Abstract
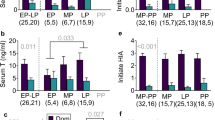
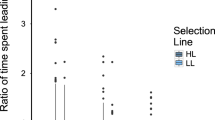
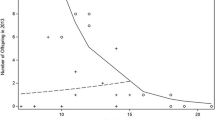
Within any hierarchical society, an individual's social rank can have profound effects on its health and reproductive success1,2, and rank-related variation in these traits is often mediated by variation in endocrine function2. Maternal effects mediated by prenatal hormone exposure are potentially important for non-genetic inheritance of phenotypic traits related to social rank3, and thus for shaping individual variation in behaviour and social structure. Here we show that androgen concentrations in wild female spotted hyaenas (Crocuta crocuta) are higher during late gestation in dominant females than in subordinate females. Furthermore, both male and female cubs born to mothers with high concentrations of androgens in late pregnancy exhibit higher rates of aggression and mounting behaviour than cubs born to mothers with lower androgen concentrations. Both behaviours are strongly affected in other mammals by organizational effects of androgens4, and both have important effects on fitness in hyaenas. Therefore, our results suggest that rank-related maternal effects of prenatal androgen exposure can adaptively influence offspring phenotype in mammals, as has previously been shown to occur in birds. They also suggest an organizational mechanism for the development of female dominance and aggressiveness in spotted hyaenas, traits that may offset the costs of extreme virilization.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Clutton-Brock, T. H. Reproductive Success (Univ. Chicago Press, Chicago, 1988)
Sapolsky, R. M. The influence of social hierarchy on primate health. Science 308, 648–652 (2005)
Schwabl, H. Yolk is a source of maternal testosterone for developing birds. Proc. Natl Acad. Sci. USA 90, 11446–11450 (1993)
Phoenix, C. H., Goy, R. W., Gerall, A. A. & Young, W. C. Organizing action of prenatally administered testosterone propionate on the tissues mediating mating behavior in the female guinea pig. Endocrinology 65, 369–382 (1959)
Mousseau, T. A. & Fox, C. W. Maternal Effects as Adaptations (Oxford Univ. Press, Oxford, 1998)
Schwabl, H. Maternal testosterone in the avian egg enhances postnatal growth. Comp. Biochem. Physiol. A 114A, 271–276 (1996)
Eising, C. M., Eikenaar, C., Schwabl, H. & Groothuis, T. G. G. Maternal androgens in black-headed gull (Larus ridibundus) eggs: consequences for chick development. Proc. Biol. Sci. 268, 839–846 (2001)
Lipar, J. L. & Ketterson, E. D. Maternally derived yolk testosterone enhances the development of the hatching muscle in the red-winged blackbird Agelaius phoeniceus. Proc. Biol. Sci. 267, 2005–2010 (2000)
Müller, W., Eising, C. M., Dijkstra, C. & Groothuis, T. G. G. Sex differences in yolk hormones depend on maternal social status in Leghorn chickens (Gallus gallus domesticus). Proc. Biol. Sci. 269, 2249–2255 (2002)
Whittingham, L. A. & Schwabl, H. Maternal testosterone in tree swallow eggs varies with female aggression. Anim. Behav. 63, 63–67 (2002)
Kruuk, H. The Spotted Hyena (Univ. Chicago Press, Chicago, 1972)
Frank, L. G. Social organisation of the spotted hyaena (Crocuta crocuta). II. Dominance and reproduction. Anim. Behav. 34, 1510–1527 (1986)
Holekamp, K. E., Smale, L. & Szykman, M. Rank and reproduction in female spotted hyenas. J. Reprod. Fertil. 108, 229–237 (1996)
Smale, L., Frank, L. G. & Holekamp, K. E. Ontogeny of dominance in free-living spotted hyaenas: juvenile rank relations with adult females and immigrant males. Anim. Behav. 46, 467–477 (1993)
Frank, L. G., Weldele, M. L. & Glickman, S. E. Masculinization costs in hyaenas. Nature 377, 584–585 (1995)
Szykman, M. et al. Rare male aggression directed toward females in a female-dominated society: Baiting behavior in the spotted hyena. Aggress. Behav. 29, 457–474 (2003)
Frank, L. G. in Carnivore Behavior, Ecology, and Evolution (ed. Gittleman, J. L.) 78–131 (Cornell Univ. Press, Ithaca, 1996)
