Abstract
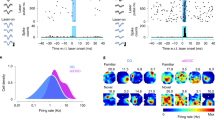
Neural stem cells in various regions of the vertebrate brain continuously generate neurons throughout life1,2,3,4. In the mammalian hippocampus, a region important for spatial and episodic memory5,6, thousands of new granule cells are produced per day7, with the exact number depending on environmental conditions and physical exercise1,8. The survival of these neurons is improved by learning and conversely learning may be promoted by neurogenesis8,9,10. Although it has been suggested that newly generated neurons may have specific properties to facilitate learning2,10,11, the cellular and synaptic mechanisms of plasticity in these neurons are largely unknown. Here we show that young granule cells in the adult hippocampus differ substantially from mature granule cells in both active and passive membrane properties. In young neurons, T-type Ca2+ channels can generate isolated Ca2+ spikes and boost fast Na+ action potentials, contributing to the induction of synaptic plasticity. Associative long-term potentiation can be induced more easily in young neurons than in mature neurons under identical conditions. Thus, newly generated neurons express unique mechanisms to facilitate synaptic plasticity, which may be important for the formation of new memories.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gage, F. H. Mammalian neural stem cells. Science 287, 1433–1438 (2000)
Nottebohm, F. Why are some neurons replaced in adult brain? J. Neurosci. 22, 624–628 (2002)
Seki, T. & Arai, Y. Highly polysialylated neural cell adhesion molecule (NCAM-H) is expressed by newly generated granule cells in the dentate gyrus of the adult rat. J. Neurosci. 13, 2351–2358 (1993)
van Praag, H. et al. Functional neurogenesis in the adult hippocampus. Nature 415, 1030–1034 (2002)
Lisman, J. E. Relating hippocampal circuitry to function: recall of memory sequences by reciprocal dentate-CA3 interactions. Neuron 22, 233–242 (1999)
Burgess, N., Maguire, E. A. & O'Keefe, J. The human hippocampus and spatial and episodic memory. Neuron 35, 625–641 (2002)
Cameron, H. A. & McKay, R. D. G. Adult neurogenesis produces a large pool of new granule cells in the dentate gyrus. J. Comp. Neurol. 435, 406–417 (2001)
van Praag, H., Christie, B. R., Sejnowski, T. J. & Gage, F. H. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc. Natl Acad. Sci. USA 96, 13427–13431 (1999)
Gould, E., Beylin, A., Tanapat, P., Reeves, A. & Shors, T. J. Learning enhances adult neurogenesis in the hippocampal formation. Nature Neurosci. 2, 260–265 (1999)
Shors, T. J. et al. Neurogenesis in the adult is involved in the formation of trace memories. Nature 410, 372–376 (2001)
Snyder, J. S., Kee, N. & Wojtowicz, J. M. Effects of adult neurogenesis on synaptic plasticity in the rat dentate gyrus. J. Neurophysiol. 85, 2423–2431 (2001)
Liu, Y.-B., Lio, P. A., Pasternak, J. F. & Trommer, B. L. Developmental changes in membrane properties and postsynaptic currents of granule cells in rat dentate gyrus. J. Neurophysiol. 76, 1074–1088 (1996)
Seki, T. Hippocampal adult neurogenesis occurs in a microenvironment provided by PSA-NCAM-expressing immature neurons. J. Neurosci. Res. 69, 772–783 (2002)
Seki, T. Expression patterns of immature neuronal markers PSA-NCAM, CRMP-4 and NeuroD in the hippocampus of young adult and aged rodents. J. Neurosci. Res. 70, 327–334 (2002)
Lübbers, K. & Frotscher, M. Differentiation of granule cells in relation to GABAergic neurons in the rat fascia dentata. Combined Golgi/EM and immunocytochemical studies. Anat. Embryol. 178, 119–127 (1988)
Rihn, L. L. & Claiborne, B. J. Dendritic growth and regression in rat dentate granule cells during late postnatal development. Dev. Brain Res. 54, 115–124 (1990)
