Abstract
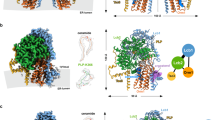
The oxysterol-binding-protein (OSBP)-related proteins (ORPs) are conserved from yeast to humans1,2, and are implicated in the regulation of sterol homeostasis3,4 and in signal transduction pathways5. Here we report the structure of the full-length yeast ORP Osh4 (also known as Kes1) at 1.5–1.9 Å resolution in complexes with ergosterol, cholesterol, and 7-, 20- and 25-hydroxycholesterol. We find that a single sterol molecule binds within a hydrophobic tunnel in a manner consistent with a transport function for ORPs. The entrance is blocked by a flexible amino-terminal lid and surrounded by basic residues that are critical for Osh4 function. The structure of the open state of a lid-truncated form of Osh4 was determined at 2.5 Å resolution. Structural analysis and limited proteolysis show that sterol binding closes the lid and stabilizes a conformation favouring transport across aqueous barriers and signal transmission. The structure of Osh4 in the absence of ligand exposes potential phospholipid-binding sites that are positioned for membrane docking and sterol exchange. On the basis of these observations, we propose a model in which sterol and membrane binding promote reciprocal conformational changes that facilitate a sterol transfer and signalling cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dawson, P. A., Ridgway, N. D., Slaughter, C. A., Brown, M. S. & Goldstein, J. L. cDNA cloning and expression of oxysterol-binding protein, an oligomer with a potential leucine zipper. J. Biol. Chem. 264, 16798–16803 (1989)
Olkkonen, V. M. & Levine, T. P. Oxysterol binding proteins: in more than one place at one time? Biochem. Cell Biol. 82, 87–98 (2004)
Beh, C. T., Cool, L., Phillips, J. & Rine, J. Overlapping functions of the yeast oxysterol-binding protein homologues. Genetics 157, 1117–1140 (2001)
Beh, C. T. & Rine, J. A role for yeast oxysterol-binding protein homologs in endocytosis and in the maintenance of intracellular sterol-lipid distribution. J. Cell Sci. 117, 2983–2996 (2004)
Wang, P., Weng, J. & Anderson, R. G. W. OSBP is a cholesterol-regulated scaffolding protein in control of ERK1/2 activation. Science 307, 1472–1476 (2005)
Kandutsch, A. A., Chen, H. W. & Heiniger, H. J. Biological activity of some oxygenated sterols. Science 201, 498–501 (1978)
Taylor, F. R., Saucier, S. E., Shown, E. P., Parish, E. J. & Kandutsch, A. A. Correlation between oxysterol binding to a cytosolic binding-protein and potency in the repression of hydroxymethylglutaryl coenzyme-A reductase. J. Biol. Chem. 259, 12382–12387 (1984)
Schroepfer, G. J. Jr Oxysterols: Modulators of cholesterol metabolism and other processes. Physiol. Rev. 80, 361–554 (2000)
Levine, T. P. & Munro, S. The pleckstrin homology domain of oxysterol-binding protein recognises a determinant specific to Golgi membranes. Curr. Biol. 8, 729–739 (1998)
Loewen, C. J. R., Roy, A. & Levine, T. P. A conserved ER targeting motif in three families of lipid binding proteins and in Opi1p binds VAP. EMBO J. 22, 2025–2035 (2003)
Ridgway, N. D., Dawson, P. A., Ho, Y. K., Brown, M. S. & Goldstein, J. L. Translocation of oxysterol binding-protein to Golgi-apparatus triggered by ligand-binding. J. Cell Biol. 116, 307–319 (1992)
Xu, Y. Q., Liu, Y. L., Ridgway, N. D. & McMaster, C. R. Novel members of the human oxysterol-binding protein family bind phospholipids and regulate vesicle transport. J. Biol. Chem. 276, 18407–18414 (2001)
Li, X. M. et al. Analysis of oxysterol binding protein homologue Kes1p function in regulation of Sec14p-dependent protein transport from the yeast Golgi complex. J. Cell Biol. 157, 63–77 (2002)
Buchanan, S. K. β-Barrel proteins from bacterial outer membranes: structure, function and refolding. Curr. Opin. Struct. Biol. 9, 455–461 (1999)
Holm, L. & Sander, C. DALI: a network tool for protein-structure comparison. Trends Biochem. Sci. 20, 478–480 (1995)
Tsujishita, Y. & Hurley, J. H. Structure and lipid transport mechanism of a StAR-related domain. Nature Struct. Biol. 7, 408–414 (2000)
Romanowski, M. J., Soccio, R. E., Breslow, J. L. & Burley, S. K. Crystal structure of the Mus musculus cholesterol-regulated START protein 4 (StarD4) containing a StAR-related lipid transfer domain. Proc. Natl Acad. Sci. USA 99, 6949–6954 (2002)
Roderick, S. L. et al. Structure of human phosphatidylcholine transfer protein in complex with its ligand. Nature Struct. Biol. 9, 507–511 (2002)
Tilley, S. J. et al. Structure-function analysis of phosphatidylinositol transfer protein alpha bound to human phosphatidylinositol. Structure 12, 317–326 (2004)
Schouten, A. et al. Structure of apo-phosphatidylinositol transfer protein alpha provides insight into membrane association. EMBO J. 21, 2117–2121 (2002)
Yoder, M. D. et al. Structure of a multifunctional protein. Mammalian phosphatidylinositol transfer protein complexed with phosphatidylcholine. J. Biol. Chem. 276, 9246–9252 (2001)
