Abstract
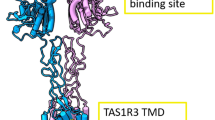
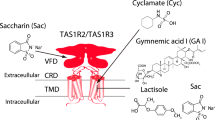
‘Water-tastes’ are gustatory after-impressions elicited by water following the removal of a chemical solution from the mouth, akin to colour after-images appearing on ‘white’ paper after fixation on coloured images. Unlike colour after-images, gustatory after-effects are poorly understood1. One theory posits that ‘water-tastes’ are adaptation phenomena, in which adaptation to one taste solution causes the water presented subsequently to act as a taste stimulus2,3. An alternative hypothesis is that removal of the stimulus upon rinsing generates a receptor-based, positive, off-response in taste-receptor cells, ultimately inducing a gustatory perception4. Here we show that a sweet ‘water-taste’ is elicited when sweet-taste inhibitors are rinsed away. Responses of cultured cells expressing the human sweetener receptor directly parallel the psychophysical responses—water rinses remove the inhibitor from the heteromeric sweetener receptor TAS1R2–TAS1R3, which activates cells and results in the perception of strong sweetness from pure water. This ‘rebound’ activity occurs when equilibrium forces on the two-state allosteric sweet receptors result in their coordinated shift to the activated state upon being released from inhibition by rinsing5,6,7.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bartoshuk, L. M. Water taste in man. Percept. Psychophys. 3, 69–72 (1968)
McBurney, D. H. & Bartoshuk, L. M. in Olfaction and taste IV (ed. Schneider, D.) 329–335 (Wissenschaftliche Verlagsgesellschaft MBH, Stuttgart, 1972)
McBurney, D. H. & Bartoshuk, L. M. Interactions between stimuli with different taste qualities. Physiol. Behav. 10, 1101–1106 (1973)
DuBois, G. E. Unraveling the biochemistry of sweet and umami tastes. Proc. Natl Acad. Sci. USA 101, 13972–13973 (2004)
Carroll, F. Y. et al. BAY36-7620: a potent non-competitive mGlu1 receptor antagonist with inverse agonist activity. Mol. Pharmacol. 59, 965–973 (2001)
Kasai, H. Tonic inhibition and rebound facilitation of a neuronal calcium channel by a GTP-binding protein. Proc. Natl Acad. Sci. USA 88, 8855–8859 (1991)
Harvey, R. D. & Belevych, A. E. Muscarinic regulation of cardiac ion channels. Br. J. Pharmacol. 139, 1074–1084 (2003)
Horne, J., Lawless, H. T., Speirs, W. & Sposato, D. Bitter taste of saccharin and acesulfame-K. Chem. Senses 27, 31–38 (2002)
Schiffman, S. S. & Gatlin, C. A. Sweeteners: State of knowledge review. Neurosci. Biobehav. Rev. 17, 313–345 (1993)
Schiffman, S. S., Booth, B. J., Losee, M. L., Pecore, S. D. & Warwick, Z. S. Bitterness of sweeteners as a function of concentration. Brain Res. Bull. 36, 505–513 (1995)
Gent, J. F. & McBurney, D. H. Time course of gustatory adaptation. Percept. Psychophys. 23, 171–175 (1978)
Li, X. et al. Human receptors for sweet and umami taste. Proc. Natl Acad. Sci. USA 99, 4692–4696 (2002)
Nelson, G. et al. Mammalian sweet taste receptors. Cell 106, 381–390 (2001)
Kuhn, C. et al. Bitter taste receptors for saccharin and acesulfame K. J. Neurosci. 24, 10260–10265 (2004)
Jiang, P. et al. Lactisole interacts with the transmembrane domains of human T1R3 to inhibit sweet taste. J. Biol. Chem. 280, 15238–15246 (2005)
Winnig, M., Bufe, B. & Meyerhof, W. Valine 738 and lysine 735 in the fifth transmembrane domain of rTas1r3 mediate insensitivity towards lactisole of the rat sweet taste receptor. BMC Neurosci. 6, 1–7 (2005)
Xu, H. et al. Different functional roles of T1R subunits in the heteromeric taste receptors. Proc. Natl Acad. Sci. USA 101, 14258–14263 (2004)
Schiffman, S. S., Booth, B. J., Sattely-Miller, E. A., Graham, B. G. & Gibes, K. M. Selective inhibition of sweetness by the sodium salt of ± 2-(4-methoxyphenoxy)propanoic acid. Chem. Senses 24, 439–447 (1999)
Dalziel, K. A kinetic interpretation of the allosteric model of Monod, Wyman and Changeaux. FEBS Lett. 1, 346–348 (1968)
