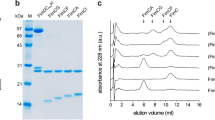
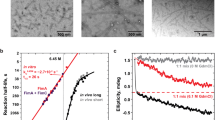
Abstract
Adhesive type 1 pili from uropathogenic Escherichia coli strains have a crucial role during infection by mediating the attachment to and potentially the invasion of host tissue. These filamentous, highly oligomeric protein complexes are assembled by the ‘chaperone–usher’ pathway1, in which the individual pilus subunits fold in the bacterial periplasm and form stoichiometric complexes with a periplasmic chaperone molecule that is essential for pilus assembly2,3,4. The chaperone subsequently delivers the subunits to an assembly platform (usher) in the outer membrane, which mediates subunit assembly and translocation to the cell surface5,6,7,8. Here we show that the periplasmic type 1 pilus chaperone FimC binds non-native pilus subunits and accelerates folding of the subunit FimG by 100-fold. Moreover, we find that the FimC–FimG complex is formed quantitatively and very rapidly when folding of FimG is initiated in the presence of both FimC and the assembly-competent subunit FimF, even though the FimC–FimG complex is thermodynamically less stable than the FimF–FimG complex. FimC thus represents a previously unknown type of protein-folding catalyst, and simultaneously acts as a kinetic trap preventing spontaneous subunit assembly in the periplasm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sauer, F. G. et al. Chaperone-assisted pilus assembly and bacterial attachment. Curr. Opin. Struct. Biol. 10, 548–556 (2000)
Lindberg, F., Tennent, J. M., Hultgren, S. J., Lund, B. & Normark, S. PapD, a periplasmic transport protein in P-pilus biogenesis. J. Bacteriol. 171, 6052–6058 (1989)
Kuehn, M. J., Normark, S. & Hultgren, S. J. Immunoglobulin-like PapD chaperone caps and uncaps interactive surfaces of nascently translocated pilus subunits. Proc. Natl Acad. Sci. USA 88, 10586–10590 (1991)
Jones, C. H. et al. FimC is a periplasmic PapD-like chaperone that directs assembly of type 1 pili in bacteria. Proc. Natl Acad. Sci. USA 90, 8397–8401 (1993)
Klemm, P. & Christiansen, G. The fimD gene required for cell surface localization of Escherichia coli type 1 fimbriae. Mol. Gen. Genet. 220, 334–338 (1990)
Dodson, K. W., Jacob-Dubuisson, F., Striker, R. T. & Hultgren, S. J. Outer-membrane PapC molecular usher discriminately recognizes periplasmic chaperone-pilus subunit complexes. Proc. Natl Acad. Sci. USA 90, 3670–3674 (1993)
Thanassi, D. G. et al. The PapC usher forms an oligomeric channel: implications for pilus biogenesis across the outer membrane. Proc. Natl Acad. Sci. USA 95, 3146–3151 (1998)
Saulino, E. T., Bullitt, E. & Hultgren, S. J. Snapshots of usher-mediated protein secretion and ordered pilus assembly. Proc. Natl Acad. Sci. USA 97, 9240–9245 (2000)
Hahn, E. et al. Exploring the 3D molecular architecture of Escherichia coli type 1 pili. J. Mol. Biol. 323, 845–857 (2002)
Choudhury, D. et al. X-ray structure of the FimC-FimH chaperone-adhesin complex from uropathogenic Escherichia coli. Science 285, 1061–1066 (1999)
Sauer, F. G. et al. Structural basis of chaperone function and pilus biogenesis. Science 285, 1058–1061 (1999)
Sauer, F. G., Pinkner, J. S., Waksman, G. & Hultgren, S. J. Chaperone priming of pilus subunits facilitates a topological transition that drives fiber formation. Cell 111, 543–551 (2002)
Zavialov, A. V. et al. Structure and biogenesis of the capsular F1 antigen from Yersinia pestis: preserved folding energy drives fiber formation. Cell 113, 587–596 (2003)
Zavialov, A. V. et al. Donor strand complementation mechanism in the biogenesis of non-pilus systems. Mol. Microbiol. 45, 983–995 (2002)
Jacob-Dubuisson, F., Striker, R. T. & Hultgren, S. J. Chaperone-assisted self-assembly of pili independent of cellular energy. J. Biol. Chem. 269, 12447–12455 (1994)
Schmid, F. X. Mechanism of folding of ribonuclease A. Slow refolding is a sequential reaction via structural intermediates. Biochemistry 22, 4690–4696 (1983)
Balbach, J. & Schmid, F. X. in Protein Folding: Frontiers in Molecular Biology (ed. Pain, R.) (Oxford Univ. Press, Oxford, 2000)
Pugsley, A. P. The complete general secretory pathway in gram-negative bacteria. Microbiol. Rev. 57, 50–108 (1993)
Vetsch, M., Sebbel, P. & Glockshuber, R. Chaperone-independent folding of type 1 pilus domains. J. Mol. Biol. 322, 827–840 (2002)
Hartl, F. U. & Hayer-Hartl, M. Molecular chaperones in the cytosol: from nascent chain to folded protein. Science 295, 1852–1858 (2002)
