Abstract
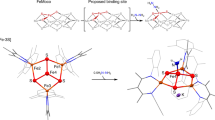
The metal-sulphur active sites of hydrogenases catalyse hydrogen evolution or uptake at rapid rates. Understanding the structure and function of these active sites—through mechanistic studies of hydrogenases1,2,3,4, synthetic assemblies5,6,7,8,9,10,11,12 and in silico models13,14,15—will help guide the design of new materials for hydrogen production or uptake16. Here we report the assembly of the iron-sulphur framework of the active site of iron-only hydrogenase (the H-cluster), and show that it functions as an electrocatalyst for proton reduction. Through linking of a di-iron subsite to a {4Fe4S} cluster, we achieve the first synthesis of a metallosulphur cluster core involved in small-molecule catalysis. In addition to advancing our understanding of the natural biological system, the availability of an active, free-standing analogue of the H-cluster may enable us to develop useful electrocatalytic materials for application in, for example, reversible hydrogen fuel cells. (Platinum is currently the preferred electrocatalyst for such applications, but is expensive, limited in availability and, in the long term, unsustainable17.)
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Peters, J. W., Lanzilotta, W. N., Lemon, B. J. & Seefeldt, L. C. X-ray crystal structure of the Fe-only hydrogenase (Cpl) from Clostridium pasteurianum to 1.8 angstrom resolution. Science 282, 1853–1858 (1998)
Nicolet, Y., Piras, C., Legrand, P., Hatchikian, C. E. & Fontecilla-Camps, J. C. Desulfovibrio desulfuricans iron hydrogenase: the structure shows unusual coordination to an active site Fe binuclear center. Struct. Fold. Des. 7, 13–23 (1999)
Nicolet, Y., Lemon, B. J., Fontecilla-Camps, J. C. & Peters, J. W. A novel FeS cluster in Fe-only hydrogenases. Trends Biochem. Sci. 25, 138–143 (2000)
Armstrong, F. A. Hydrogenases: active site puzzles and progress. Curr. Opin. Chem. Biol. 8, 133–140 (2004)
Evans, D. J. & Pickett, C. J. Chemistry and the hydrogenases. Chem. Soc. Rev. 32, 268–275 (2003)
Lyon, E. J., Georgakaki, I. P., Reibenspies, J. H. & Darensbourg, M. Y. Carbon monoxide and cyanide ligands in a classical organometallic complex model for Fe-only hydrogenase. Angew. Chem. Int. Edn 38, 3178–3180 (1999)
Schmidt, M., Contakes, S. M. & Rauchfuss, T. B. First generation analogues of the binuclear site in the Fe-only hydrogenases: Fe2(µ-SR)2(CO)4(CN)2 2- . J. Am. Chem. Soc. 121, 9736–9737 (1999)
Le Cloirec, A. et al. A di-iron dithiolate possessing structural elements of the carbonyl/cyanide sub-site of the H-centre of Fe-only hydrogenase. Chem. Commun. 2285–2286 (1999)
Razavet, M. et al. Transient FTIR spectroelectrochemical and stopped-flow detection of a mixed valence {Fe(I)-Fe(II)} bridging carbonyl intermediate with structural elements and spectroscopic characteristics of the di-iron sub-site of all-iron hydrogenase. Chem. Commun. 700–701 (2002)
George, S. J., Cui, Z., Razavet, M. & Pickett, C. J. The di-iron subsite of all-iron hydrogenase: Mechanism of cyanation of a synthetic {2Fe3S} – carbonyl assembly. Chem. Eur. J. 8, 4037–4046 (2002)
Gloaguen, F., Lawrence, J. D., Rauchfuss, T. B., Benard, M. & Rohmer, M. M. Bimetallic carbonyl thiolates as functional models for Fe-only hydrogenases. Inorg. Chem. 41, 6573–6582 (2002)
Ott, S., Kritikos, M., Akermark, B., Sun, L. C. & Lomoth, R. A biomimetic pathway for hydrogen evolution from a model of the iron hydrogenase active site. Angew. Chem. Int. Edn 43, 1006–1009 (2004)
Cao, Z. X. & Hall, M. B. Modeling the active sites in metalloenzymes. 3. Density functional calculations on models for Fe-hydrogenase: Structures and vibrational frequencies of the observed redox forms and the reaction mechanism at the diiron active center. J. Am. Chem. Soc. 123, 3734–3742 (2001)
Liu, Z. P. & Hu, P. A density functional theory study on the active center of Fe-only hydrogenase: Characterization and electronic structure of the redox states. J. Am. Chem. Soc. 124, 5175–5182 (2002)
Bruschi, M., Fantucci, P. & De Gioia, L. Density functional theory investigation of the active site of Fe-hydrogenases. Systematic study of the effects of redox state and ligands hardness on structural and electronic properties of complexes related to the [2Fe]H subcluster. Inorg. Chem. 43, 3733–3741 (2004)
Basic Research Needs for the Hydrogen Economy (Report of the Basic Energy Sciences Workshop on Hydrogen Production, Storage, and Use, 13–15 May 2003, Office of Science, US Department of Energy); available at http://www.eere.energy.gov/hydrogenandfuelcells/pdfs/bes_project.pdf.
