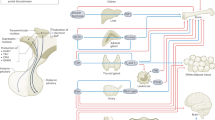
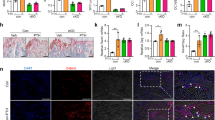
Abstract
Bone remodelling, the mechanism by which vertebrates regulate bone mass, comprises two phases, namely resorption by osteoclasts and formation by osteoblasts; osteoblasts are multifunctional cells also controlling osteoclast differentiation. Sympathetic signalling via β2-adrenergic receptors (Adrb2) present on osteoblasts controls bone formation downstream of leptin1. Here we show, by analysing Adrb2-deficient mice, that the sympathetic nervous system favours bone resorption by increasing expression in osteoblast progenitor cells of the osteoclast differentiation factor Rankl. This sympathetic function requires phosphorylation (by protein kinase A) of ATF4, a cell-specific CREB-related transcription factor essential for osteoblast differentiation and function2. That bone resorption cannot increase in gonadectomized Adrb2-deficient mice highlights the biological importance of this regulation, but also contrasts sharply with the increase in bone resorption characterizing another hypogonadic mouse with low sympathetic tone, the ob/ob mouse3. This discrepancy is explained, in part, by the fact that CART (‘cocaine amphetamine regulated transcript’), a neuropeptide whose expression is controlled by leptin and nearly abolished in ob/ob mice4, inhibits bone resorption by modulating Rankl expression. Our study establishes that leptin-regulated neural pathways control both aspects of bone remodelling, and demonstrates that integrity of sympathetic signalling is necessary for the increase in bone resorption caused by gonadal failure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Takeda, S. et al. Leptin regulates bone formation via the sympathetic nervous system. Cell 111, 305–317 (2002)
Yang, X. et al. ATF4 is a substrate of RSK2 and an essential regulator of osteoblast biology; implication for Coffin-Lowry Syndrome. Cell 117, 387–398 (2004)
Ducy, P. et al. Leptin inhibits bone formation through a hypothalamic relay: A central control of bone mass. Cell 100, 197–207 (2000)
Kristensen, P. et al. Hypothalamic CART is a new anorectic peptide regulated by leptin. Nature 393, 72–76 (1998)
Chruscinski, A. J. et al. Targeted disruption of the beta2 adrenergic receptor gene. J. Biol. Chem. 274, 16694–16700 (1999)
Thomas, S. A., Matsumoto, A. M. & Palmiter, R. D. Noradrenaline is essential for mouse fetal development. Nature 374, 643–646 (1995)
Friedman, J. M. & Halaas, J. L. Leptin and the regulation of body weight in mammals. Nature 395, 763–770 (1998)
Dominici, M. et al. Hematopoietic cells and osteoblasts are derived from a common marrow progenitor after bone marrow transplantation. Proc. Natl Acad. Sci. USA 101, 11761–11766 (2004)
Teitelbaum, S. L. & Ross, F. P. Genetic regulation of osteoclast development and function. Nature Rev. Genet. 4, 638–649 (2003)
Lacey, D. L. et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176 (1998)
Berkowitz, L. A., Riabowol, K. T. & Gilman, M. Z. Multiple sequence elements of a single functional class are required for cyclic AMP responsiveness of the mouse c-fos promoter. Mol. Cell. Biol. 9, 4272–4281 (1989)
Asnicar, M. A. et al. Absence of cocaine- and amphetamine-regulated transcript results in obesity in mice fed a high caloric diet. Endocrinology 142, 4394–4400 (2001)
Satoh, N. et al. Sympathetic activation of leptin via the ventromedial hypothalamus: leptin-induced increase in catecholamine secretion. Diabetes 48, 1787–1793 (1999)
Orwoll, B., Bouxsein, M. L., Marks, D. L., Cone, R. D. & Klein, R. F. in ORS/AAOS Presentations 2003, 71st Annual Meeting of the AAOS (ORS, San Francisco, CA, 2004).
Rohrer, D. K., Chruscinski, A., Schauble, E. H., Bernstein, D. & Kobilka, B. K. Cardiovascular and metabolic alterations in mice lacking both beta1- and beta2-adrenergic receptors. J. Biol. Chem. 274, 16701–16708 (1999)
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997)
Takahashi, N. et al. Osteoblastic cells are involved in osteoclast formation. Endocrinology 123, 2600–2602 (1988)
Ducy, P. & Karsenty, G. Two distinct osteoblast-specific cis-acting elements control expression of a mouse osteocalcin gene. Mol. Cell. Biol. 15, 1858–1869 (1995)
Acknowledgements
We thank T. Townes and A. Hanauer for Atf4 and Rsk2 - / - mice, A. Hanauer and M. Montminy for RSK2 and CREB antibodies, M. Huelskamp and D.A. Horst for Dpd and CTX measurements, P. Ducy for suggestions and critical readings of the manuscript, and L. Li for technical assistance This work was supported by grants from NIH, NSBRI and CNRC (G.K., F.E.), Arthritis Foundation (S.T.) and Children's Brittle Bone Foundation (X.Y.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figure S1-S9. (PDF 378 kb)
Supplementary Figure Legends
This file contains legends to accompany the above Supplementary Figures. (DOC 22 kb)
Rights and permissions
About this article
Cite this article
Elefteriou, F., Ahn, J., Takeda, S. et al. Leptin regulation of bone resorption by the sympathetic nervous system and CART. Nature 434, 514–520 (2005). https://doi.org/10.1038/nature03398
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature03398
This article is cited by
-
Cracking the Code: The Role of Peripheral Nervous System Signaling in Fracture Repair
Current Osteoporosis Reports (2024)
-
The roles of bone remodeling in normal hematopoiesis and age-related hematological malignancies
Bone Research (2023)
-
Neuro–bone tissue engineering: emerging mechanisms, potential strategies, and current challenges
Bone Research (2023)
-
The interaction between the nervous system and the stomatognathic system: from development to diseases
International Journal of Oral Science (2023)
-
Hallmarks of peripheral nerve function in bone regeneration
Bone Research (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.