Sha, B. D., Phillips, S. E., Bankaitis, V. A. & Luo, M. Crystal structure of the Saccharomyces cerevisiae phosphatidylinositol-transfer protein. Nature 391, 506–510 (1998)
Meier, R., Tomizaki, T., Schulze-Briese, C., Baumann, U. & Stocker, A. The molecular basis of vitamin E retention: structure of human alpha-tocopherol transfer protein. J. Mol. Biol. 331, 725–734 (2003)
Min, K. C., Kovall, R. A. & Hendrickson, W. A. Crystal structure of human alpha-tocopherol transfer protein bound to its ligand: implications for ataxia with vitamin E deficiency. Proc. Natl Acad. Sci. USA 100, 14713–14718 (2003)
Wright, C. S., Li, S. C. & Rastinejad, F. Crystal structure of human GM2-activator protein with a novel β-cup topology. J. Mol. Biol. 304, 411–422 (2000)
Friedland, N., Liou, H. L., Lobel, P. & Stock, A. M. Structure of a cholesterol-binding protein deficient in Niemann-Pick type C2 disease. Proc. Natl Acad. Sci. USA 100, 2512–2517 (2003)
Choinowski, T., Hauser, H. & Piontek, K. Structure of sterol carrier protein 2 at 1.8 Å resolution reveals a hydrophobic tunnel suitable for lipid binding. Biochemistry 39, 1897–1902 (2000)
Lopez-Garcia, F. et al. NMR structure of the sterol carrier protein-2: implications for the biological role. J. Mol. Biol. 295, 595–603 (2000)
Malinina, L., Malakhova, M. L., Teplov, A., Brown, R. E. & Patel, D. J. Structural basis for glycosphingolipid transfer specificity. Nature 430, 1048–1053 (2004)
Williams, S. et al. X-ray crystal structure of the liver X receptor β ligand binding domain: regulation by a histidine-tryptophan switch. J. Biol. Chem. 278, 27138–27143 (2003)
Acknowledgements
We thank T. Levine, V. Bankaitis and M. Brown for discussions and sharing unpublished data, N. DeAngelis for technical assistance, R. Craigie for assistance with protein sequencing, J. Kim for advice on the early stages of this project, G. Miller and H. Shi for collecting synchrotron data, C. Beh and R. Scheckman for yeast strains, F. Dyda for maintaining the home X-ray facility, and the staff of beamline X25, National Synchrotron Light Source, Brookhaven National Laboratory and of SER-CAT, Advanced Photon Source, Argonne National Laboratory for assistance with data collection. This research was supported by the intramural program of the NIDDK. Y.J.I. thanks S. H. Eom for mentoring and support. Y.J.I. was partly supported by the Korea Science and Engineering Foundation. Research carried out at the National Synchrotron Light Source is supported by the US Department of Energy, Division of Materials Sciences and Division of Chemical Sciences. Use of the Advanced Photon Source was supported by the US Department of Energy, Basic Energy Sciences, Office of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Coordinates have been deposited with the Protein Data Bank with accession numbers 1ZHT, 1ZHW, 1ZHX, 1ZHY, 1ZHZ and 1ZI7. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
Word file containing Supplementary Methods, additional references, and Supplementary Figure Legends. (DOC 39 kb)
Supplementary Table S1
Statistics of data collection, MIR phasing, and crystallographic refinement. (DOC 40 kb)
Supplementary Figure S1
Experimental electron density. (PDF 453 kb)
Supplementary Figure S2*
Topology of the ORD fold. *Supplementary Fig. 2 of this Letter was substituted on 07/09/05 for an inadvertently duplicated version of Fig. S3. (PDF 132 kb)
Supplementary Figure S3
Comparison to bacterial outer membrane transporters. (PDF 267 kb)
Supplementary Figure S4a
Structure-based alignment of ORDs. (PDF 659 kb)
Supplementary Figure S4b
Structure-based alignment of ORDs. (PDF 388 kb)
Supplementary Figure S5
25-hydroxycholesterol binding to Osh4. (PDF 47 kb)
Supplementary Figure S6
Quantitative immunoblotting. (PDF 71 kb)
Supplementary Figure S7
Sterol-dependent changes in lid conformation. (PDF 73 kb)
Rights and permissions
About this article
Cite this article
Im, Y., Raychaudhuri, S., Prinz, W. et al. Structural mechanism for sterol sensing and transport by OSBP-related proteins. Nature 437, 154–158 (2005). https://doi.org/10.1038/nature03923
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03923
This article is cited by
-
Nanoscale architecture of a VAP-A-OSBP tethering complex at membrane contact sites
Nature Communications (2021)
-
ORP/Osh mediate cross-talk between ER-plasma membrane contact site components and plasma membrane SNAREs
Cellular and Molecular Life Sciences (2021)
-
An electrostatic switching mechanism to control the lipid transfer activity of Osh6p
Nature Communications (2019)
-
Allosteric enhancement of ORP1-mediated cholesterol transport by PI(4,5)P2/PI(3,4)P2
Nature Communications (2019)
-
ER–lysosome contacts enable cholesterol sensing by mTORC1 and drive aberrant growth signalling in Niemann–Pick type C
Nature Cell Biology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.