Abstract
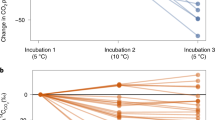
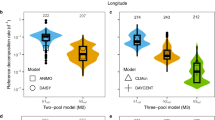
Our understanding of the relationship between the decomposition of soil organic matter (SOM) and soil temperature affects our predictions of the impact of climate change on soil-stored carbon1. One current opinion is that the decomposition of soil labile carbon is sensitive to temperature variation whereas resistant components are insensitive2,3,4. The resistant carbon or organic matter in mineral soil is then assumed to be unresponsive to global warming2,4. But the global pattern and magnitude of the predicted future soil carbon stock will mainly rely on the temperature sensitivity of these resistant carbon pools. To investigate this sensitivity, we have incubated soils under changing temperature. Here we report that SOM decomposition or soil basal respiration rate was significantly affected by changes in SOM components associated with soil depth, sampling method and incubation time. We find, however, that the temperature sensitivity for SOM decomposition was not affected, suggesting that the temperature sensitivity for resistant organic matter pools does not differ significantly from that of labile pools, and that both types of SOM will therefore respond similarly to global warming.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lenton, T. M. & Huntingford, C. Global terrestrial carbon storage and uncertainties in its temperature sensitivity examined with a simple model. Glob. Change Biol. 9, 1333–1352 (2003)
Liski, J., Ilvesniemi, H., Mäkelä, A. & Westman, C. J. CO2 emissions from soil in response to climatic warming are overestimated—The decomposition of old soil organic matter is tolerant of temperature. Ambio 28, 171–174 (1999)
Thornley, J. H. M. & Cannell, M. G. R. Soil carbon storage response to temperature: a hypothesis. Ann. Bot. 87, 591–598 (2001)
Giardina, C. P. & Ryan, M. G. Evidence that decomposition rates of organic carbon in mineral soil do not vary with temperature. Nature 404, 858–861 (2000)
Fang, C. & Moncrieff, J. B. The dependence of soil CO2 efflux on temperature. Soil Biol. Biochem. 33, 155–165 (2001)
Sanderman, J., Amundson, R. G. & Baldocchi, D. D. Application of eddy covariance measurements to the temperature dependence of soil organic matter mean residence time. Glob. Biogeochem. Cycles 17, doi:10.1029/2001GB001833 (2003)
Schlesinger, W. H. & Andrews, J. A. Soil respiration and the global carbon cycle. Biogeochemistry 48, 7–20 (2000)
Jobbágy, E. G. & Jackson, R. B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl. 10, 423–436 (2000)
Rustad, L. E. et al. A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126, 543–562 (2001)
Peterjohn, W. T., Melillo, J. M. & Bowles, S. T. Soil warming and trace gas fluxes: experimental design and preliminary flux results. Oecologia 93, 18–24 (1993)
Peterjohn, W. T. et al. Response of trace gas fluxes and N availability to experimentally elevated soil temperature. Ecol. Appl. 4, 617–625 (1994)
Dalias, P., Anderson, J. M., Bottner, P. & Coûteaux, M.-M. Temperature responses of carbon mineralization in conifer forest soils from different regional climates incubated under standard laboratory conditions. Glob. Change Biol. 6, 181–192 (2001)
Trumbore, S. E., Chadwick, O. A. & Amundson, R. Rapid exchange between soil carbon and atmospheric carbon dioxide driven by temperature change. Science 272, 393–396 (1996)
Winkler, J. P., Cherry, R. S. & Schlesinger, W. H. The Q 10 relationship of microbial respiration in a temperate forest soil. Soil Biol. Biochem. 28, 1067–1072 (1996)
MacDonald, N. W., Zak, D. R. & Pregitzer, K. S. Temperature effects on kinetics of microbial respiration and net nitrogen and sulfur mineralization. Soil Sci. Soc. Am. J. 59, 233–240 (1995)
Ross, D. J. & Tate, K. R. Microbial C and N, and respiratory activity, in litter and soil of a southern beech (Nothofagus) forest: distribution and properties. Soil Biol. Biochem. 25, 477–483 (1994)