Platinum and Hydrogen for Fuel Cell Vehicles (UK Department for Transport, September 2003); available at http://www.dft.gov.uk/stellent/groups/dft_roads/documents/page/dft_roads_024056.hcsp.
Reihlen, H., Gruhl, A. & Hessling, G. Über den photochemischen und oxydativen Abbau von Carbonylen. Liebigs Ann. Chem. 472, 268–287 (1929)
Razavet, M. et al. All-iron hydrogenase: synthesis, structure and properties of {2Fe3S}-assemblies related to the di-iron sub-site of the H-cluster. Dalton Trans. 586–595 (2003)
Stack, T. D. P. & Holm, R. H. Subsite-specific functionalization of the [4Fe-4S]2+ analog of iron sulfur protein clusters. J. Am. Chem. Soc. 109, 2546–2547 (1987)
Perdew, J. P. Density-functional approximation for the correlation-energy of the inhomogeneous electron-gas. Phys. Rev. B 33, 8822–8824 (1986)
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic-behavior. Phys. Rev. A. 38, 3098–3100 (1988)
Schafer, A., Huber, C. & Ahlrichs, R. Fully optimized contracted Gaussian-basis sets of triple zeta valence quality for atoms Li to Kr. J. Chem. Phys. 100, 5829–5835 (1994)
Wang, L. S., Ding, C. F., Wang, X. B. & Barlow, S. E. Photodetachment photoelectron spectroscopy of multiply charged anions using electrospray ionization. Rev. Sci. Instrum. 70, 1957–1966 (1999)
Yang, X., Razavet, M., Wang, X. B., Pickett, C. J. & Wang, L. S. Probing the electronic structure of the di-iron subsite of Fe-hydrogenase: A photoelectron spectroscopic study of Fe(I)-Fe(I) model complexes. J. Phys. Chem. A 107, 4612–4618 (2003)
Wang, X. B. et al. Probing the intrinsic electronic structure of the cubane [4Fe4S] cluster: Nature's favorite cluster for electron transfer and storage. J. Am. Chem. Soc. 125, 14072–14081 (2003)
Zhao, X. et al. Catalysis of H2/D2 scrambling and other H/D exchange processes by [Fe]-hydrogenase model complexes. Inorg. Chem. 41, 3917–3928 (2002)
Tard, C., Liu, X., Hughes, D. L. & Pickett, C. J. A novel {FeI-FeII-FeII-FeI} iron thiolate carbonyl assembly which electrocatalyses hydrogen evolution. Chem. Commun. 133–135 (2005)
Popescu, C. V. & Munck, E. Electronic structure of the H cluster in Fe-hydrogenases. J. Am. Chem. Soc. 121, 7877–7884 (1999)
Pickett, C. J. & Ryder, K. S. Bioinorganic reaction centers on electrodes — Modified electrodes possessing amino-acid, peptide and ferredoxin-type groups on a poly(pyrrole) backbone. J. Chem. Soc. Dalton Trans. 2181–2189 (1994)
Acknowledgements
This work was supported by the BBSRC and the John Innes Foundation. We thank D. J. Evans and J. E. Barclay for Mössbauer spectroscopy and discussions; L. Hill for ESMS; and R. R. Eady and R. A. Dixon for discussions and comments on the manuscript. Research done at Washington State University was supported by NIH and performed at EMSL, a national scientific user facility sponsored by the US DOE's Office of Biological and Environmental Research and located at PNNL, which is operated for DOE by Battelle.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Methods
Experimental, ESMS and PES information. (DOC 42 kb)
Supplementary Notes
Note on dissociation step in Supplementary Figure 4. (DOC 29 kb)
Supplementary Figure 1
X-ray structure of B. (JPG 167 kb)
Supplementary Figure 2
PES spectrum of [Fe4S4(SEt)3{Fe2(CH3C(CH2S)3)(CO)5}]2-. (DOC 30 kb)
Supplementary Figure 3
Simulation 10th scan of D. (JPG 25 kb)
Supplementary Figure 4
Proposed mechanism for electrochemistry of D. (GIF 12 kb)
Supplementary Figure Legends
(DOC 26 kb)
Supplementary Data
Crystallographic data of B. (DOC 34 kb)
Rights and permissions
About this article
Cite this article
Tard, C., Liu, X., Ibrahim, S. et al. Synthesis of the H-cluster framework of iron-only hydrogenase. Nature 433, 610–613 (2005). https://doi.org/10.1038/nature03298
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03298
This article is cited by
-
A Tetranuclear Propane-1,3-Dithiolate Complex, [Fe2(CO)5{Ph2P(CH2)3}{(µ-SCH2)2CH2}]2, with a Bridging 1,6-Bis(diphenylphosphino)hexane Ligand
Journal of Chemical Crystallography (2022)
-
A Trimetallic Cu(II) Derivative as an Efficient and Stable Electrocatalyst for Reduction of Proton to Molecular Hydrogen
Catalysis Letters (2020)
-
Graphene Supported Rhodium Nanoparticles for Enhanced Electrocatalytic Hydrogen Evolution Reaction
Scientific Reports (2019)
-
Planar three-coordinate iron sulfide in a synthetic [4Fe-3S] cluster with biomimetic reactivity
Nature Chemistry (2019)
-
Synthesis, structures, and electrochemistry of diiron toluene-3,4-dithiolate complexes containing phosphine ligands
Transition Metal Chemistry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.