Abstract
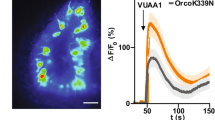
All animals exhibit innate behaviours in response to specific sensory stimuli that are likely to result from the activation of developmentally programmed neural circuits. Here we observe that Drosophila exhibit robust avoidance to odours released by stressed flies. Gas chromatography and mass spectrometry identifies one component of this ‘Drosophila stress odorant (dSO)’ as CO2. CO2 elicits avoidance behaviour, at levels as low as 0.1%. We used two-photon imaging with the Ca2+-sensitive fluorescent protein G-CaMP to map the primary sensory neurons governing avoidance to CO2. CO2 activates only a single glomerulus in the antennal lobe, the V glomerulus; moreover, this glomerulus is not activated by any of 26 other odorants tested. Inhibition of synaptic transmission in sensory neurons that innervate the V glomerulus, using a temperature-sensitive Shibire gene (Shits)1, blocks the avoidance response to CO2. Inhibition of synaptic release in the vast majority of other olfactory receptor neurons has no effect on this behaviour. These data demonstrate that the activation of a single population of sensory neurons innervating one glomerulus is responsible for an innate avoidance behaviour in Drosophila.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kitamoto, T. Conditional modification of behavior in Drosophila by targeted expression of a temperature-sensitive shibire allele in defined neurons. J. Neurobiol. 47, 81–92 (2001)
Dudai, Y., Jan, Y. N., Byers, D., Quinn, W. G. & Benzer, S. dunce, a mutant of Drosophila deficient in learning. Proc. Natl Acad. Sci. USA 73, 1684–1688 (1976)
Stocker, R. F. The organization of the chemosensory system in Drosophila melanogaster: a review. Cell Tissue Res. 275, 3–26 (1994)
Heisenberg, M. Mushroom body memoir: from maps to models. Nature Rev. Neurosci. 4, 266–275 (2003)
Heimbeck, G., Bugnon, V., Gendre, N., Keller, A. & Stocker, R. F. A central neural circuit for experience-independent olfactory and courtship behavior in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 98, 15336–15341 (2001)
Wang, Y. et al. Blockade of neurotransmission in Drosophila mushroom bodies impairs odor attraction, but not repulsion. Curr. Biol. 13, 1900–1904 (2003)
de Belle, J. S. & Heisenberg, M. Associative odor learning in Drosophila abolished by chemical ablation of mushroom bodies. Science 263, 692–695 (1994)
Wang, J. W., Wong, A. M., Flores, J., Vosshall, L. B. & Axel, R. Two-photon calcium imaging reveals an odor-evoked map of activity in the fly brain. Cell 112, 271–282 (2003)
Stange, G. & Stowe, S. Carbon-dioxide sensing structures in terrestrial arthropods. Microsc. Res. Tech. 47, 416–427 (1999)
Stensmyr, M. C., Giordano, E., Balloi, A., Angioy, A. M. & Hansson, B. S. Novel natural ligands for Drosophila olfactory receptor neurones. J. Exp. Biol. 206, 715–724 (2003)
de Bruyne, M., Foster, K. & Carlson, J. R. Odor coding in the Drosophila antenna. Neuron 30, 537–552 (2001)
Kent, K. S., Harrow, I. D., Quartararo, P. & Hildebrand, J. G. An accessory olfactory pathway in Lepidoptera: the labial pit organ and its central projections in Manduca sexta and certain other sphinx moths and silk moths. Cell Tissue Res. 245, 237–245 (1986)
Bogner, F., Boppre, M., Ernst, K. D. & Boeckh, J. CO2 sensitive receptors on labial palps of Rhodogastria moths (Lepidoptera: Arctiidae): physiology, fine structure and central projection. J. Comp. Physiol. A 158, 741–749 (1986)
Distler, P. & Boeckh, J. Central projections of the maxillary and antennal nerves in the mosquito Aedes aegypti. J. Exp. Biol. 200, 1873–1879 (1997)
Anton, S. Central olfactory pathways in mosquitoes and other insects. Ciba Found. Symp. 200, 184–192; discussions 192–196, 226–232 (1996)
Scott, K. et al. A chemosensory gene family encoding candidate gustatory and olfactory receptors in Drosophila. Cell 104, 661–673 (2001)