Dloniak, S. M. et al. Non-invasive monitoring of fecal androgens in spotted hyenas (Crocuta crocuta). Gen. Comp. Endocrinol. 135, 51–61 (2004)
Yalcinkaya, T. M. et al. A mechanism for virilization of female spotted hyenas in utero. Science 260, 1929–1931 (1993)
Licht, P. et al. Androgens and masculinization of genitalia in the spotted hyaena (Crocuta crocuta). 1. Urogenital morphology and placental androgen production during fetal life. J. Reprod. Fertil. 113, 105–116 (1998)
Becker, J. B. & Breedlove, S. M. in Behavioral Endocrinology (eds Becker, J. B., Breedlove, S. M., Crews, D. & McCarthy, M. M.) 1–38 (MIT Press, Cambridge, Massachusetts, 2002)
Marchlewska-Koj, A., Kapusta, J. & Kruczek, M. Prenatal stress modifies behavior in offspring of bank voles (Clethrionomys glareolus). Physiol. Behav. 79, 671–678 (2003)
Goy, R. W., Bercovitch, F. B. & McBrair, M. C. Behavioral masculinization is independent of genital masculinization in prenatally androgenized female rhesus macaques. Horm. Behav. 22, 552–571 (1988)
East, M. L., Burke, T., Wilhelm, K., Greig, C. & Hofer, H. Sexual conflicts in spotted hyaenas: male and female mating tactics and their reproductive outcome with respect to age, social status, and tenure. Proc. R. Soc. Lond. B 270, 1247–1254 (2003)
Drea, C. M. et al. Exposure to naturally circulating androgens during foetal life incurs direct reproductive costs in female spotted hyaenas, but is prerequisite for male mating. Proc. R. Soc. Lond. B 269, 1981–1987 (2002)
Altmann, J. Observational study of behaviour: Sampling methods. Behaviour 49, 227–256 (1974)
Goymann, W., Möstl, E., Van't Hof, T., East, M. L. & Hofer, H. Noninvasive fecal monitoring of glucocorticoids in spotted hyenas, Crocuta crocuta. Gen. Comp. Endocrinol. 114, 340–348 (1999)
Acknowledgements
We thank the Office of the President of Kenya for permission to conduct field research. We also thank the Kenya Wildlife Service, the Narok County Council and the Senior Warden of the Masai Mara National Reserve for their cooperation. The following individuals provided assistance in the field: E. Boydston, M. Durham, A. Engh, L. G. Frank, P. Garrett, I. Graham, T. Harty, K. Kapheim, J. Kolowski, K. Nelson, G. Ording, L. Smale, M. Szykman, J. Tanner, K. Theis, R. Van Horn, S. Wahaj, H. Watts and K. Weibel. P. Bills and P. Van Meter provided technical assistance. This research was supported by NSF grants to K.E.H. and J.A.F., and fellowships from Michigan State University and the International Foundation for Ethical Research to S.M.D. Author Contributions Laboratory work, including development and validation of faecal hormone assays, was performed by S.M.D. and J.A.F. Field work was conducted by S.M.D. and K.E.H. The original idea for the study was conceived by K.E.H. S.M.D. conducted the statistical analyses and drafted the manuscript, which was further developed by all three authors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Dloniak, S., French, J. & Holekamp, K. Rank-related maternal effects of androgens on behaviour in wild spotted hyaenas. Nature 440, 1190–1193 (2006). https://doi.org/10.1038/nature04540
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04540
This article is cited by
-
Plasticity in metabolism of maternal androgens in avian embryos
Scientific Reports (2023)
-
Mirror image stimulation could reverse social-isolation-induced aggressiveness in the high-level subsocial lactating spider
Animal Cognition (2022)
-
Effects of prenatal testosterone on cumulative markers of oxidative damage to organs of young adult zebra finches (Taeniopygia guttata)
Journal of Comparative Physiology A (2022)
-
The value of individual identification in studies of free-living hyenas and aardwolves
Mammalian Biology (2022)
-
Suitability of drone olfactory sensitivity as a selection trait for Varroa-resistance in honeybees
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.