Abstract
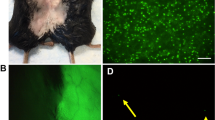
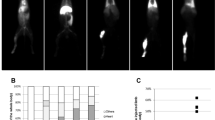
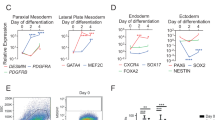
Duchenne muscular dystrophy remains an untreatable genetic disease that severely limits motility and life expectancy in affected children. The only animal model specifically reproducing the alterations in the dystrophin gene and the full spectrum of human pathology is the golden retriever dog model. Affected animals present a single mutation in intron 6, resulting in complete absence of the dystrophin protein, and early and severe muscle degeneration with nearly complete loss of motility and walking ability. Death usually occurs at about 1 year of age as a result of failure of respiratory muscles. Here we report that intra-arterial delivery of wild-type canine mesoangioblasts (vessel-associated stem cells) results in an extensive recovery of dystrophin expression, normal muscle morphology and function (confirmed by measurement of contraction force on single fibres). The outcome is a remarkable clinical amelioration and preservation of active motility. These data qualify mesoangioblasts as candidates for future stem cell therapy for Duchenne patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
20 February 2013
Nature 444, 574–579 (2006), doi:10.1038/nature05282 In Fig. 4b of this Article, the gel for the loading control MyHC for the dog Vaccin was an unintentional duplication of the loading control for the dog Vampire (which is correct). The correct gel is shown below in Fig. 1. The error does not affect any of our results.
12 March 2014
Nature 444, 574–579 (2006), doi:10.1038/nature05282 and corrigendum Nature 494, 506 (2013); doi:10.1038/nature11976 In Fig. 4b of this Article, the gel for the loading control MyHC for the dog Varus was an unintentional duplication of the loading controls for the dog Vampire (which is correct). The correct gel is shown below in Fig.
References
Emery, A. E. The muscular dystrophies. Lancet 359, 687–695 (2002)
Cossu, G. & Sampaolesi, M. New therapies for muscular dystrophy: cautious optimism. Trends Mol. Med. 10, 516–520 (2004)
Qu-Petersen, Z. et al. Identification of a novel population of muscle stem cells in mice: potential for muscle regeneration. J. Cell Biol. 157, 851–864 (2002)
Sampaolesi, M. et al. Cell therapy of α-sarcoglycan null dystrophic mice through intra-arterial delivery of mesoangioblasts. Science 301, 487–492 (2003)
Bachrach, E. et al. Systemic delivery of human microdystrophin to regenerating mouse dystrophic muscle by muscle progenitor cells. Proc. Natl Acad. Sci. USA 101, 3581–3586 (2004)
Torrente, Y. et al. Human circulating AC133+ stem cells restore dystrophin expression and ameliorate function in dystrophic skeletal muscle. J. Clin. Invest. 114, 182–195 (2004)
Rodriguez, A. M. et al. Transplantation of a multipotent cell population from human adipose tissue induces dystrophin expression in the immunocompetent mdx mouse. J. Exp. Med. 201, 1397–1405 (2005)
Dezawa, M. et al. Bone marrow stromal cells generate muscle cells and repair muscle degeneration. Science 309, 314–317 (2005)
Kornegay, J. N., Tuler, S. M., Miller, D. M. & Levesque, D. C. Muscular dystrophy in a litter of golden retriever dogs. Muscle Nerve 11, 1056–1064 (1988)
Sharp, N. J. et al. An error in dystrophin mRNA processing in golden retriever muscular dystrophy, an animal homologue of Duchenne muscular dystrophy. Genomics 13, 115–121 (1992)
Harper, S. Q. et al. Modular flexibility of dystrophin: implication for gene therapy of Duchenne muscular dystrophy. Nature Med. 8, 253–261 (2004)
Minasi, M. G. et al. The meso-angioblast: a multipotent, self-renewing cell that originates from the dorsal aorta and differentiates into most mesodermal tissues. Development 129, 2773–2783 (2002)
Galvez, B. G. et al. Complete rescue of dystrophic muscle by mesoangioblasts with enhanced migratory ability. J. Cell Biol. 174, 231–243 (2006)
Kornegay, J. N., Cundiff, D. D., Bogan, D. J., Bogan, J. R. & Okamura, C. S. The cranial sartorius muscle undergoes true hypertrophy in dogs with golden retriever muscular dystrophy. Neuromuscul. Disord. 13, 493–500 (2003)
Galli, D. et al. Mesoangioblasts, vessel-associated multipotent stem cells, repair the infarcted heart by multiple cellular mechanisms: a comparison with bone marrow progenitors, fibroblasts, and endothelial cells. Arterioscler. Thromb. Vasc. Biol. 25, 692–697 (2005)
Childers, M. K. et al. Skinned single fibers from normal and dystrophin-deficient dogs incur comparable stretch-induced force deficits. Muscle Nerve 31, 768–771 (2005)
