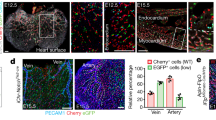
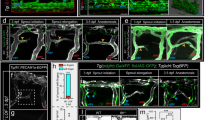
Abstract
Arteries and veins are anatomically, functionally and molecularly distinct. The current model of arterial–venous identity proposes that binding of vascular endothelial growth factor to its heterodimeric receptor—Flk1 and neuropilin 1 (NP-1; also called Nrp1)—activates the Notch signalling pathway in the endothelium, causing induction of ephrin B2 expression and suppression of ephrin receptor B4 expression to establish arterial identity1,2,3,4. Little is known about vein identity except that it involves ephrin receptor B4 expression, because Notch signalling is not activated in veins; an unresolved question is how vein identity is regulated. Here, we show that COUP-TFII (also known as Nr2f2), a member of the orphan nuclear receptor superfamily, is specifically expressed in venous but not arterial endothelium. Ablation of COUP-TFII in endothelial cells enables veins to acquire arterial characteristics, including the expression of arterial markers NP-1 and Notch signalling molecules, and the generation of haematopoietic cell clusters. Furthermore, ectopic expression of COUP-TFII in endothelial cells results in the fusion of veins and arteries in transgenic mouse embryos. Thus, COUP-TFII has a critical role in repressing Notch signalling to maintain vein identity, which suggests that vein identity is under genetic control and is not derived by a default pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lawson, N. D. et al. Notch signaling is required for arterial-venous differentiation during embryonic vascular development. Development 128, 3675–3683 (2001)
Gerety, S. S. & Anderson, D. J. Cardiovascular ephrinB2 function is essential for embryonic angiogenesis. Development 129, 1397–1410 (2002)
Lawson, N. D., Vogel, A. M. & Weinstein, B. M. Sonic hedgehog and vascular endothelium growth factor act upstream of Notch pathway during arterial endothelial differentiation. Dev. Cell 3, 127–136 (2002)
Gu, C. et al. Neuropilin-1 conveys Semaphorin and VEGF signaling during neural and cardiovascular development. Dev. Cell 5, 45–57 (2003)
Pereira, F. A., Qiu, Y. H., Chou, G., Tsai, M.-J. & Tsai, S. Y. The orphan nuclear receptor COUP-TFII is required for angiogenesis and heart development. Genes Dev. 13, 1037–1049 (1999)
Takamoto, N. et al. COUP-TFII is essential for radial and anterior-posterior patterning of the stomach. Development 132, 2179–2182 (2005)
Sato, T. N. et al. Distinct roles of the receptor kinase Tie-1 and Tie-2 in blood vessel formation. Nature 376, 70–74 (1995)
Suri, C. et al. Requisite role of angiopoietin-1, a ligand for TIE2 receptor, during embryonic angiogenesis. Cell 87, 1171–1180 (1996)
Kisanuki, Y. Y. et al. Tie2-Cre transgenic mice: a new model for endothelial cell-lineage analysis in vivo . Dev. Biol. 230, 230–242 (2001)
Huppert, S. S. et al. Embryonic lethality in mice homozygous for a processing-deficient allele of Notch1. Nature 405, 966–970 (2000)
Krebs, L. T. et al. Notch signaling is essential for vascular morphogenesis in mice. Genes Dev. 14, 1343–1352 (2000)
Xue, Y. et al. Embryonic lethality and vascular defects in mice lacking the Notch ligand Jagged1. Hum. Mol. Genet. 8, 723–730 (1999)
Villa, N. et al. Vascular expression of Notch pathway receptors and ligands is restricted to arterial vessels. Mech. Dev. 108, 161–164 (2001)
Wang, H. U., Chen, Z.-F. & Anderson, D. J. Molecular distinction and angiogenic interaction between embryonic arteries and veins revealed by ephrin-B2 and its receptor Eph-B4. Cell 93, 741–753 (1998)
Gerety, S. S., Wang, H. U., Chen, Z.-F. & Anderson, D. J. Symmetrical mutant phenotypes of the receptor EphB4 and its specific transmembrane ligand ephrin-B2 in cardiovascular development. Mol. Cell 4, 403–414 (1999)
