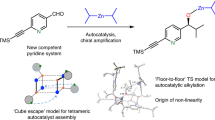
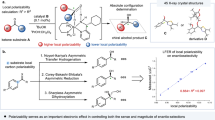
Abstract
Ever since Pasteur noticed that tartrate crystals exist in two non-superimposable forms that are mirror images of one another—as are left and right hands—the phenomenon of chirality has intrigued scientists. On the molecular level, chirality often has a profound impact on recognition and interaction events and is thus important to biochemistry and pharmacology. In chemical synthesis, much effort has been directed towards developing asymmetric synthesis strategies that yield product molecules with a significant excess of either the left-handed or right-handed enantiomer. This is usually achieved by making use of chiral auxiliaries or catalysts that influence the course of a reaction, with the enantiomeric excess (ee) of the product linearly related to the ee of the auxiliary or catalyst used. In recent years, however, an increasing number of asymmetric reactions have been documented where this relationship is nonlinear1, an effect that can lead to asymmetric amplification. Theoretical models2,3 have long suggested that autocatalytic processes can result in kinetically controlled asymmetric amplification, a prediction that has now been verified experimentally4,5,6 and rationalized mechanistically7,8,9,10,11,12,13,14 for an autocatalytic alkylation reaction. Here we show an alternative mechanism that gives rise to asymmetric amplification based on the equilibrium solid-liquid phase behaviour of amino acids in solution. This amplification mechanism is robust and can operate in aqueous systems, making it an appealing proposition for explaining one of the most tantalizing examples of asymmetric amplification—the development of high enantiomeric excess in biomolecules from a presumably racemic prebiotic world.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Girard, C. & Kagan, H. B. Nonlinear effects in asymmetric synthesis and stereoselective reactions: ten years of investigation. Angew. Chem. Int. Edn 37, 2923–2959 (1998)
Frank, F. C. Spontaneous asymmetric synthesis. Biochim. Biophys. Acta 11, 459–463 (1953)
Calvin, M. Molecular Evolution (Oxford Univ. Press, Oxford, UK, 1969)
Shibata, T., Morioka, H., Hayase, T., Choji, K. & Soai, K. Highly enantioselective catalytic asymmetric automultiplication of chiral pyrimidyl alcohol. J. Am. Chem. Soc. 118, 471–472 (1996)
Shibata, T., Choji, K., Hayase, T., Aizu, Y. & Soai, K. Asymmetric autocatalytic reaction of 3-quinolylalkanol with amplification of enantiomeric excess. Chem. Commun., 1235–1236 (1996)
Soai, K., Shibata, T., Morioka, H. & Choji, K. Asymmetric autocatalysis and amplification of enantiomeric excess of a chiral molecule. Nature 378, 767–768 (1995)
Blackmond, D. G., McMillan, C. R., Ramdeehul, S., Schorm, A. & Brown, J. M. Origins of asymmetric amplification in autocatalytic alkylzinc additions. J. Am. Chem. Soc. 123, 10103–10104 (2001)
Blackmond, D. G. Description of the condition for asymmetric amplification in autocatalytic reactions. Adv. Synth. Catal. 344, 156–158 (2002)
Buono, F. G. & Blackmond, D. G. Kinetic evidence for a tetrameric transition state in the asymmetric autocatalytic alkylation of pyrimidyl aldehydes. J. Am. Chem. Soc. 125, 8978–8979 (2003)
Buono, F. G., Iwamura, H. & Blackmond, D. G. Physical and chemical rationalization for asymmetric amplification in autocatalytic reactions. Angew. Chem. Int. Edn 43, 2099–2103 (2004)
Blackmond, D. G. Asymmetric autocatalysis and its implications for the origin of homochirality. Proc. Natl. Acad. Sci. USA 101, 5732–5736 (2004)
Gridnev, I. D. & Brown, J. M. Asymmetric autocatalysis: novel structures, novel mechanism? Proc. Natl. Acad. Sci. USA 101, 5727–5731 (2004)
Gridnev, I. D., Serafimov, J. M. & Brown, J. M. Solution structure and reagent binding of the zinc alkoxide catalyst in the Soai asymmetric autocatalytic reaction. Angew. Chem. Int. Edn 43, 4884–4887 (2004)