Pavlath, G. K. Regulation of class I MHC expression in skeletal muscle: deleterious effect of aberrant expression on myogenesis. J. Neuroimmunol. 125, 42–50 (2002)
Krampera, M. et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101, 3722–3729 (2003)
Reinhardt, R. L. et al. Visualizing the generation of memory CD4 T cells in the whole body. Nature 410, 101–105 (2001)
Dell’Agnola, C. et al. Hematopoietic stem cell transplantation does not restore dystrophin expression in Duchenne muscular dystrophy dogs. Blood 104, 4311–4318 (2004)
Karpati, G., Gilbert, R., Petrof, B. J. & Nalbantoglu, J. Gene therapy research for Duchenne and Becker muscular dystrophies. Curr. Opin. Neurol. 10, 430–435 (1997)
Howell, J. M. et al. Use of the dog model for Duchenne muscular dystrophy in gene therapy trials. Neuromuscul. Disord. 7, 325–328 (1997)
Cerletti, M. et al. Dystrophic phenotype of canine X-linked muscular dystrophy is mitigated by adenovirus-mediated utrophin gene transfer. Gene Ther. 10, 750–757 (2003)
Emery, A. E. Clinical and molecular studies in Duchenne muscular dystrophy. Prog. Clin. Biol. Res. 306, 15–28 (1989)
Ricordi, C. & Strom, T. M. Clinical islet transplantation: advances and clinical challenges. Nature Rev. Immunol. 4, 259–270 (2004)
Acknowledgements
We thank M. G. Roncarolo for helpful discussions; J. Chamberlain for the gift of the lentiviral vector expressing human microdystrophin; C. Drougard for technical assistance; X. Cauchois, I. Gruyer, S. Kouamé, E. Wembe and A. Brindejont and M. Carré at the Centre d'Elevage du Domaine des Souches for their care of the dogs; and N. Borenstein for the systemic delivery of cells. M.S. and G.C. thank P. Luban for a donation. This work was supported by grants from the Association Française contre les Myopathies, Telethon, the Muscular Dystrophy Association, the Parent Project Onlus, the European Community ‘Eurostemcell’, the Cariplo Foundation and the Italian Ministries of Health and Research. B.G.G. was supported by a 3+3 fellowship from the Centro Nacional de Investigationes Cardiovasculares, Spain. Author Contributions M.S. coordinated the work on cells with R.T. and M.G.C.D.; S.B. coordinated the work on dogs with N.G., J.L.T. and I.B.; R.B. and G.D.A. coordinated the physiology experiments with O.P., C.R. and P.M., who also developed, with S.M., the instrument to measure dog tetanic force; A.I. did the histology work; B.G.G. performed the homing experiments; L.P. and M.S. conducted the western blot analysis; M.G. did the immunology experiment; Y.T. and C.B. evaluated the clinical aspects of the work; G.C. coordinated the whole project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods, Supplementary Figures and their legends. (PDF 24478 kb)
Supplementary Video 1
Untreated GRMD dog (MPG 5474 kb)
Supplementary Video 2
Valgus, GRMD dog treated with donor mesoangioblasts; inset movie, Valgus before the first treatment. (MPG 6378 kb)
Supplementary Video 3
Varus, GRMD dog treated with donor mesoangioblasts. (MPG 7875 kb)
Supplementary Video 4
Vampire, GRMD dog treated with autologous genetically modified mesoangioblasts. (MPG 2516 kb)
Supplementary Video 5
Azor, GRMD dog treated late with donor mesoangioblasts; inset movie, Azor before the first treatment. (MPG 7454 kb)
Supplementary Video 6
Azur, GRMD dog treated late with donor mesoangioblasts; inset movie, Azur before the first treatment. (MPG 4804 kb)
Rights and permissions
About this article
Cite this article
Sampaolesi, M., Blot, S., D’Antona, G. et al. Mesoangioblast stem cells ameliorate muscle function in dystrophic dogs. Nature 444, 574–579 (2006). https://doi.org/10.1038/nature05282
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05282
This article is cited by
-
Cell-mediated exon skipping normalizes dystrophin expression and muscle function in a new mouse model of Duchenne Muscular Dystrophy
EMBO Molecular Medicine (2024)
-
Dystrophin Expressing Chimeric (DEC) Cell Therapy for Duchenne Muscular Dystrophy: A First-in-Human Study with Minimum 6 Months Follow-up
Stem Cell Reviews and Reports (2023)
-
Cell-Based and Gene-Based Therapy Approaches in Neuro-orthopedic Disorders: a Literature Review
Regenerative Engineering and Translational Medicine (2023)
-
Human MuStem cells repress T-cell proliferation and cytotoxicity through both paracrine and contact-dependent pathways
Stem Cell Research & Therapy (2022)
-
Long-Term Protective Effect of Human Dystrophin Expressing Chimeric (DEC) Cell Therapy on Amelioration of Function of Cardiac, Respiratory and Skeletal Muscles in Duchenne Muscular Dystrophy
Stem Cell Reviews and Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.