin-resistant mice. Biochem. Biophys. Res. Commun. 321, 154–160 (2004).
Fukuhara, A. et al. Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science 307, 426–430 (2005).
Stephens, J. M. & Vidal-Puig, A. J. An update on visfatin/pre-B cell colony-enhancing factor, an ubiquitously expressed, illusive cytokine that is regulated in obesity. Curr. Opin. Lipidol. 17, 128–131 (2006).
Yang, H., Lavu, S. & Sinclair, D. A. Nampt/PBEF/Visfatin: a regulator of mammalian health and longevity? Exp. Gerontol. 41, 718–726 (2006).
Yang, R. Z. et al. Identification of omentin as a novel depot-specific adipokine in human adipose tissue: possible role in modulating insulin action. Am. J. Physiol. Endocrinol. Metab. 290, E1253–E1261 (2006).
Hotamisligil, G. S. The role of TNFα and TNF receptors in obesity and insulin resistance. J. Intern. Med. 245, 621–625 (1999).
Xu, H. et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Invest. 112, 1821–1830 (2003).
Carey, A. L. et al. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes 55, 2688–2697 (2006).
Rotter, V., Nagaev, I. & Smith, U. Interleukin-6 (IL-6) induces insulin resistance in 3T3-L1 adipocytes and is, like IL-8 and tumor necrosis factor-α, overexpressed in human fat cells from insulin-resistant subjects. J. Biol. Chem. 278, 45777–45784 (2003).
Hirosumi, J. et al. A central role for JNK in obesity and insulin resistance. Nature 420, 333–336 (2002).
Shoelson, S. E., Lee, J. & Yuan, M. Inflammation and the IKKβ/IκB/NF-κB axis in obesity- and diet-induced insulin resistance. Int. J. Obes. Relat. Metab. Disord. 27 (Suppl. 3), S49–S52 (2003).
Howard, J. K. & Flier, J. S. Attenuation of leptin and insulin signaling by SOCS proteins. Trends Endocrinol. Metab. 17, 365–371 (2006).
Houstis, N., Rosen, E. D. & Lander, E. S. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature 440, 944–948 (2006).
Steppan, C. M. et al. A family of tissue-specific resistin-like molecules. Proc. Natl Acad. Sci. USA 98, 502–506 (2001).
Steppan, C. M. et al. The hormone resistin links obesity to diabetes. Nature 409, 307–312 (2001).
Banerjee, R. R. et al. Regulation of fasted blood glucose by resistin. Science 303, 1195–1198 (2004).
Steppan, C. M. & Lazar, M. A. The current biology of resistin. J. Intern. Med. 255, 439–447 (2004).
Patel, S. D., Rajala, M. W., Rossetti, L., Scherer, P. E. & Shapiro, L. Disulfide-dependent multimeric assembly of resistin family hormones. Science 304, 1154–1158 (2004).
Kaser, S. et al. Resistin messenger-RNA expression is increased by proinflammatory cytokines in vitro. Biochem. Biophys. Res. Commun. 309, 286–290 (2003).
Patel, L. et al. Resistin is expressed in human macrophages and directly regulated by PPARγ activators. Biochem. Biophys. Res. Commun. 300, 472–476 (2003).
Yang, Q. et al. Serum retinol binding protein 4 contributes to insulin resistance in obesity and type 2 diabetes. Nature 436, 356–362 (2005).
Graham, T. E. et al. Retinol-binding protein 4 and insulin resistance in lean, obese, and diabetic subjects. N. Engl. J. Med. 354, 2552–2563 (2006).
Roden, M. et al. Mechanism of free fatty acid-induced insulin resistance in humans. J. Clin. Invest. 97, 2859–2865 (1996).
Roden, M. et al. Effects of free fatty acid elevation on postabsorptive endogenous glucose production and gluconeogenesis in humans. Diabetes 49, 701–707 (2000).
Griffin, M. E. et al. Free fatty acid-induced insulin resistance is associated with activation of protein kinase C theta and alterations in the insulin signaling cascade. Diabetes 48, 1270–1274 (1999).