Gridnev, I. D., Serafimov, J. M., Quiney, H. & Brown, J. M. Reflections on spontaneous asymmetric synthesis by amplifying autocatalysis. Org. Biomol. Chem. 1, 3811–3819 (2003)
Mathew, S. P., Iwamura, H. & Blackmond, D. G. Amplification of enantiomeric excess in a proline-mediated reaction. Angew. Chem. Int. Edn 43, 3317–3321 (2004)
Iwamura, H., Mathew, S. P. & Blackmond, D. G. In situ catalyst improvement in the proline-mediated α-amination of aldehydes. J. Am. Chem. Soc. 126, 11770–11771 (2004)
Hoang, L., Bahmanyar, S., Houk, K. N. & List, B. Kinetic and stereochemical evidence for the involvement of only one proline molecule in the transition states of proline-catalyzed intra- and intermolecular aldol reactions. J. Am. Chem. Soc. 125, 16–17 (2003)
Agami, C. Mechanism of the proline-catalyzed enantioselective aldol reaction. Recent advances. Bull. Soc. Chim. Fr.(3), 499–507 (1988)
Blackmond, D. G. Kinetic aspects of nonlinear effects in asymmetric catalysis. Acc. Chem. Res. 33, 402–411 (2000)
Roozeboom, H. W. B. Solubility and melting-point as criteria for racemate compounds, pseudoracemic mix-crystals and inactive conglomerates. Z. Phys. Chem. Stoechiometrie Verwandtschaftslehre 28, 494–517 (1899)
Jacques, J., Collet, A. & Wilen, S. H. Enantiomers, Racemates and Resolution Ch. 3 (John Wiley, New York, 1981)
Rodrigo, A. A., Lorenz, H. & Seidel-Morgenstern, A. Online monitoring of preferential crystallization of enantiomers. Chirality 16, 499–508 (2004)
Podlech, J. Origin of organic molecules and biomolecular homochirality. Cell. Mol. Life Sci. 58, 44–60 (2001)
Cordova, A. et al. Acyclic amino acid-catalyzed direct asymmetric aldol reactions: alanine, the simplest stereoselective organocatalyst. Chem. Commun. 3586 (2005)
Borsenberger, V. et al. Exploratory studies to investigate a linked prebiotic origin of RNA and coded peptides. Chem. Biodivers. 1, 203–246 (2004)
Takats, Z., Nanita, S. C. & Cooks, R. G. Serine octamer reactions: indicators of prebiotic relevance. Angew. Chem. Int. Edn 42, 3521–3523 (2003)
Morowitz, H. J. A mechanism for the amplification of fluctuations in racemic mixtures. J. Theor. Biol 25, 491 (1969)
Welch, C. J. Formation of highly enantioenriched microenvironments by stochastic sorting of conglomerate crystals: a plausible mechanism for generation of enantioenrichment on the prebiotic earth. Chirality 13, 425–427 (2001)
Kondepudi, D. K. & Asakura, K. Chiral autocatalysis, spontaneous symmetry breaking, and stochastic behavior. Acc. Chem. Res. 34, 946–954 (2001)
Kondepudi, D. K., Kaufman, R. J. & Singh, N. Chiral symmetry breaking in sodium chlorate crystallization. Science 250, 975–977 (1990)
Acknowledgements
Funding from the EPSRC and AstraZeneca is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods, Supplementary Tables 1–5, Supplementary Figures 1 and 2 and Supplementary Equations 1 and 2. (PDF 101 kb)
Rights and permissions
About this article
Cite this article
Klussmann, M., Iwamura, H., Mathew, S. et al. Thermodynamic control of asymmetric amplification in amino acid catalysis. Nature 441, 621–623 (2006). https://doi.org/10.1038/nature04780
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04780
This article is cited by
-
Divergence of catalytic systems in the zinc-catalysed alkylation of benzaldehyde mediated by chiral proline-based ligands
Nature Synthesis (2024)
-
Symmetry breaking and chiral amplification in prebiotic ligation reactions
Nature (2024)
-
Condensation and asymmetric amplification of chirality in achiral molecules adsorbed on an achiral surface
Nature Communications (2023)
-
Sequential Amplification of Amino Acid Enantiomeric Excess by Conglomerate and Racemic Compound: Plausible Prebiotic Route Towards Homochirality
Origins of Life and Evolution of Biospheres (2023)
-
On the Origin of Sugar Handedness: Facts, Hypotheses and Missing Links-A Review
Origins of Life and Evolution of Biospheres (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.