Brinker, A. et al. Dual function of protein confinement in chaperonin-assisted protein folding. Cell 107, 223–233 (2001)
Jones, C. H., Danese, P. N., Pinkner, J. S., Silhavy, T. J. & Hultgren, S. J. The chaperone-assisted membrane release and folding pathway is sensed by two signal transduction systems. EMBO J. 16, 6394–6406 (1997)
Eder, J. & Fersht, A. R. Pro-sequence-assisted protein folding. Mol. Microbiol. 16, 609–614 (1995)
Baker, D., Sohl, J. L. & Agard, D. A. A protein-folding reaction under kinetic control. Nature 356, 263–265 (1992)
Frand, A. R., Cuozzo, J. W. & Kaiser, C. A. Pathways for protein disulphide bond formation. Trends Cell Biol. 10, 203–210 (2000)
Schiene-Fischer, C., Habazettl, J., Schmid, F. X. & Fischer, G. The hsp70 chaperone DnaK is a secondary amide peptide bond cis-trans isomerase. Nature Struct. Biol. 9, 419–424 (2002)
Hermanns, U., Sebbel, P., Eggli, V. & Glockshuber, R. Characterization of FimC, a periplasmic assembly factor for biogenesis of type 1 pili in Escherichia coli. Biochemistry 39, 11564–11570 (2000)
Nishiyama, M., Vetsch, M., Puorger, C., Jelesarov, I. & Glockshuber, R. Identification and characterization of the chaperone-subunit complex-binding domain from the type 1 pilus assembly platform FimD. J. Mol. Biol. 330, 513–525 (2003)
Acknowledgements
We thank A. Bachmann and T. Kiefhaber for their assistance and discussions; A. Fritz for technical assistance; and R. Brunisholz for Edman sequencing and MALDI–TOF mass spectrometry. This work was supported by the Schweizerische Nationalfonds and the Swiss NCCR Program in Structural Biology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Table S1
The data in this table are derived from the fluorescence traces of FimGt folding kinetics (Supplementary Fig. S3) in the presence of the chaperone FimCYY, the subunit FimFF and the donor strand peptides DSFimC, DSFimF, and DSFimG. (PDF 98 kb)
Supplementary Figure S1
List of recombinant type1 pilus subunit constructs and synthetic donor strand peptides used in this study. (PDF 84 kb)
Supplementary Figure S2
Spectroscopic properties and thermodynamic stability of FimGt. The data illustrate that the tryptophan fluorescence of FimGt changes upon folding and that FimGt can fold also in the absence of any donor strand. (PDF 91 kb)
Supplementary Figure S3
Stopped flow fluorescence traces of FimGt refolding. Folding of FimGt is slow and neither the partner subunit FimFF nor synthetic donor strand peptides influence FimGt folding significantly. In contrast, the reaction catalysed by the chaperone FimC exhibits very rapid and complex folding kinetics. (PDF 294 kb)
Supplementary Figure S4
The figure shows interrupted refolding experiments that follow the formation of native complexes between FimGt and various donor strand peptides. In addition, the determination of the apparent affinity of FimGt for the corresponding donor strand peptides is shown. (PDF 95 kb)
Supplementary Figure S5
Comparison of FimC and the tryptophan-free variant FimCYY with respect to stability, spectroscopic properties and function. The results indicate that FimCYY retains the ability of FimC to form soluble complexes with subunits in vivo despite its lowered intrinsic stability. (PDF 254 kb)
Supplementary Figure S6
Interrupted refolding was used to monitor the formation of native FimGt in the presence of both FimCYY and cyclophilin. Periplasmic cyclophilin does not abolish the slow phase in chaperone-assisted formation of native FimGt. (PDF 79 kb)
Rights and permissions
About this article
Cite this article
Vetsch, M., Puorger, C., Spirig, T. et al. Pilus chaperones represent a new type of protein-folding catalyst. Nature 431, 329–333 (2004). https://doi.org/10.1038/nature02891
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02891
This article is cited by
-
The assembly platform FimD is required to obtain the most stable quaternary structure of type 1 pili
Nature Communications (2024)
-
Stochastic chain termination in bacterial pilus assembly
Nature Communications (2023)
-
The periplasmic chaperone protein Psg_2795 contributes to the virulence of Pseudomonas savastanoi pv. glycinea: the causal agent of bacterial blight of soybean
Journal of Microbiology (2022)
-
A comprehensive guide to pilus biogenesis in Gram-negative bacteria
Nature Reviews Microbiology (2017)
-
Chaperone–substrate interactions monitored via a robust TEM-1 β-lactamase fragment complementation assay
Biotechnology Letters (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.