Stocker, R. F., Heimbeck, G., Gendre, N. & de Belle, J. S. Neuroblast ablation in Drosophila P[GAL4] lines reveals origins of olfactory interneurons. J. Neurobiol. 32, 443–456 (1997)
Shorey, H. H. Behavioral responses to insect pheromones. Annu. Rev. Entomol. 18, 349–380 (1973)
Stange, G. in Advances in Bioclimatology (ed. Stanhill, G.) 223–253 (Springer, Berlin, 1996)
Enserink, M. What mosquitoes want: secrets of host attraction. Science 298, 90–92 (2002)
Galizia, C. G., Sachse, S., Rappert, A. & Menzel, R. The glomerular code for odor representation is species specific in the honeybee Apis mellifera. Nature Neurosci. 2, 473–478 (1999)
Wang, Y. et al. Genetic manipulation of the odor-evoked distributed neural activity in the Drosophila mushroom body. Neuron 29, 267–276 (2001)
Ng, M. et al. Transmission of olfactory information between three populations of neurons in the antennal lobe of the fly. Neuron 36, 463–474 (2002)
Wilson, R. I., Turner, G. C. & Laurent, G. Transformation of olfactory representations in the Drosophila antennal lobe. Science 303, 366–370 (2004)
Laurent, G. Olfactory network dynamics and the coding of multidimensional signals. Nature Rev. Neurosci. 3, 884–895 (2002)
Christensen, T. A., Harrow, I. D., Cuzzocrea, C., Randolph, P. W. & Hildebrand, J. G. Distinct projections of two populations of olfactory receptor axons in the antennal lobe of the sphinx moth Manduca sexta. Chem. Senses 20, 313–323 (1995)
Hansson, B. S., Carlsson, M. A. & Kalinova, B. Olfactory activation patterns in the antennal lobe of the sphinx moth, Manduca sexta. J. Comp. Physiol. A 189, 301–308 (2003)
Troemel, E. R., Kimmel, B. E. & Bargmann, C. I. Reprogramming chemotaxis responses: sensory neurons define olfactory preferences in C. elegans. Cell 91, 161–169 (1997)
Connolly, J. B. et al. Associative learning disrupted by impaired Gs signaling in Drosophila mushroom bodies. Science 274, 2104–2107 (1996)
Beck, C. D., Schroeder, B. & Davis, R. L. Learning performance of normal and mutant Drosophila after repeated conditioning trials with discrete stimuli. J Neurosci 20, 2944–53 (2000)
Acknowledgements
We thank J.-S. Chang for technical assistance, L. Vosshall for providing Or83b-Gal4 and Or47b-Gal4 flies and for other unpublished information, D. Armstrong for Gal4 enhancer trap lines 103Y, 253Y, c747 and c761, T. Kitamoto for UAS-Shits flies, U. Heberlein for the HU protocol and R. I. Wilson for discussion of unpublished data and comments on the manuscript. G.S.B.S. is a recipient of a National Research Service Award. A.C.H. is supported by a Howard Hughes Predoctoral fellowship. This work was supported by the HHMI (R.A. and D.J.A.) and by the NSF (S.B.). R.A. and D.J.A. are Investigators of the Howard Hughes Medical Institute.Author contributions S.B., R.A. and D.J.A. made equally minimal contributions to this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure S1
Dose-response curve for avoidance response to CO2 (a) and dose-response curve for activation of GR21A+ neurons to CO2 (b). (DOC 197 kb)
Rights and permissions
About this article
Cite this article
Suh, G., Wong, A., Hergarden, A. et al. A single population of olfactory sensory neurons mediates an innate avoidance behaviour in Drosophila. Nature 431, 854–859 (2004). https://doi.org/10.1038/nature02980
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature02980
This article is cited by
-
Neural manifolds for odor-driven innate and acquired appetitive preferences
Nature Communications (2023)
-
Threat gates visual aversion via theta activity in Tachykinergic neurons
Nature Communications (2023)
-
Stability of olfactory behavior syndromes in the Drosophila larva
Scientific Reports (2023)
-
The power of Drosophila genetics in studying insect toxicology and chemical ecology
Crop Health (2023)
-
Molecular sensors in the taste system of Drosophila
Genes & Genomics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.