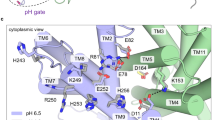
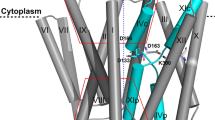
Abstract
The control by Na+/H+ antiporters of sodium/proton concentration and cell volume is crucial for the viability of all cells. Adaptation to high salinity and/or extreme pH in plants and bacteria or in human heart muscles requires the action of Na+/H+ antiporters. Their activity is tightly controlled by pH. Here we present the crystal structure of pH-downregulated NhaA, the main antiporter of Escherichia coli and many enterobacteria. A negatively charged ion funnel opens to the cytoplasm and ends in the middle of the membrane at the putative ion-binding site. There, a unique assembly of two pairs of short helices connected by crossed, extended chains creates a balanced electrostatic environment. We propose that the binding of charged substrates causes an electric imbalance, inducing movements, that permit a rapid alternating-access mechanism. This ion-exchange machinery is regulated by a conformational change elicited by a pH signal perceived at the entry to the cytoplasmic funnel.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
West, I. & Mitchell, P. Proton/sodium ion antiport in Escherichia coli. Biochem. J. 144, 87–90 (1974)
Padan, E., Venturi, M., Gerchman, Y. & Dover, N. Na+/H+ antiporters. Biochim. Biophys. Acta 1505, 144–157 (2001)
Orlowski, J. & Grinstein, S. Diversity of the mammalian sodium/proton exchanger SLC9 gene family. Pflugers Arch. 447, 549–565 (2004)
Counillon, L. & Pouysségur, J. The expanding family of eucaryotic Na+/H+ exchangers. J. Biol. Chem. 275, 1–4 (2000)
Engelhardt, S., Hein, L., Keller, U., Klambt, K. & Lohse, M. J. Inhibition of Na+-H+ exchange prevents hypertrophy, fibrosis, and heart failure in β1-adrenergic receptor transgenic mice. Circ. Res. 90, 814–819 (2002)
Myers, M. L. in The Sodium–Hydrogen Exchange, from Molecule to its Role in Disease (eds Karmazyn, N., Avkiran, M. & Fliegel, L.) 279–290 (Kluwer Academic, Boston, Massachusetts, 2003)
Apse, M. P., Sottosanto, J. B. & Blumwald, E. Vacuolar cation/H+ exchange, ion homeostasis, and leaf development are altered in a T-DNA insertional mutant of AtNHX1, the Arabidopsis vacuolar Na+/H+ antiporter. Plant J. 36, 229–239 (2003)
Qiu, Q. S., Barkla, B. J., Vera-Estrella, R., Zhu, J. K. & Schumaker, K. S. Na+/H+ exchange activity in the plasma membrane of Arabidopsis. Plant Physiol. 132, 1041–1052 (2003)
Apse, M. P., Aharon, G. S., Snedden, W. A. & Blumwald, E. Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport in Arabidopsis. Science 285, 1256–1258 (1999)
Krulwich, T. A., Ito, M. & Guffanti, A. A. The Na+-dependence of alkaliphily in Bacillus. Biochim. Biophys. Acta 1505, 158–168 (2001)
Hase, C. C. & Mekalanos, J. J. Effects of changes in membrane sodium flux on virulence gene expression in Vibrio cholerae. Proc. Natl Acad. Sci. USA 96, 3183–3187 (1999)
Padan, E., Tzubery, T., Herz, K., Kozachkov, L. & Galili, L. NhaA of Escherichia coli, as a model of a pH regulated Na+/H+ antiporter. Biochim. Biophys. Acta 1658, 2–13 (2004)
Taglicht, D., Padan, E. & Schuldiner, S. Overproduction and purification of a functional Na+/H+ antiporter coded by nhaA (ant) from Escherichia coli. J. Biol. Chem. 266, 11289–11294 (1991)
Putney, L. K., Denker, S. P. & Barber, D. L. The changing face of the Na+/H+ exchanger, NHE1: structure, regulation and cellular actions. Annu. Rev. Pharmacol. Toxicol. 42, 527–552 (2002)
Wakabayashi, S., Pang, T., Hisamitsu, T. & Shigekawa, M. in The Sodium–Hydrogen Exchange, from Molecule to its Role in Disease (eds Karmazyn, N., Avkiran, M. & Fliegel, L.) 35–49 (Kluwer Academic, Boston, Massachusetts, 2003)
Gerchman, Y., Rimon, A. & Padan, E. A pH-dependent conformational change of NhaA Na+/H+ antiporter of Escherichia coli involves loop VIII–IX, plays a role in the pH response of the protein, and is maintained by the pure protein in dodecyl maltoside. J. Biol. Chem. 274, 24617–24624 (1999)