Rao, M. S. & Shetty, A. K. Efficacy of doublecortin as a marker to analyse the absolute number and dendritic growth of newly generated neurons in the adult dentate gyrus. Eur. J. Neurosci. 19, 234–246 (2004)
Jonas, P., Bischofberger, J., Fricker, D. & Miles, R. Interneuron diversity series: Fast in, fast out—temporal and spatial signal processing in hippocampal interneurons. Trends Neurosci. 27, 30–40 (2004)
Liu, S. et al. Generation of functional inhibitory neurons in the adult rat hippocampus. J. Neurosci. 23, 732–736 (2003)
Spruston, N. & Johnston, D. Perforated patch-clamp analysis of the passive membrane properties of three classes of hippocampal neurons. J. Neurophysiol. 67, 508–529 (1992)
Spruston, N., Jonas, P. & Sakmann, B. Dendritic glutamate receptor channels in rat hippocampal CA3 and CA1 pyramidal neurons. J. Physiol. (Lond.) 482, 325–352 (1995)
Eliot, L. S. & Johnston, D. Multiple components of calcium current in acutely dissociated dentate gyrus granule neurons. J. Neurophysiol. 72, 762–777 (1994)
Blaxter, T. J., Carlen, P. L. & Niesen, C. Pharmacological and anatomical separation of calcium currents in rat dentate granule neurones in vitro. J. Physiol. (Lond.) 412, 93–112 (1989)
Debanne, D., Gähwiler, B. H. & Thompson, S. M. Cooperative interactions in the induction of long-term potentiation and depression of synaptic excitation between hippocampal CA3–CA1 cell pairs in vitro. Proc. Natl Acad. Sci. USA 93, 11225–11230 (1996)
Magee, J. C. & Johnston, D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science 275, 209–213 (1997)
Normann, C. et al. Associative long-term depression in the hippocampus is dependent on postsynaptic N-type Ca2+ channels. J. Neurosci. 20, 8290–8297 (2000)
Jung, M. W. & McNaughton, B. L. Spatial selectivity of unit activity in the hippocampal granular layer. Hippocampus 3, 165–182 (1993)
Skaggs, W. E., McNaughton, B. L., Wilson, M. A. & Barnes, C. A. Theta phase precession in hippocampal neuronal populations and the compression of temporal sequences. Hippocampus 6, 149–172 (1996)
Pike, F. G., Meredith, R. M., Olding, A. W. A. & Paulsen, O. Postsynaptic bursting is essential for ‘Hebbian’ induction of associative long-term potentiation at excitatory synapses in rat hippocampus. J. Physiol. (Lond.) 518, 571–576 (1999)
Dayer, A. G., Ford, A. A., Cleaver, K. M., Yassaee, M. & Cameron, H. A. Short-term and long-term survival of new neurons in the rat dentate gyrus. J. Comp. Neurol. 460, 563–572 (2003)
Acknowledgements
We thank M. Frotscher, E. Förster and G. Stuart for critically reading the manuscript, and K. Winterhalter and A. Blomenkamp for technical assistance. Work was supported by the Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Schmidt-Hieber, C., Jonas, P. & Bischofberger, J. Enhanced synaptic plasticity in newly generated granule cells of the adult hippocampus. Nature 429, 184–187 (2004). https://doi.org/10.1038/nature02553
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature02553
This article is cited by
-
Subfield-specific interneuron circuits govern the hippocampal response to novelty in male mice
Nature Communications (2024)
-
Burst firing is required for induction of Hebbian LTP at lateral perforant path to hippocampal granule cell synapses
Molecular Brain (2023)
-
The times they are a-changin’: a proposal on how brain flexibility goes beyond the obvious to include the concepts of “upward” and “downward” to neuroplasticity
Molecular Psychiatry (2023)
-
Enhanced excitability but mature action potential waveforms at mossy fiber terminals of young, adult-born hippocampal neurons in mice
Communications Biology (2023)
-
Assessments of dentate gyrus function: discoveries and debates
Nature Reviews Neuroscience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.