Abstract
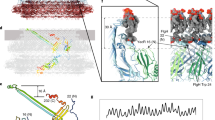
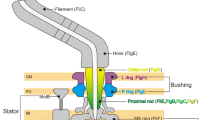
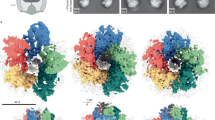
The bacterial flagellar motor is an amazing nanomachine: built from approximately 25 different proteins, it uses an electrochemical ion gradient to drive rotation at speeds of up to 300 Hz (refs 1, 2). The flagellar motor consists of a fixed, membrane-embedded, torque-generating stator and a typically bidirectional, spinning rotor that changes direction in response to chemotactic signals. Most structural analyses so far have targeted the purified rotor3,4, and hence little is known about the stator and its interactions. Here we show, using electron cryotomography of whole cells, the in situ structure of the complete flagellar motor from the spirochaete Treponema primitia at 7 nm resolution. Twenty individual motor particles were computationally extracted from the reconstructions, aligned and then averaged. The stator assembly, revealed for the first time, possessed 16-fold symmetry and was connected directly to the rotor, C ring and a novel P-ring-like structure. The unusually large size of the motor suggested mechanisms for increasing torque and supported models wherein critical interactions occur atop the C ring, where our data suggest that both the carboxy-terminal and middle domains of FliG are found.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kojima, S. & Blair, D. F. The bacterial flagellar motor: structure and function of a complex molecular machine. Int. Rev. Cytol. 233, 93–134 (2004)
Berg, H. C. The rotary motor of bacterial flagella. Annu. Rev. Biochem. 72, 19–54 (2003)
Francis, N. R., Sosinsky, G. E., Thomas, D. & DeRosier, D. J. Isolation, characterization and structure of bacterial flagellar motors containing the switch complex. J. Mol. Biol. 235, 1261–1270 (1994)
Thomas, D., Morgan, D. G. & DeRosier, D. J. Structures of bacterial flagellar motors from two FliF–FliG gene fusion mutants. J. Bacteriol. 183, 6404–6412 (2001)
Sosinsky, G. E., Francis, N. R., Stallmeyer, M. J. & DeRosier, D. J. Substructure of the flagellar basal body of Salmonella typhimurium. J. Mol. Biol. 223, 171–184 (1992)
Stallmeyer, M. J., Hahnenberger, K. M., Sosinsky, G. E., Shapiro, L. & DeRosier, D. J. Image reconstruction of the flagellar basal body of Caulobacter crescentus. J. Mol. Biol. 205, 511–518 (1989)
Suzuki, H., Yonekura, K. & Namba, K. Structure of the rotor of the bacterial flagellar motor revealed by electron cryomicroscopy and single-particle image analysis. J. Mol. Biol. 337, 105–113 (2004)
Young, H. S., Dang, H., Lai, Y., DeRosier, D. J. & Khan, S. Variable symmetry in Salmonella typhimurium flagellar motors. Biophys. J. 84, 571–577 (2003)
Khan, S., Dapice, M. & Reese, T. S. Effects of mot gene expression on the structure of the flagellar motor. J. Mol. Biol. 202, 575–584 (1988)
Khan, S., Ivey, D. M. & Krulwich, T. A. Membrane ultrastructure of alkaliphilic Bacillus species studied by rapid-freeze electron microscopy. J. Bacteriol. 174, 5123–5126 (1992)
Khan, S., Khan, I. H. & Reese, T. S. New structural features of the flagellar base in Salmonella typhimurium revealed by rapid-freeze electron microscopy. J. Bacteriol. 173, 2888–2896 (1991)
Coulton, J. W. & Murray, R. G. Cell envelope associations of Aquaspirillum serpens flagella. J. Bacteriol. 136, 1037–1049 (1978)
Yonekura, K. et al. Electron cryomicroscopic visualization of PomA/B stator units of the sodium-driven flagellar motor in liposomes. J. Mol. Biol. 357, 73–81 (2006)
Braun, T. F., Al-Mawsawi, L. Q., Kojima, S. & Blair, D. F. Arrangement of core membrane segments in the MotA/MotB proton-channel complex of Escherichia coli. Biochemistry 43, 35–45 (2004)
Limberger, R. J. The periplasmic flagellum of spirochetes. J. Mol. Microbiol. Biotechnol. 7, 30–40 (2004)
Jackson, S. & Black, S. H. Ultrastructure of Treponema pallidum Nichols following lysis by physical and chemical methods. II. Axial filaments. Arch. Mikrobiol. 76, 325–340 (1971)
Katayama, E., Shiraishi, T., Oosawa, K., Baba, N. & Aizawa, S. Geometry of the flagellar motor in the cytoplasmic membrane of Salmonella typhimurium as determined by stereo-photogrammetry of quick-freeze deep-etch replica images. J. Mol. Biol. 255, 458–475 (1996)
Charon, N. W. & Goldstein, S. F. Genetics of motility and chemotaxis of a fascinating group of bacteria: the spirochetes. Annu. Rev. Genet. 36, 47–73 (2002)
Brown, P. N., Hill, C. P. & Blair, D. F. Crystal structure of the middle and C-terminal domains of the flagellar rotor protein FliG. EMBO J. 21, 3225–3234 (2002)
Lowder, B. J., Duyvesteyn, M. D. & Blair, D. F. FliG subunit arrangement in the flagellar rotor probed by targeted cross-linking. J. Bacteriol. 187, 5640–5647 (2005)
Leadbetter, J. R., Schmidt, T. M., Graber, J. R. & Breznak, J. A. Acetogenesis from H2 plus CO2 by spirochetes from termite guts. Science 283, 686–689 (1999)
Acknowledgements
We thank E. Matson for growing the T. primitia cultures. This work was supported, in part, by NIH grants to G.J.J., a DOE grant to G.J.J., a Searle Scholar Award to G.J.J., an NSF grant to J.R.L., an NIH graduate fellowship to G.E.M., and gifts to Caltech from the Ralph M. Parsons Foundation, the Agouron Institute, and the Gordon and Betty Moore Foundation. Author Contributions G.E.M. collected and analysed the data, and drafted the text and figures; J.R.L. provided cells and discourse; and G.J.J. guided the research and manuscript editing throughout.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The averaged and symmetrized structure has been deposited in the EM Data Bank (http://www.ebi.ac.uk/msd/index.html) with the accession code EMD-1235. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
Supplementary Figures and Legends 1-5; Supplementary Video Legend; Supplementary Methods; Supplementary Notes. (PDF 620 kb)
Supplementary Video
The movie shows the reconstruction of a single intact T. primitia cell and its periplasmic flagella in three-dimensions, plus the average flagellar motor structure and its presumed rotation. (MOV 77676 kb)
Rights and permissions
About this article
Cite this article
Murphy, G., Leadbetter, J. & Jensen, G. In situ structure of the complete Treponema primitia flagellar motor. Nature 442, 1062–1064 (2006). https://doi.org/10.1038/nature05015
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature05015
This article is cited by
-
The secret lives of cells — as never seen before
Nature (2021)
-
FliL association with flagellar stator in the sodium-driven Vibrio motor characterized by the fluorescent microscopy
Scientific Reports (2018)
-
A new view into prokaryotic cell biology from electron cryotomography
Nature Reviews Microbiology (2016)
-
Architecture of the major component of the type III secretion system export apparatus
Nature Structural & Molecular Biology (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.