Gerchman, Y., Rimon, A., Venturi, M. & Padan, E. Oligomerization of NhaA, the Na+/H+ antiporter of Escherichia coli in the membrane and its functional and structural consequences. Biochemistry 40, 3403–3412 (2001)
Williams, K. A., Geldmacher-Kaufer, U., Padan, E., Schuldiner, S. & Kuhlbrandt, W. Projection structure of NhaA, a secondary transporter from Escherichia coli, at 4.0 Å resolution. EMBO J. 18, 3558–3563 (1999)
Williams, K. A. Three-dimensional structure of the ion-coupled transport protein NhaA. Nature 403, 112–115 (2000)
Rothman, A., Padan, E. & Schuldiner, S. Topological analysis of NhaA, a Na+/H+ antiporter from Escherichia coli. J. Biol. Chem. 271, 32288–32292 (1996)
Dutzler, R., Campbell, E. B., Cadene, M., Chait, B. T. & MacKinnon, R. X-ray structure of a ClC chloride channel at 3.0 Å reveals the molecular basis of anion selectivity. Nature 415, 287–294 (2002)
Palsdottir, H. & Hunte, C. Lipids in membrane protein structures. Biochim. Biophys. Acta 1666, 2–18 (2004)
Fu, D. et al. Structure of a glycerol-conducting channel and the basis for its selectivity. Science 290, 481–486 (2000)
Khademi, S. et al. Mechanism of ammonia transport by Amt/MEP/Rh: structure of AmtB at 1.35 Å. Science 305, 1587–1594 (2004)
von Heijne, G. Membrane protein structure prediction: hydrophobicity analysis and the ‘positive inside’ rule. J. Mol. Biol. 225, 487–494 (1992)
Glusker, J. P. Structural aspects of metal liganding to functional groups in proteins. Adv. Protein Chem. 42, 1–76 (1991)
Harding, M. M. The architecture of metal coordination groups in proteins. Acta Crystallogr. D Biol. Crystallogr. 60, 849–859 (2004)
Inoue, H., Noumi, T., Tsuchiya, T. & Kanazawa, H. Essential aspartic acid residues, Asp-133, Asp-163 and Asp-164, in the transmembrane helices of a Na+/H+ antiporter (NhaA) from Escherichia coli. FEBS Lett. 363, 264–268 (1995)
Galili, L., Rothman, A., Kozachkov, L., Rimon, A. & Padan, E. Trans membrane domain IV is involved in ion transport activity and pH regulation of the NhaA-Na+/H+ antiporter of Escherichia coli. Biochemistry 41, 609–617 (2002)
Rimon, A., Tzubery, T., Galili, L. & Padan, E. Proximity of cytoplasmic and periplasmic loops in NhaA Na+/H+ antiporter of Escherichia coli as determined by site-directed thiol cross-linking. Biochemistry 41, 14897–14905 (2002)
Tzubery, T., Rimon, A. & Padan, E. Mutation E252C increases drastically the Km for Na and causes an alkaline shift of the pH dependence of NhaA Na+/H+ antiporter of Escherichia coli. J. Biol. Chem. 279, 3265–3272 (2004)
Venturi, M. et al. The monoclonal antibody 1F6 identifies a pH-dependent conformational change in the hydrophilic NH2 terminus of NhaA Na+/H+ antiporter of Escherichia coli. J. Biol. Chem. 275, 4734–4742 (2000)
Zilberstein, D., Agmon, V., Schuldiner, S. & Padan, E. The sodium/proton antiporter is part of the pH homeostasis mechanism in Escherichia coli. J. Biol. Chem. 257, 3687–3691 (1982)
Jin, J., Guffanti, A. A., Bechhofer, D. H. & Krulwich, T. A. Tet(L) and tet(K) tetracycline-divalent metal/H+ antiporters: characterization of multiple catalytic modes and a mutagenesis approach to differences in their efflux substrate and coupling ion preferences. J. Bacteriol. 184, 4722–4732 (2002)
Jencks, W. P. The utilization of binding energy in coupled vectorial processes. Adv. Enzymol. 51, 75–106 (1980)
West, I. C. Ligand conduction and the gated-pore mechanism of transmembrane transport. Biochim. Biophys. Acta 1331, 213–234 (1997)
Taglicht, D., Padan, E. & Schuldiner, S. Proton–sodium stoichiometry of NhaA, an electrogenic antiporter from Escherichia coli. J. Biol. Chem. 268, 5382–5387 (1993)
Galili, L., Hertz, K., Dym, O. & Padan, E. Unraveling functional and structural interactions between trans membrane domains IV and XI of NhaA Na+/H+ antiporter of Escherichia coli. J. Biol. Chem. 279, 23104–23113 (2004)
Rimon, A., Gerchman, Y., Kariv, Z. & Padan, E. A point mutation (G338S) and its suppressor mutations affect both the pH response of the NhaA-Na+/H+ antiporter as well as the growth phenotype of Escherichia coli. J. Biol. Chem. 273, 26470–26476 (1998)
