Abstract
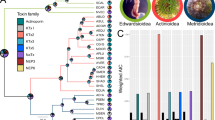
Among extant reptiles only two lineages are known to have evolved venom delivery systems, the advanced snakes and helodermatid lizards (Gila Monster and Beaded Lizard)1. Evolution of the venom system is thought to underlie the impressive radiation of the advanced snakes (2,500 of 3,000 snake species)2,3,4,5. In contrast, the lizard venom system is thought to be restricted to just two species and to have evolved independently from the snake venom system1. Here we report the presence of venom toxins in two additional lizard lineages (Monitor Lizards and Iguania) and show that all lineages possessing toxin-secreting oral glands form a clade, demonstrating a single early origin of the venom system in lizards and snakes. Construction of gland complementary-DNA libraries and phylogenetic analysis of transcripts revealed that nine toxin types are shared between lizards and snakes. Toxinological analyses of venom components from the Lace Monitor Varanus varius showed potent effects on blood pressure and clotting ability, bioactivities associated with a rapid loss of consciousness and extensive bleeding in prey. The iguanian lizard Pogona barbata retains characteristics of the ancestral venom system, namely serial, lobular non-compound venom-secreting glands on both the upper and lower jaws, whereas the advanced snakes and anguimorph lizards (including Monitor Lizards, Gila Monster and Beaded Lizard) have more derived venom systems characterized by the loss of the mandibular (lower) or maxillary (upper) glands. Demonstration that the snakes, iguanians and anguimorphs form a single clade provides overwhelming support for a single, early origin of the venom system in lizards and snakes. These results provide new insights into the evolution of the venom system in squamate reptiles and open new avenues for biomedical research and drug design using hitherto unexplored venom proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kochva, E. in Biology of the Reptilia Vol 8 (eds Gans, S. K. & Gans, C.) 43–162 (Academic, London, 1978)
Vidal, N. Colubroid systematics: evidence for an early appearance of the venom apparatus followed by extensive evolutionary tinkering. J. Toxicol. Toxin Rev. 21, 21–41 (2002)
Vidal, N. & Hedges, S. B. Higher-level relationships of caenophidian snakes inferred from four nuclear and mitochondrial genes. C. R. Biol. 325, 987–995 (2002)
Fry, B. G. et al. Isolation of a neurotoxin (alpha-colubritoxin) from a ‘non-venomous’ colubrid: evidence for early origin of venom in snakes. J. Mol. Evol. 57, 446–452 (2003)
Fry, B. G. et al. LC/MS (liquid chromatography, mass spectrometry) analysis of Colubroidea snake venoms: evolutionary and toxinological implications. Rapid Commun. Mass Spectrom. 17, 2047–2062 (2003)
Kochva, E. Development of the venom gland and trigeminal muscles in Vipera palaestinae. Acta Anat. 52, 49–89 (1963)
Kochva, E. The development of the venom gland in the opisthoglyph snake Telescopus fallax with remarks on Thamnophis sirtalis (Colubridae, Reptilia). Copeia 2, 147–154 (1965)
Kochva, E. The origin of snakes and evolution of the venom apparatus. Toxicon 25, 65–106 (1987)
Kochva, E. Atractaspis (Serpentes, Atractaspididae) the Burrowing Asp; a multidisciplinary minireview. Bull. Nat. Hist. Mus. Lond. Zool. 68, 91–99 (2002)
Underwood, G. & Kochva, E. On the affinities of the burrowing asps Atractaspis (Serpentes: Atractaspididae). Zool. J. Linn. Soc. 107, 3–64 (1993)
Underwood, G. in Venomous Snakes: Ecology, Evolution and Snakebite (eds Thorpe, R. S., Wüster, W. & Malhotra, A.) 1–13 (Symp. Zool. Soc. Lond. no. 70, Clarendon, Oxford, 1997)
Jackson, K. The evolution of venom-delivery systems in snakes. Zool. J. Linn. Soc. 137, 337–354 (2003)
Fry, B. G. & Wüster, W. Assembling an arsenal: Origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences. Mol. Biol. Evol. 21, 870–883 (2004)
Vidal, N. & Hedges, S. B. Molecular evidence for a terrestrial origin of snakes. Proc. R. Soc. Lond. B Suppl. 271, S226–S229 (2004)
Townsend, T. M., Larson, A., Louis, E. & Macey, J. R. Molecular phylogenetics of Squamata: The position of snakes, Amphisbaenians, and Dibamids, and the root of the squamate tree. Syst. Biol. 53, 735–757 (2004)
Gabe, M. & Saint Girons, H. Données histologiques sur les glandes salivaires des lépidosauriens. Mém. Mus. Natl Hist. Nat. Paris 58, 1–118 (1969)
Gabe, M. & Saint Girons, H. in Toxins of Animal and Plant Origin (eds de Vries, A. & Kochva, E.) 65–68 (Gordon & Breach, London, 1971)
Gygax, P. Entwicklung, Bau und Funktion der Giftdruse (Duvernoy's gland) von Natrix tessellata. Acta Trop. Zool. 28, 225–274 (1971)
Fry, B. G. From genome to ‘venome’: Molecular origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences and related body proteins. Genome Res. 15, 403–420 (2005)
Fry, B. G. et al. Molecular evolution of elapid snake venom three finger toxins. J. Mol. Evol. 57, 110–129 (2003)
Huang, T. F. & Chiang, H. S. Effect on human platelet aggregation of phospholipase A2 purified from Heloderma horridum (beaded lizard) venom. Biochim. Biophys. Acta 1211, 61–68 (1994)
