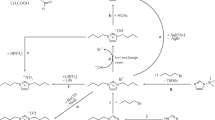
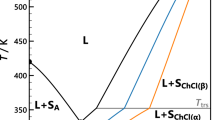
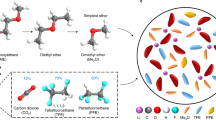
Abstract
It is widely believed that a defining characteristic of ionic liquids (or low-temperature molten salts) is that they exert no measurable vapour pressure, and hence cannot be distilled1,2. Here we demonstrate that this is unfounded, and that many ionic liquids can be distilled at low pressure without decomposition. Ionic liquids represent matter solely composed of ions, and so are perceived as non-volatile substances. During the last decade, interest in the field of ionic liquids has burgeoned3, producing a wealth of intellectual and technological challenges and opportunities for the production of new chemical and extractive processes4,5,6, fuel cells and batteries7, and new composite materials8,9. Much of this potential is underpinned by their presumed involatility. This characteristic, however, can severely restrict the attainability of high purity levels for ionic liquids (when they contain poorly volatile components) in recycling schemes, as well as excluding their use in gas-phase processes. We anticipate that our demonstration that some selected families of commonly used aprotic ionic liquids can be distilled at 200–300 °C and low pressure, with concomitant recovery of significant amounts of pure substance, will permit these currently excluded applications to be realized.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Seddon, K. R. Room-temperature ionic liquids—neoteric solvents for clean catalysis. Kinet. Catal. 37, 693–697 (1996)
Seddon, K. R. in Molten Salt Forum: Proceedings of 5th International Conference on Molten Salt Chemistry and Technology (ed. Wendt, H.) 53–62 (Trans Tech Publications, Zürich, 1998)
ISI Essential Science Indicators Special Topics: Ionic Liquidshttp://www.esi-topics.com/ionic-liquids/index.html (ISI, 2004).
Rogers, R. D. & Seddon, K. R. (eds) Ionic Liquids IIIB: Fundamentals, Progress, Challenges, and Opportunities—Transformations and Processes (American Chemical Society, Washington DC, 2005)
Rogers, R. D. & Seddon, K. R. (eds) Ionic Liquids IIIA: Fundamentals, Progress, Challenges, and Opportunities—Properties and Structure (American Chemical Society, Washington DC, 2005)
Atkins, M. P. et al. Ionic Liquids: A Map for Industrial Innovation: Q001 (January 2004) (QUILL, Belfast, 2004)
Ohno, H. (ed.) Electrochemical Aspects of Ionic Liquids (Wiley-Interscience, Hoboken, 2005)
Rogers, R. D. & Seddon, K. R. (eds) Ionic Liquids: Industrial Applications for Green Chemistry (American Chemical Society, Washington DC, 2002)
Rogers, R. D., Seddon, K. R. & Volkov, S. (eds) Green Industrial Applications of Ionic Liquids (Kluwer, Dordrecht, 2002)
Wasserscheid, P. & Welton, T. (eds) Ionic Liquids in Synthesis (Wiley-VCH, Weinheim, 2003)
Dymek, C. J. Jr, Hussey, C. L., Wilkes, J. S. & Øye, H. A. in Joint (Sixth) International Symposium on Molten Salts (eds Mamantov, G. et al.) 93–104 (The Electrochemical Society, Pennington, New Jersey, 1987)
Øye, H. A. & Wahnsiedler, W. E. in The Fourth International Symposium on Molten Salts (eds Blander, M., Newman, D. S., Saboungi, M. L., Mamantov, G. & Johnson, K.) 58–73 (The Electrochemical Society, Pennington, New Jersey, 1984)
Øye, H. A., Jagtoyen, M., Oksefjell, T. & Wilkes, J. S. Vapour pressure and thermodynamics of the system 1-methyl-3-ethylimidazolium chloride-aluminium chloride. Mater. Sci. Forum 73–75, 183–190 (1991)
Jeapes, A. J. et al. Process for recycling ionic liquids. World Patent WO 01/15175 (2001)
Yoshizawa, M., Xu, W. & Angell, C. A. Ionic liquids by proton transfer: Vapor pressure, conductivity, and the relevance of ΔpKa from aqueous solutions. J. Am. Chem. Soc. 125, 15411–15419 (2003)
