Abstract
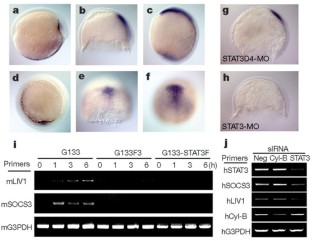
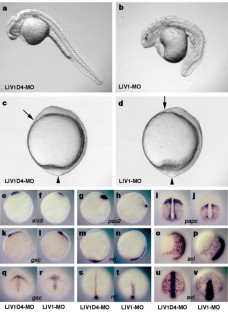
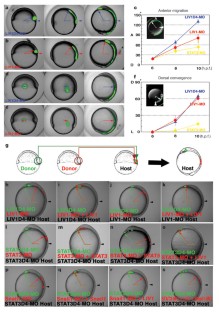
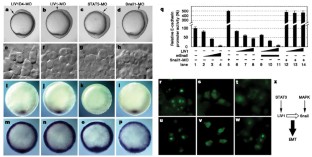
Vertebrate gastrulation is a critical step in the establishment of body plan. During gastrulation, epithelial-mesenchymal transition (EMT) occurs1. EMT is one of the central events of embryonic development, organ and tissue regeneration, and cancer metastasis1,2. Signal transducers and activators of transcription (STATs) mediate biological actions such as cell proliferation, differentiation and survival in response to cytokines and growth factors, in a variety of biological processes3,4,5,6. STATs are also important in EMT during gastrulation, organogenesis, wound healing and cancer progression7,8,9. We previously showed that STAT3 is activated in the organizer during zebrafish gastrulation and its activity is essential for gastrulation movements. The requirement for STAT3 is cell-autonomous for the anterior migration of gastrula organizer cells, and non-cell-autonomous for the convergence of neighbouring cells10. The molecular mechanisms of STAT's action in EMT, however, are unknown. Here we identify LIV1, a breast-cancer-associated zinc transporter protein11,12,13, as a downstream target of STAT3 that is essential and sufficient for STAT3's cell-autonomous role in the EMT of zebrafish gastrula organizer cells. Furthermore, we demonstrate that LIV1 is essential for the nuclear localization of zinc-finger protein Snail, a master regulator of EMT1,2,14,15. These results establish a molecular link between STAT3, LIV1 and Snail in EMT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Thiery, J. P. Epithelial-mesenchymal transitions in tumour progression. Nature Rev. Cancer 2, 442–454 (2002)
Savagner, P. Leaving the neighborhood: molecular mechanisms involved during epithelial-mesenchymal transition. Bioessays 23, 912–923 (2001)
Darnell, J. E. Jr. STATs and gene regulation. Science 277, 1630–1635 (1997)
Bromberg, J. & Darnell, J. E. Jr The role of STATs in transcriptional control and their impact on cellular function. Oncogene 19, 2468–2473 (2000)
Hirano, T., Ishihara, K. & Hibi, M. Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene 19, 2548–2556 (2000)
Kamimura, D. & Hirano, T. in Signal Transducers and Activators of Transcription (STATs): Activation and Biology (eds Sehgal, P. B., Levy, D. E. & Hirano, T.) 155–175 (Kluwer Academic, Dordrecht, 2003)
Sano, S. et al. Keratinocyte-specific ablation of Stat3 exhibits impaired skin remodeling, but does not affect skin morphogenesis. EMBO J. 18, 4657–4668 (1999)
Silver, D. L. & Montell, D. J. Paracrine signaling through the JAK/STAT pathway activates invasive behavior of ovarian epithelial cells in Drosophila. Cell 107, 831–841 (2001)
Yamashita, S. & Hirano, T. in Signal Transducers and Activators of Transcription (STATs): Activation and Biology (eds Sehgal, P. B., Levy, D. E. & Hirano, T.) 595–607 (Kluwer Academic, Dordrecht, 2003)
Yamashita, S. et al. Stat3 controls cell movements during zebrafish gastrulation. Dev. Cell 2, 363–375 (2002)
Manning, D. L., Daly, R. J., Lord, P. G., Kelly, K. F. & Green, C. D. Effects of oestrogen on the expression of a 4.4 kb mRNA in the ZR-75-1 human breast cancer cell line. Mol. Cell. Endocrinol. 59, 205–212 (1988)
Manning, D. L. et al. Oestrogen-regulated genes in breast cancer: association of pLIV1 with lymph node involvement. Eur. J. Cancer 30A, 675–678 (1994)
Taylor, K. M., Morgan, H. E., Johnson, A., Hadley, L. J. & Nicholson, R. I. Structure-function analysis of LIV-1, the breast cancer-associated protein that belongs to a new subfamily of zinc transporters. Biochem. J. 375, 51–59 (2003)
Batlle, E. et al. The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nature Cell Biol. 2, 84–89 (2000)
Cano, A. et al. The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nature Cell Biol. 2, 76–83 (2000)