Fry, B. G. et al. Electrospray liquid chromatography/mass spectrometry fingerprinting of Acanthophis (death adder) venoms: taxonomic and toxinological implications. Rapid Commun. Mass Spectrom. 16, 600–608 (2002)
Fry, B. G. et al. Novel natriuretic peptides from the venom of the inland taipan (Oxyuranus microlepidotus): Isolation, chemical and biological characterization. Biochem. Biophys. Res. Commun. 327, 1011–1015 (2005)
Sopiev, O., Makeev, B. M., Kudryavtsev, S. B. & Makarov, A. N. A case of intoxication by a bite of the gray monitor (Varanus griseus). Izv. Akad. Nauk Turkm. SSR. Ser. Biol. Nauk 87, 78 (1987)
Gorelov, Y. U. K. About the toxicity of the saliva of the gray monitor. Izv. Akad. Nauk Turkm. SSR. Ser. Biol. Nauk 71, 74 (1971)
Pregill, G. K., Gauthier, J. A. & Greene, H. W. The evolution of Helodermatid squamates, with description of a new taxon and an overview of Varanoidea. Trans. San Diego Soc. Nat. Hist. 21, 167–202 (1986)
Norell, M. A., McKenna, M. C. & Novacek, M. J. Estesia mongoliensis, a new fossil varanoid from the Late Cretaceous Barun Goyot Formation of Mongolia. Am. Mus. Novit. 3045, 1–24 (1992)
Evans, S. E. At the feet of the dinosaurs: the origin, evolution and early diversification of squamate reptiles (Lepidosauria: Diapsida). Biol. Rev. Cambr. 78, 513–551 (2003)
Guex, N. & Peitsch, M. C. SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling. Electrophoresis 18, 2714–2723 (1997)
Koradi, R., Billeter, M. & Wuthrich, K. MOLMOL: a program for display and analysis of macromolecular structures. J. Mol. Graph. 14(1M), 51–55 (1996)
Acknowledgements
We thank the following persons and institutions who helped us or contributed tissue samples used in this study: A. Fry, Alice Springs Reptile Centre, Australian Reptile Park, M. A. G. de Bakker, R. L. Bezy, B. Branch, J. Campbell, N. Clemann, C. Clemente, C. Cicero, K. Daoues, A. S. Delmas, B. Demeter, J. Haberfield, A. Hassanin, Healesville Sanctuary, M. Hird, Louisiana State University Museum of Zoology, P. Moler, T. Moncuit, P. Moret, National Museum of Natural History Naturalis Leiden (J. W. Arntzen), T. Pappenfus, J.-C. Rage, C. Skliris, J. Smith, S. Sweet, Ultimate Reptiles (South Australia), University of California Museum of Vertebrate Zoology (Berkeley), J. Walker, R. Waters, J. Weigel and B. Wilson. We also thank A. Webb and T. Purcell for providing HPLC access; N. Williamson for help with preliminary mass spectrometry characterization; E. V. Grishin for help in obtaining the references in Russian; S. Edwards for comments; and T. van Wagner and V. Wexler for artwork. This work was funded by the Service de Systématique moléculaire of the Muséum National d'Histoire Naturelle, Institut de Systématique (N.V.) and by grants from the Australian Academy of Science (B.G.F.), Australian Geographic Society (B.G.F.), Australia & Pacific Science Foundation (B.G.F.), Australian Research Council (B.G.F.), CASS Foundation (B.G.F.), Commonwealth of Australia Department of Health and Aging (B.G.F.), Ian Potter Foundation (B.G.F.), International Human Frontiers Science Program Organisation (B.G.F.), Leiden University (F.J.V., M.K.R.), NASA Astrobiology Institute (S.B.H.), National Science Foundation (S.B.H.) and University of Melbourne (B.G.F.). We thank the relevant wildlife departments for granting the scientific permits for field collection of required specimens.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The sequences of the cDNA clones have been deposited in GenBank (accession numbers DQ139877–DQ139931 and DQ184481), as have the nuclear gene sequences (DQ119594–DQ119641). Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains 13 Supplementary Figures, including the molecular phylogenetic analysis of squamate nuclear genes, the phylogenetic analyses and sequence alignments of the toxin types analysed in this study as well as the liquid chromatography-mass spectrometry analysis of Varanus varius (Lace monitor) venom. (PDF 1238 kb)
Supplementary Methods
This file contains detailed descriptions of materials and methods not already described in the main article. (DOC 52 kb)
Rights and permissions
About this article
Cite this article
Fry, B., Vidal, N., Norman, J. et al. Early evolution of the venom system in lizards and snakes. Nature 439, 584–588 (2006). https://doi.org/10.1038/nature04328
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04328
This article is cited by
-
Non-transgenic, PAMAM co-delivery DNA of interactive proteins NbCRVP and NbCalB endows Nicotiana benthamiana with a stronger antiviral effect to RNA viruses
Journal of Nanobiotechnology (2024)
-
Genomic and transcriptomic analyses support a silk gland origin of spider venom glands
BMC Biology (2023)
-
Monitor Lizard Envenomation Treated with ASV
Indian Journal of Pediatrics (2023)
-
Jellyfish Acromitus flagellatus (Maas) nematocyst venom-mediated biogenic synthesis of gold nanoparticles and its anti-proliferative effects
Aquaculture International (2023)
-
Dynamic genetic differentiation drives the widespread structural and functional convergent evolution of snake venom proteinaceous toxins
BMC Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.