Kreher, U. P., Rosamilia, A. E., Raston, C. L., Scott, J. L. & Strauss, C. R. Self-associated, “distillable” ionic media. Molecules 9, 387–393 (2004)
Abdul-Sada, A. K., Elaiwi, A. E., Greenway, A. M. & Seddon, K. R. Evidence for the clustering of substituted imidazolium salts via hydrogen bonding under the conditions of fast atom bombardment mass spectrometry. Eur. Mass Spectrom. 3, 245–247 (1997)
Rebelo, L. P. N., Canongia Lopes, J. N., Esperança, J. M. S. S. & Filipe, E. On the critical temperature, normal boiling point, and vapor pressure of ionic liquids. J. Phys. Chem. B 109, 6040–6043 (2005)
Paulechka, Y. U., Zaitsau, Dz. H., Kabo, G. J. & Strechan, A. A. Vapor pressure and thermal stability of ionic liquid 1-butyl-3-methylimidazolium Bis(trifluoromethylsulfonyl)amide. Thermochim. Acta 439, 158–160 (2005)
Maase, M. et al. Method for the separation of acids from chemical reaction mixtures by means of ionic fluids. World Patent WO 03/062171 (2003).
Baranyai, K. J., Deacon, G. B., MacFarlane, D. R., Pringle, J. M. & Scott, J. L. Thermal degradation of ionic liquids at elevated temperatures. Aust. J. Chem. 57, 145–147 (2004)
Acknowledgements
This work was supported by a grant from FC&T, Portugal (to J.M.S.S.E. and L.P.N.R.). The Belfast team wishes to thank the industrial members of QUILL for support and Al Robertson (Cytec) for supplying the phosphonium ionic liquids. For J.W.M. and J.A.W. this work represents an official contribution of the National Institute of Standards and Technology and is not subject to copyright in the USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Describes in detail the methodologies of most of the distillations performed as well as those of the NMR Spectra. (DOC 43 kb)
Supplementary Data
Plots of the 1H, 13C and 19F NMR spectra of distilled and undistilled ionic liquids corresponding to the presentation of 30 figures, labelled from Supplementary Figure 1a to Supplementary Figure 12b. (DOC 243 kb)
Supplementary Figures
Supplementary Figure 13: [C6mim][NTf2] in the sublimation apparatus: photographs of atmospheric pressure distillation; and Supplementary Figure 14: < 0.001 mbar distillation compared. (DOC 1143 kb)
Supplementary Video 1
This movie (56 seconds) shows the Kugelrohr distillation process of [C2mim][NTf2] in action – 100 times speeded up. (AVI 4359 kb)
Supplementary Video 2
This movie (50 seconds) shows the Kugelrohr distillation process of [C10mim][NTf2] in action – 150 times speeded up. (AVI 6062 kb)
Supplementary Video Legends
Video 1. Timelapse video of the distillation of [C2mim][NTf2]. Video 2. Timelapse video of the distillation of [C10mim][NTf2]. (DOC 27 kb)
Rights and permissions
About this article
Cite this article
Earle, M., Esperança, J., Gilea, M. et al. The distillation and volatility of ionic liquids. Nature 439, 831–834 (2006). https://doi.org/10.1038/nature04451
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04451
This article is cited by
-
Ionic liquids in green energy storage devices: lithium-ion batteries, supercapacitors, and solar cells
Monatshefte für Chemie - Chemical Monthly (2024)
-
Efficient extraction of lipids from magnetically separated microalgae using ionic liquids and their transesterification to biodiesel
Biomass Conversion and Biorefinery (2024)
-
Recent advances in the use of ionic liquids in the CO2 conversion to CO and C2+ hydrocarbons
Clean Technologies and Environmental Policy (2024)
-
Determination of vapor pressure, evaporation enthalpy and polarity of imidazolium-based ionic liquids
Journal of Thermal Analysis and Calorimetry (2024)
-
Role of ZnO-nano-thin-film-layered micro/nanostructured surfaces on flow boiling heat transfer characteristics
Journal of Thermal Analysis and Calorimetry (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.