Abstract
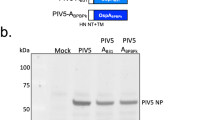
The Lyme disease agent, Borrelia burgdorferi, is maintained in a tick–mouse cycle1,2. Here we show that B. burgdorferi usurps a tick salivary protein, Salp15 (ref. 3), to facilitate the infection of mice. The level of salp15 expression was selectively enhanced by the presence of B. burgdorferi in Ixodes scapularis, first indicating that spirochaetes might use Salp15 during transmission. Salp15 was then shown to adhere to the spirochaete, both in vitro and in vivo, and specifically interacted with B. burgdorferi outer surface protein C. The binding of Salp15 protected B. burgdorferi from antibody-mediated killing in vitro and provided spirochaetes with a marked advantage when they were inoculated into naive mice or animals previously infected with B. burgdorferi. Moreover, RNA interference-mediated repression of salp15 in I. scapularis drastically reduced the capacity of tick-borne spirochaetes to infect mice. These results show the capacity of a pathogen to use a secreted arthropod protein to help it colonize the mammalian host.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Burgdorfer, W. et al. Lyme disease—a tick-borne spirochetosis? Science 216, 1317–1319 (1982)
Ribeiro, J. M., Mather, T. N., Piesman, J. & Spielman, A. Dissemination and salivary delivery of Lyme disease spirochetes in vector ticks (Acari: Ixodidae). J. Med. Entomol. 24, 201–205 (1987)
Anguita, J. et al. Salp15, an Ixodes scapularis salivary protein, inhibits CD4+ T cell activation. Immunity 16, 849–859 (2002)
Steere, A. C., Coburn, J. & Glickstein, L. The emergence of Lyme disease. J. Clin. Invest. 113, 1093–1101 (2004)
Schwan, T. G. & Piesman, J. Temporal changes in outer surface proteins A and C of the Lyme disease-associated spirochete, Borrelia burgdorferi, during the chain of infection in ticks and mice. J. Clin. Microbiol. 38, 382–388 (2000)
Pal, U. et al. OspC facilitates Borrelia burgdorferi invasion of Ixodes scapularis salivary glands. J. Clin. Invest. 113, 220–230 (2004)
Grimm, D. et al. Outer-surface protein C of the Lyme disease spirochete: a protein induced in ticks for infection of mammals. Proc. Natl Acad. Sci. USA 101, 3142–3147 (2004)
Wikel, S. K. Host immunity to ticks. Annu. Rev. Entomol. 41, 1–22 (1996)
Sauer, J. R., McSwain, J. L., Bowman, A. S. & Essenberg, R. C. Tick salivary gland physiology. Annu. Rev. Entomol. 40, 245–267 (1995)
Ribeiro, J. M. & Francischetti, I. M. Role of arthropod saliva in blood feeding: sialome and post-sialome perspectives. Annu. Rev. Entomol. 48, 73–88 (2003)
Das, S. et al. Salp25D, an Ixodes scapularis antioxidant, is 1 of 14 immunodominant antigens in engorged tick salivary glands. J. Infect. Dis. 184, 1056–1064 (2001)
Zeidner, N. S., Schneider, B. S., Nuncio, M. S., Gern, L. & Piesman, J. Coinoculation of Borrelia spp. with tick salivary gland lysate enhances spirochete load in mice and is tick species-specific. J. Parasitol. 88, 1276–1278 (2002)
Steere, A. C. & Glickstein, L. Elucidation of Lyme arthritis. Nature Rev. Immunol. 4, 143–152 (2004)
Wooten, R. M. et al. Toll-like receptor 2 is required for innate, but not acquired, host defense to Borrelia burgdorferi. J. Immunol. 168, 348–355 (2002)
McKisic, M. D. & Barthold, S. W. T-cell-independent responses to Borrelia burgdorferi are critical for protective immunity and resolution of Lyme disease. Infect. Immun. 68, 5190–5197 (2000)
Fikrig, E., Barthold, S. W., Chen, M., Chang, C. H. & Flavell, R. A. Protective antibodies develop, and murine Lyme arthritis regresses, in the absence of MHC class II and CD4 + T cells. J. Immunol. 159, 5682–5686 (1997)
Sadziene, A., Thompson, P. A. & Barbour, A. G. In vitro inhibition of Borrelia burgdorferi growth by antibodies. J. Infect. Dis. 167, 165–172 (1993)
