Abstract
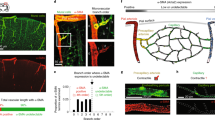
Neural activity increases local blood flow in the central nervous system (CNS), which is the basis of BOLD (blood oxygen level dependent) and PET (positron emission tomography) functional imaging techniques1,2,3. Blood flow is assumed to be regulated by precapillary arterioles, because capillaries lack smooth muscle. However, most (65%) noradrenergic innervation of CNS blood vessels terminates near capillaries rather than arterioles4, and in muscle and brain a dilatory signal propagates from vessels near metabolically active cells to precapillary arterioles5,6, suggesting that blood flow control is initiated in capillaries. Pericytes, which are apposed to CNS capillaries and contain contractile proteins7, could initiate such signalling. Here we show that pericytes can control capillary diameter in whole retina and cerebellar slices. Electrical stimulation of retinal pericytes evoked a localized capillary constriction, which propagated at ∼2 µm s-1 to constrict distant pericytes. Superfused ATP in retina or noradrenaline in cerebellum resulted in constriction of capillaries by pericytes, and glutamate reversed the constriction produced by noradrenaline. Electrical stimulation or puffing GABA (γ-amino butyric acid) receptor blockers in the inner retina also evoked pericyte constriction. In simulated ischaemia, some pericytes constricted capillaries. Pericytes are probably modulators of blood flow in response to changes in neural activity, which may contribute to functional imaging signals and to CNS vascular disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Roy, C. & Sherrington, C. On the regulation of the blood supply of the brain. J. Physiol. (Lond.) 11, 85–100 (1890)
Raichle, M. E. Behind the scenes of functional brain imaging: a historical and physiological perspective. Proc. Natl Acad. Sci. USA 95, 765–772 (1998)
Attwell, D. & Iadecola, C. The neural basis of functional brain imaging signals. Trends Neurosci. 25, 621–625 (2002)
Cohen, Z., Molinatti, G. & Hamel, E. Astroglial and vascular interactions of noradrenaline terminals in the rat cerebral cortex. J. Cereb. Blood Flow Metab. 17, 894–904 (1997)
Berg, B. R., Cohen, K. D. & Sarelius, I. H. Direct coupling between blood flow and metabolism at the capillary level in striated muscle. Am. J. Physiol. 272, H2693–H2700 (1997)
Iadecola, C., Yang, G., Ebner, T. J. & Chen, G. Local and propagated vascular responses evoked by focal synaptic activity in cerebellar cortex. J. Neurophysiol. 78, 651–659 (1997)
Herman, I. M. & D'Amore, P. A. Microvascular pericytes contain muscle and nonmuscle actins. J. Cell Biol. 101, 43–52 (1985)
Rucker, H. K., Wynder, H. J. & Thomas, W. E. Cellular mechanisms of CNS pericytes. Brain Res. Bull. 51, 363–369 (2000)
Kawamura, H. et al. ATP: a vasoactive signal in the pericyte-containing microvasculature of the rat retina. J. Physiol. (Lond.) 551, 787–799 (2003)
Kawamura, H. et al. Effects of angiotensin II on the pericyte-containing microvasculature of the rat retina. J. Physiol. (Lond.) 561, 671–683 (2004)
Wu, D. M., Kawamura, H., Sakagami, K., Kobayashi, M. & Puro, D. G. Cholinergic regulation of pericyte-containing retinal microvessels. Am. J. Physiol. Heart Circ. Physiol. 284, H2083–H2090 (2003)
Hirase, H., Creso, J., Singleton, M., Bartho, P. & Buzsaki, G. Two-photon imaging of brain pericytes in vivo using dextran-conjugated dyes. Glia 46, 95–100 (2004)
Hughes, S. & Chan-Ling, T. Characterization of smooth muscle cell and pericyte differentiation in the rat retina in vivo. Invest. Ophthalmol. Vis. Sci. 45, 2795–2806 (2004)
Zonta, M. et al. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nature Neurosci. 6, 43–50 (2003)
Mulligan, S. J. & MacVicar, B. A. Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 431, 195–199 (2004)
Takano, T. et al. Astrocyte-mediated control of cerebral blood flow. Nature Neurosci. 9, 260–267 (2006)