Aronson, P. Kinetic properties of the plasma membrane Na+/H+ exchanger. Annu. Rev. Physiol. 47, 545–560 (1985)
Wakabayashi, S., Hisamitsu, T., Pang, T. & Shigekawa, M. Kinetic dissection of two distinct proton binding sites in Na+/H+ exchangers by measurement of reverse mode reaction. J. Biol. Chem. 278, 43580–43585 (2003)
Murakami, S., Nakashima, R., Yamashita, E. & Yamaguchi, A. Crystal structure of bacterial multidrug efflux transporter AcrB. Nature 419, 587–593 (2002)
Abramson, J. et al. Structure and mechanism of the lactose permease of Escherichia coli. Science 301, 610–615 (2003)
Pebay-Peyroula, E. et al. Structure of mitochondrial ADP/ATP carrier in complex with carboxyatractyloside. Nature 426, 39–44 (2003)
Huang, Y., Lemieux, M. J., Song, J., Auer, M. & Wang, D. N. Structure and mechanism of the glycerol-3-phosphate transporter from Escherichia coli. Science 301, 616–620 (2003)
Viitanen, P., Garcia, M. L. & Kaback, H. R. Purified reconstituted lac carrier protein from Escherichia coli is fully functional. Proc. Natl Acad. Sci. USA 81, 1629–1633 (1984)
Klingenberg, M. in The ADP/ATP Carrier in Mitochondrial Membranes (ed. Martonosi, A. N.) (Plenum, New York, 1985)
Auer, M. et al. High-yield expression and functional analysis of Escherichia coli glycerol-3-phosphate transporter. Biochemistry 40, 6628–6635 (2001)
Liang, J., Edelsbrunner, H. & Woodward, C. Anatomy of protein pockets and cavities: measurement of binding site geometry and implications for ligand design. Protein Sci. 7, 1884–1897 (1998)
Kleywegt, G. J. & Jones, T. A. Detection, delineation, measurement and display of cavities in macromolecular structures. Acta Crystallogr. D Biol. Crystallogr. 50, 178–185 (1994)
Acknowledgements
We acknowledge the beamtime and the assistance of the personnel at beamlines ID29 and ID23 at the European Synchrotron Facility and at PX06 at the Swiss Light Source. This study was supported by grants from the German Israeli Foundation for Scientific Research and Development (to H.M. and E.P.), the Max Planck Society, the Fonds der Chemischen Industrie and the Israel Science Foundation (to E.P.). E.S. was supported by the International Max Planck Research School (IMPResS).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Coordinates have been deposited in the Protein Data Bank under accession code 1ZCD. Reprints and permissions information is available at npg.nature.com/reprintsandpermissions. The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Over-expression, purification, crystallization of NhaA; data collection and structure determination. (DOC 40 kb)
Supplementary Table S1
Data collection and refinement statistics for NhaA structure determination. (DOC 33 kb)
Supplementary Figure S1
Organisation of the 12 TMSs of NhaA in two main helical bundles; periplasmic and cytoplasmic view. (PDF 1127 kb)
Supplementary Figure S2
Structural homology between TMS III, IV and V onto TMS X, XI, and XII of NhaA; stereo view of the superimposed elements. (PDF 966 kb)
Rights and permissions
About this article
Cite this article
Hunte, C., Screpanti, E., Venturi, M. et al. Structure of a Na+/H+ antiporter and insights into mechanism of action and regulation by pH. Nature 435, 1197–1202 (2005). https://doi.org/10.1038/nature03692
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03692
This article is cited by
-
Ion and lipid orchestration of secondary active transport
Nature (2024)
-
Architecture and autoinhibitory mechanism of the plasma membrane Na+/H+ antiporter SOS1 in Arabidopsis
Nature Communications (2023)
-
Crystal structure of the Na+/H+ antiporter NhaA at active pH reveals the mechanistic basis for pH sensing
Nature Communications (2022)
-
Structural insights into the HBV receptor and bile acid transporter NTCP
Nature (2022)
-
Structures and mechanism of the plant PIN-FORMED auxin transporter
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.