Abstract
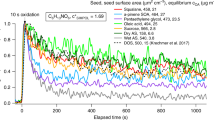
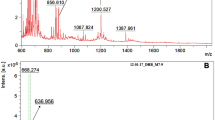
Biemann1 calls into question our preliminary interpretations of our experimental results2. A comparison of laboratory and flight measurements should settle the uncertainties he raises. In addition to evaluating instrumental characteristics such as the 'piston effect', a technique we used for injecting oven-gas content for gas chromatography–mass spectrometry (GC–MS), such studies should enable a wide range of possible compositions for Titan's aerosols to be investigated. Our aerosol collection and pyrolysis (ACP) measurements in Titan's atmosphere can then be revisited.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Biemann, K. Nature 444, doi:10.1038/nature05417 (2006).
Israël, G. et al. Nature 438, 796–799 (2005).
Khare, B. N. et al. Icarus 60, 127–137 (1984).
McDonald, G. D. et al. Icarus 108, 137–145 (1994).
Coll, P. et al. Planet. Space Sci. 47, 1331–1340 (1999).
Ramirez, S. I. et al. Icarus 156, 515–529 (2002).
Bernard, J. M. et al. Icarus 185, 301–307 (2006).
Thompson, W. R., Henry, T. J., Schwartz, J. M., Khare, B. N. & Sagan, C. Origins of Life 19, 475–476 (1989).
Lichtin, D. A. & Lin, M. C. Chem. Phys. 104, 325–330 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Israël, G., Szopa, C., Raulin, F. et al. Complex organic matter in Titan's aerosols? (Reply). Nature 444, E6–E7 (2006). https://doi.org/10.1038/nature05418
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05418
This article is cited by
-
Results from the Huygens probe on Titan
The Astronomy and Astrophysics Review (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.