Newman, E. A. Propagation of intercellular calcium waves in retinal astrocytes and Muller cells. J. Neurosci. 21, 2215–2223 (2001)
Leffler, C. W., Beasley, D. G. & Busija, D. W. Cerebral ischemia alters cerebral microvascular reactivity in newborn pigs. Am. J. Physiol. 257, H266–H271 (1989)
Nelson, C. W., Wei, E. P., Povlishock, J. T., Kontos, H. A. & Moskowitz, M. A. Oxygen radicals in cerebral ischemia. Am. J. Physiol. 263, H1356–H1362 (1992)
Hauck, E. F., Apostel, S., Hoffmann, J. F., Heimann, A. & Kempski, O. Capillary flow and diameter changes during reperfusion after global cerebral ischemia studied by intravital video microscopy. J. Cereb. Blood Flow Metab. 24, 383–391 (2004)
Hansen, A. J. Effect of anoxia on ion distribution in the brain. Physiol. Rev. 65, 101–148 (1985)
Butryn, R. K., Ruan, H., Hull, C. M. & Frank, R. N. Vasoactive agonists do not change the caliber of retinal capillaries of the rat. Microvasc. Res. 50, 80–93 (1995)
Schonfelder, U., Hofer, A., Paul, M. & Funk, R. H. In situ observation of living pericytes in rat retinal capillaries. Microvasc. Res. 56, 22–29 (1998)
Shepro, D. & Morel, N. M. Pericyte physiology. FASEB J. 7, 1031–1038 (1993)
Sakagami, K., Kawamura, H., Wu, D. M. & Puro, D. G. Nitric oxide/cGMP-induced inhibition of calcium and chloride currents in retinal pericytes. Microvasc. Res. 62, 196–203 (2001)
Haefliger, I. O., Zschauer, A. & Anderson, D. R. Relaxation of retinal pericyte contractile tone through the nitric oxide–cyclic guanosine monophosphate pathway. Invest. Ophthalmol. 35, 991–997 (1994)
Simard, M., Arcuino, G., Takano, T., Liu, O. S. & Nedergaard, M. Signaling at the gliovascular interface. J. Neurosci. 23, 9254–9262 (2003)
Duffy, S. & MacVicar, B. A. Adrenergic calcium signaling in astrocyte networks within the hippocampal slice. J. Neurosci. 15, 5535–5550 (1995)
Cauli, B. et al. Cortical GABA interneurons in neurovascular coupling: relays for subcortical vasoactive pathways. J. Neurosci. 24, 8940–8949 (2004)
Metea, M. R. & Newman, E. A. Glial cells dilate and constrict blood vessels: a mechanism of neurovascular coupling. J. Neurosci. 26, 2862–2870 (2006)
Acknowledgements
We thank C. Auger for initiating the experiments on cerebellar capillaries. This work was supported by the Wellcome Trust, the EU and a Wolfson-Royal Society Award. C.H. is in the 4-year PhD Programme in Neuroscience at UCL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods and Supplementary Figure and Movie legends. (PDF 435 kb)
Supplementary Movie 1
Electrically-evoked pericyte constriction and propagation of pericyte constriction along retinal capillaries. (MOV 679 kb)
Supplementary Movie 2
UTP-evoked constriction of retinal capillaries. (MOV 640 kb)
Supplementary Movie 3
Noradrenaline-evoked constriction and glutamate-evoked dilation in a molecular layer capillary in a cerebellar slice. (MOV 1107 kb)
Rights and permissions
About this article
Cite this article
Peppiatt, C., Howarth, C., Mobbs, P. et al. Bidirectional control of CNS capillary diameter by pericytes. Nature 443, 700–704 (2006). https://doi.org/10.1038/nature05193
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05193
This article is cited by
-
Sex, hormones and cerebrovascular function: from development to disorder
Fluids and Barriers of the CNS (2024)
-
Bicarbonate signalling via G protein-coupled receptor regulates ischaemia-reperfusion injury
Nature Communications (2024)
-
Systemic Immune-Inflammation Response is Associated with Futile Recanalization After Endovascular Treatment
Neurocritical Care (2024)
-
Brain Maturation as a Fundamental Factor in Immune-Neurovascular Interactions in Stroke
Translational Stroke Research (2024)
-
On the origin and development of glioblastoma: multifaceted role of perivascular mesenchymal stromal cells
Acta Neuropathologica Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.