Schmitz-Peiffer, C. et al. Alterations in the expression and cellular localization of protein kinase C isozymes epsilon and theta are associated with insulin resistance in skeletal muscle of the high-fat-fed rat. Diabetes 46, 169–178 (1997).
Paolisso, G. et al. Does free fatty acid infusion impair insulin action also through an increase in oxidative stress? J. Clin. Endocrinol. Metab. 81, 4244–4248 (1996).
Hajduch, E. et al. Ceramide impairs the insulin-dependent membrane recruitment of protein kinase B leading to a loss in downstream signalling in L6 skeletal muscle cells. Diabetologia 44, 173–183 (2001).
Shi, H. et al. TLR4 links innate immunity and fatty acid-induced insulin resistance. J. Clin. Invest. 116, 3015–3025 (2006).
Song, M. J., Kim, K. H., Yoon, J. M. & Kim, J. B. Activation of Toll-like receptor 4 is associated with insulin resistance in adipocytes. Biochem. Biophys. Res. Commun. 346, 739–745 (2006).
Eldor, R. & Raz, I. Lipotoxicity versus adipotoxicity — the deleterious effects of adipose tissue on beta cells in the pathogenesis of type 2 diabetes. Diabetes Res. Clin. Pract. 74, S3–S8 (2006).
Lowell, B. B. & Shulman, G. I. Mitochondrial dysfunction and type 2 diabetes. Science 307, 384–387 (2005).
Simha, V. & Garg, A. Lipodystrophy: lessons in lipid and energy metabolism. Curr. Opin. Lipidol. 17, 162–169 (2006).
Yamauchi, T. et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nature Med. 7, 941–946 (2001).
Weisberg, S. P. et al. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 112, 1796–1808 (2003).
Weisberg, S. P. et al. CCR2 modulates inflammatory and metabolic effects of high-fat feeding. J. Clin. Invest. 116, 115–124 (2006).
Kanda, H. et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Invest. 116, 1494–1505 (2006).
Kamei, N. et al. Overexpression of monocyte chemoattractant protein-1 in adipose tissues causes macrophage recruitment and insulin resistance. J. Biol. Chem. 281, 26602–26614 (2006).
Di Gregorio, G. B. et al. Expression of CD68 and macrophage chemoattractant protein-1 genes in human adipose and muscle tissues: association with cytokine expression, insulin resistance, and reduction by pioglitazone. Diabetes 54, 2305–2313 (2005).
Gnudi, L., Tozzo, E., Shepherd, P. R., Bliss, J. L. & Kahn, B. B. High level overexpression of glucose transporter-4 driven by an adipose-specific promoter is maintained in transgenic mice on a high fat diet, but does not prevent impaired glucose tolerance. Endocrinology 136, 995–1002 (1995).
Abel, E. D. et al. Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature 409, 729–733 (2001).
Fukuchi, K. et al. Radionuclide imaging metabolic activity of brown adipose tissue in a patient with pheochromocytoma. Exp. Clin. Endocrinol. Diabetes 112, 601–603 (2004).
Acknowledgements
We thank B. Lowell and M. Herman for a critical reading of the manuscript.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Rosen, E., Spiegelman, B. Adipocytes as regulators of energy balance and glucose homeostasis. Nature 444, 847–853 (2006). https://doi.org/10.1038/nature05483
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05483
This article is cited by
-
Polarized Th2 cells attenuate high-fat-diet induced obesity through the suppression of lipogenesis
BMC Immunology (2024)
-
Obesity causes mitochondrial fragmentation and dysfunction in white adipocytes due to RalA activation
Nature Metabolism (2024)
-
Madhuca longifolia-hydro-ethanolic-fraction reverses mitochondrial dysfunction and modulates selective GLUT expression in diabetic mice fed with high fat diet
Molecular Biology Reports (2024)
-
Fat as a Friend or Foe of the Bone
Current Osteoporosis Reports (2024)
-
Arsenic and adipose tissue: an unexplored pathway for toxicity and metabolic dysfunction
Environmental Science and Pollution Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.