Bunikis, J. et al. Borrelia burgdorferi infection in a natural population of Peromyscus leucopus mice: a longitudinal study in an area where Lyme borreliosis is highly endemic. J. Infect. Dis. 189, 1515–1523 (2004)
Brunet, L. R., Sellitto, C., Spielman, A. & Telford, S. R. Antibody response of the mouse reservoir of Borrelia burgdorferi in nature. Infect. Immun. 63, 3030–3036 (1995)
de Silva, A. M. et al. Immune evasion by tickborne and host-adapted Borrelia burgdorferi. J. Infect. Dis. 177, 395–400 (1998)
Malawista, S. E., Barthold, S. W. & Persing, D. H. Fate of Borrelia burgdorferi DNA in tissues of infected mice after antibiotic treatment. J. Infect. Dis. 170, 1312–1316 (1994)
Coleman, J. L. et al. Plasminogen is required for efficient dissemination of Borrelia burgdorferi in ticks and for enhancement of spirochetemia in mice. Cell 89, 1111–1119 (1997)
Nagamune, K. et al. Surface sialic acids taken from the host allow trypanosome survival in tsetse fly vectors. J. Exp. Med. 199, 1445–1450 (2004)
Nazario, S. et al. Prevention of Borrelia burgdorferi transmission in guinea pigs by tick immunity. Am. J. Trop. Med. Hyg. 58, 780–785 (1998)
Barthold, S. W., Beck, D. S., Hansen, G. M., Terwilliger, G. A. & Moody, K. D. Lyme borreliosis in selected strains and ages of laboratory mice. J. Infect. Dis. 162, 133–138 (1990)
Piesman, J. Standard system for infecting ticks (Acari: Ixodidae) with the Lyme disease spirochete, Borrelia burgdorferi. J. Med. Entomol. 30, 199–203 (1993)
Pal, U. et al. Inhibition of Borrelia burgdorferi-tick interactions in vivo by outer surface protein A antibody. J. Immunol. 166, 7398–7403 (2001)
Narasimhan, S. et al. Disruption of Ixodes scapularis anticoagulation by using RNA interference. Proc. Natl Acad. Sci. USA 101, 1141–1146 (2004)
Acknowledgements
We thank D. Beck for help with the in vivo experiments, K. DePonte and N. Marcantonio for assistance with the microinjection and RNA interference experiments, M. Vasil for the maintenance of ticks, and M. Papero and L. Rollend for guidance with the experiments using Peromyscus leucopus. This work was supported by grants from the American Heart Association, National Institutes of Health, and Centers for Disease Control and Prevention. E.F. is the recipient of a Burroughs Wellcome Clinical Scientist Award in Translational Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Table S1
Peptide sequences obtained through matrix-assisted laser desorption ionization mass spectrometry peptide analysis. (PDF 1263 kb)
Supplementary Table S2
Oligonucleotide primers used for expression studies using RT-PCR and quantitative PCR. (PDF 51 kb)
Supplementary Figure S1
Salp15 injected at a different site from Borrelia burgdorferi did not alter spirochete load in the murine host. (PDF 41 kb)
Supplementary Figure S2
Reduced transmission of B. burgdorferi to Peromyscus leucopus mice by salp15 dsRNA. (PDF 49 kb)
Supplementary Notes
Legends for Supplementary Figures S1 and S2 (PDF 86 kb)
Rights and permissions
About this article
Cite this article
Ramamoorthi, N., Narasimhan, S., Pal, U. et al. The Lyme disease agent exploits a tick protein to infect the mammalian host. Nature 436, 573–577 (2005). https://doi.org/10.1038/nature03812
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03812
This article is cited by
-
Tick saliva and its role in pathogen transmission
Wiener klinische Wochenschrift (2023)
-
The evolving story of Borrelia burgdorferi sensu lato transmission in Europe
Parasitology Research (2022)
-
Prevention of tick-borne diseases: challenge to recent medicine
Biologia (2022)
-
Functional implication of heat shock protein 70/90 and tubulin in cold stress of Dermacentor silvarum
Parasites & Vectors (2021)
-
Impact of tick salivary gland extracts on cytotoxic activity of mouse natural killer cells
Biologia (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.