Shalaby, F. et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 376, 62–66 (1995)
Kawasaki, T. et al. A requirement for neuropilin-1 in embryonic vessel formation. Development 126, 4895–4902 (1999)
Zhong, T. P., Childs, S., Leu, J. P. & Fishman, M. C. Gridlock signalling pathway fashions the first embryonic artery. Nature 414, 216–220 (2001)
Fischer, A., Schumacher, N., Maier, M., Sendtner, M. & Gessler, M. The Notch target genes Hey1 and Hey2 are required for embryonic vascular development. Genes Dev. 18, 901–911 (2004)
Garcia-Porrero, J. A., Godin, I. E. & Dieterlen-lievre, F. Potential intraembryonic hemogenic sites at pre-liver stages in the mouse. Anat. Embryol. (Berl.) 192, 427–435 (1995)
Tavian, M. et al. Aorta-associated CD34+ hematopoietic cells in the early human embryo. Blood 87, 67–72 (1996)
Medvinsky, A. L., Samoylina, N. L., Muller, A. M. & Dzierzak, E. A. An early pre-liver intraembryonic source of CFU-S in the developing mouse. Nature 364, 64–67 (1993)
Godin, I. E., Garcia-Porrero, J. A., Coutinho, A., Dieterlen-lievre, F. & Macros, M. A. Para-aortic splanchnopleura from early mouse embryos contains B1a cell progenitors. Nature 364, 67–69 (1993)
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003)
Calvi, L. M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003)
Hogan, B. L. M., Beddington, R. S. P., Costantini, E. & Lacy, E. Manipulating the Mouse Embryo, a Laboratory Manual (Cold Spring Harbor Laboratory Press, New Yolk, 1994)
Adams, R. H. Molecular control of arterial-venous blood vessel identity. J. Anat. 202, 105–112 (2003)
Acknowledgements
We thank W. Qian and C. Yang for technical assistance; M. Yanagisawa for providing the Tie2-Cre transgenic mice; Y. Furuta for providing the Efnb2 construct for RNA in situ hybridization; and F. Petit for the COUP-TFII minigene construct. We also thank L.-Y. Yu-Lee and H. J. Bellen for discussions and critical reading of the manuscript. This work was supported by NIH grants to S.Y.T. and M.-J.T.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure S1
Differential expression pattern of COUP-TFII using knockin lacZ reporter as a marker in the vasculature. (PDF 158 kb)
Supplementary Figure S2
Model of cell autonomous versus non-cell autonomous functions. (PDF 50 kb)
Supplementary Figure S3
Endothelial-specific knockout of COUP-TFII by Tie2-Cre. (PDF 125 kb)
Supplementary Figure S4
The endothelial-specific COUP-TFII null mutants exhibited a variety of vascular defects. (PDF 138 kb)
Supplementary Figure S5
The haematopoietic-specific markers in haematopoietic cell clusters of the mutant. (PDF 99 kb)
Supplementary Figure Legends
Legends to accompany the above Supplementary Figures. (DOC 25 kb)
Rights and permissions
About this article
Cite this article
You, LR., Lin, FJ., Lee, C. et al. Suppression of Notch signalling by the COUP-TFII transcription factor regulates vein identity. Nature 435, 98–104 (2005). https://doi.org/10.1038/nature03511
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03511
This article is cited by
-
Apela promotes blood vessel regeneration and remodeling in zebrafish
Scientific Reports (2024)
-
Sinus venosus adaptation models prolonged cardiovascular disease and reveals insights into evolutionary transitions of the vertebrate heart
Nature Communications (2023)
-
Understanding the development, pathogenesis, and injury response of meningeal lymphatic networks through the use of animal models
Cellular and Molecular Life Sciences (2023)
-
An NKX-COUP-TFII morphogenetic code directs mucosal endothelial addressin expression
Nature Communications (2022)
-
Notch signaling pathway: architecture, disease, and therapeutics
Signal Transduction and Targeted Therapy (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.