Kunishima, N. et al. Structural basis of glutamate recognition by a dimeric metabotropic glutamate receptor. Nature 407, 971–977 (2000)
Samama, P., Pei, G., Costa, T., Cotecchia, S. & Lefkowitz, R. J. Negative antagonists promote an inactive conformation of the beta 2-adrenergic receptor. Mol. Pharmacol. 45, 390–394 (1994)
Cooperman, B. S. in Encyclopedia of Biological Chemistry (eds Lennarz, W. J. & Lane, M. D.) 68–73 (Elsevier, New York, 2004)
Kashlan, O. B., Scott, C. P., Lear, J. D. & Cooperman, B. S. A comrehensive model for the allosteric regulation of mammalian ribonucleotide reductase. Functional consequences of ATP- and dATP-induced oligomerization of the large subunit. Biochemistry 41, 462–474 (2002)
Christopoulos, A. & Kenakin, T. G protein-coupled receptor allosterism and complexing. Pharmacol. Rev. 54, 323–374 (2002)
Costa, T. & Cotecchia, S. Historical review: Negative efficacy and the constitutive activity of G-protein-coupled receptors. Trends Pharmacol. Sci. 26, 618–624 (2005)
Jiang, P. et al. Identification of the cyclamate interaction site within the transmembrane domain of the human sweet taste receptor subunit T1R3. J. Biol. Chem. 280, 34296–34305 (2005)
Kenakin, T. G-protein coupled receptors as allosteric machines. Receptors Channels 10, 51–60 (2004)
Pin, J. P., Galvez, T. & Prezeau, L. Evolution, structure, and activation mechanism of family 3/C G-protein-coupled receptors. Pharmacol. Ther. 98, 325–354 (2003)
Green, B. G., Dalton, P., Cowart, B. J., Shaffer, G. S. & Rankin, K. Evaluating the ‘labelled magnitude scale’ for measuring sensations of taste and smell. Chem. Senses 21, 323–334 (1996)
Bartoshuk, L. M. Comparing sensory experience across individuals: recent psychophysical advances illuminate genetic variation in taste perception. Chem. Senses 25, 447–460 (2000)
Acknowledgements
The authors thank E. Chudoba, E. Schöley-Pohl and S. Demgensky for technical assistance. We thank G. Beauchamp, B. Cooperman and D. Krautwurst for comments and discussion. We are grateful to H. Schmale for the rat Tas1r3 cDNA and J. Slack for the HEK293T Gα16gust44-expressing cell line. This research was supported in part by NIH and US-Israel BARD grants to P.A.S.B. and the German Science Foundation (W.M.). Author Contributions: V.G.-C. and P.A.S.B. discovered the phenomenon of sweet ‘water-taste’ with Na-saccharin and other sweetness inhibitors, conceived and designed the perceptual experiments and collected the behavioural data. M.W. conducted the molecular, histological and imaging experiments. B.B. participated in the functional studies and coordination of the molecular experiments. All authors contributed to the design of parallel in vivo/ex vivo control experiments and to writing and editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Figures 1–6 and Supplementary Methods. (PDF 617 kb)
Rights and permissions
About this article
Cite this article
Galindo-Cuspinera, V., Winnig, M., Bufe, B. et al. A TAS1R receptor-based explanation of sweet ‘water-taste’. Nature 441, 354–357 (2006). https://doi.org/10.1038/nature04765
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04765
This article is cited by
-
Prediction of dynamic allostery for the transmembrane domain of the sweet taste receptor subunit, TAS1R3
Communications Biology (2023)
-
Isolation and identification of sweet substances and sweet aftertaste substances in the fruit of Phyllanthus emblica
European Food Research and Technology (2022)
-
Sweet taste of heavy water
Communications Biology (2021)
-
Taste and the Gastrointestinal tract: from physiology to potential therapeutic target for obesity
International Journal of Obesity Supplements (2019)
-
The Heptahelical Domain of the Sweet Taste Receptor T1R2 Is a New Allosteric Binding Site for the Sweet Taste Modulator Amiloride That Modulates Sweet Taste in a Species-Dependent Manner
Journal of Molecular Neuroscience (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.