Taylor, K. M. & Nicholson, R. I. The LZT proteins; the LIV-1 subfamily of zinc transporters. Biochim. Biophys. Acta 1611, 16–30 (2003)
Fukada, T. et al. Two signals are necessary for cell proliferation induced by a cytokine receptor gp130: involvement of STAT3 in anti-apoptosis. Immunity 5, 449–460 (1996)
Nasevicius, A. & Ekker, S. C. Effective targeted gene ‘knockdown’ in zebrafish. Nature Genet. 26, 216–220 (2000)
Kozlowski, D. J. & Weinberg, E. S. Photoactivatable (caged) fluorescein as a cell tracer for fate mapping in the zebrafish embryo. Methods Mol. Biol. 135, 349–355 (2000)
Dominguez, D. et al. Phosphorylation regulates the subcellular location and activity of the snail transcriptional repressor. Mol. Cell. Biol. 23, 5078–5089 (2003)
Van Doren, M. et al. Fear of intimacy encodes a novel transmembrane protein required for gonad morphogenesis in Drosophila. Development 130, 2355–2364 (2003)
Thisse, C., Thisse, B., Schilling, T. F. & Postlethwait, J. H. Structure of the zebrafish snail1 gene and its expression in wild-type, spadetail and no tail mutant embryos. Development 119, 1203–1215 (1993)
Thisse, C., Thisse, B. & Postlethwait, J. H. Expression of snail2, a second member of the zebrafish snail family, in cephalic mesendoderm and presumptive neural crest of wild-type and spadetail mutant embryos. Dev. Biol. 172, 86–99 (1995)
Solnica-Krezel, L., Stemple, D. L. & Driever, W. Transparent things: cell fates and cell movements during early embryogenesis of zebrafish. Bioessays 17, 931–939 (1995)
Blanco, M. J. et al. Correlation of Snail expression with histological grade and lymph node status in breast carcinomas. Oncogene 21, 3241–3246 (2002)
Bowman, T., Garcia, R., Turkson, J. & Jove, R. STATs in oncogenesis. Oncogene 19, 2474–2488 (2000)
Ciruna, B. & Rossant, J. FGF signaling regulates mesoderm cell fate specification and morphogenetic movement at the primitive streak. Dev. Cell 1, 37–49 (2001)
Peinado, H., Quintanilla, M. & Cano, A. Transforming growth factor beta-1 induces snail transcription factor in epithelial cell lines: mechanisms for epithelial mesenchymal transitions. J. Biol. Chem. 278, 21113–21123 (2003)
Acknowledgements
We thank A. G. De Herreros for the pGL3-E-cadh promoter plasmid and pcDNA3-mm snail-HA plasmid, and many colleagues for providing reagents. We also thank R. Masuda and A. Kubota for secretarial assistance. This work was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology in Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1
Primary structure of zebrafish LIV1. (PDF 192 kb)
Supplementary Figure 2
Independence of non-cell-autonomous role of STAT3 in gastrulation movements on LIV1 activity. (PDF 56 kb)
Rights and permissions
About this article
Cite this article
Yamashita, S., Miyagi, C., Fukada, T. et al. Zinc transporter LIVI controls epithelial-mesenchymal transition in zebrafish gastrula organizer. Nature 429, 298–302 (2004). https://doi.org/10.1038/nature02545
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature02545
This article is cited by
-
SLC39A10 promotes malignant phenotypes of gastric cancer cells by activating the CK2-mediated MAPK/ERK and PI3K/AKT pathways
Experimental & Molecular Medicine (2023)
-
Uterine epithelial Gp130 orchestrates hormone response and epithelial remodeling for successful embryo attachment in mice
Scientific Reports (2023)
-
Treatment landscape of triple-negative breast cancer — expanded options, evolving needs
Nature Reviews Clinical Oncology (2022)
-
Triptonide effectively inhibits triple-negative breast cancer metastasis through concurrent degradation of Twist1 and Notch1 oncoproteins
Breast Cancer Research (2021)
-
ADMA mediates gastric cancer cell migration and invasion via Wnt/β-catenin signaling pathway
Clinical and Translational Oncology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.