Reichstein, M., Bednorz, F., Broll, G. & Kätterer, T. Temperature dependence of carbon mineralisation: conclusions from a long-term incubation of subalpine soil samples. Soil Biol. Biochem. 32, 947–958 (2000)
Fierer, N., Allen, A. S., Schimel, J. P. & Holden, P. A. Controls on microbial CO2 production: a comparison of surface and subsurface soil horizon. Glob. Change Biol. 9, 1322–1332 (2003)
Lovell, R. D. & Jarvis, S. C. Soil microbial biomass and activity in soil from different grassland management treatments stored under controlled conditions. Soil Biol. Biochem. 30, 2077–2085 (1998)
Lomander, A., Kätterer, T. & Andrén, O. Carbon dioxide evolution from top- and subsoil as affected by moisture and constant and fluctuating temperature. Soil Biol. Biochem. 30, 2017–2022 (1998)
Grisi, B., Grace, C., Brookes, P. C., Benedetti, A. & Dell'abate, M. T. Temperature effects on organic matter and microbial biomass dynamics in temperate and tropical soils. Soil Biol. Biochem. 30, 1309–1315 (1998)
IPCC. Special Report on Emissions Scenarios (Cambridge Univ. Press, Cambridge, UK, 2000)
Coleman, K. & Jenkinson, D. S. in Evaluation of Soil Organic Matter Models Using Existing Long-Term Datasets (eds Powlson, D. S., Smith, P. & Smith, J. U.) 237–246 (NATO ASI Series I Vol. 38, Springer, Heidelberg, 1996)
Allen, S. E., Grimshaw, H. M., Parkingson, J. A. & Quarmby, C. Chemical Analysis of Ecological Materials 137–139 (Blackwell Scientific, Oxford, 1974)
Martin-Olmedo, P. & Rees, R. M. Short-term N availability in response to dissolved organic-carbon from poultry manure, alone or in combination with cellulose. Biol. Fert. Soils 29, 386–393 (1999)
Öhlinger, R. in Methods in Soil Biology (eds Schinner, F. et al.) 56–58 (Springer, Berlin, 1995)
Acknowledgements
We thank M. Wattenbarch and C. Zhang for assistance with the modelling. The pan-European modelling used data sets arising from the EU-funded ATEAM project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Methods
This file explains how the measured soil respiration data were edited, the methods we used to estimate Q10 values from respiration rates at different temperatures, and how we assessed the contribution of resistant carbon pool to the total soil organic carbon decomposition and the Q10 value. (DOC 53 kb)
Supplementary Figures S1–3
Supplementary Figure S1 shows the impact of temperature and incubation time on soil basal respiration rate of root-free samples. Supplementary Figure S2 shows contribution of resistant and labile components to soil basal respiration and the relative importance of resistant carbon decompostition with incubation time. Supplementary Figure S3 shows impacts of changing Q10 value for resistant carbon (humus) decomposition on the temperature dependence of soil basal respiration. The figures show the variation in soil respiration rate with time and temperature, contributions of resistant carbon pool to the total respiration rate and the Q10 value. Figures support that the resistant carbon pool is equally sensitive to temperature change to labile pools. (DOC 139 kb)
Rights and permissions
About this article
Cite this article
Fang, C., Smith, P., Moncrieff, J. et al. Similar response of labile and resistant soil organic matter pools to changes in temperature. Nature 433, 57–59 (2005). https://doi.org/10.1038/nature03138
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature03138
This article is cited by
-
Thermal responses of dissolved organic matter under global change
Nature Communications (2024)
-
Drought may exacerbate dryland soil inorganic carbon loss under warming climate conditions
Nature Communications (2024)
-
Comparing the temperature sensitivity of organic matter decomposition in oxic and oxygen-deprived soils
Soil Ecology Letters (2024)
-
Substrate availability reconciles the contrasting temperature response of SOC mineralization in different soil profiles
Journal of Soils and Sediments (2024)
-
Substantial halogenated organic chemicals stored in permafrost soils on the Tibetan Plateau
Nature Geoscience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.