Abstract
Jepson et al. reply — We did not, as Piantadosi and Thalmann suggest1, present our findings as conclusive evidence of decompression sickness (DCS). We stated neither that DCS occurs naturally in cetaceans, nor that exposure to active sonar increases its occurrence2. However, we restate that there is now a generally accepted link between some beaked-whale strandings and sonar use, and that lesions in some cetaceans demonstrate that in vivo bubble formation (embolus) can occur and persist.
Similar content being viewed by others
Main
Progressively increasing concentrations of nitrogen in cetacean tissues after repetitive diving have been studied empirically in bottlenose dolphins (Tursiops truncatus)3 and higher levels are predicted for northern bottlenose whales (Hyperoodon ampullatus) on the basis of their rate of descent or ascent and depth of diving4. Nitrogen supersaturation could be further increased by an accelerated rate of ascent, possibly to a critical point where bubbles form.
Even if naturally occurring levels of nitrogen supersaturation in the tissues of diving cetaceans are normally insufficient to initiate bubble formation, a theoretical possibility remains that cetaceans with nitrogen-supersaturated tissues could experience bubble growth or formation as a result of intense acoustic exposure4,5. There was a clear spatial and temporal link with active naval sonar exposure in the case of the beaked whales in the Canary Islands, as well as in previously reported beaked-whale strandings 6,7.
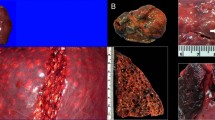
The lesions in the Canary Island beaked whales and in the UK cases (mainly dolphins) differed. The beaked whales had acute, systemic and widely disseminated lesions consistent with, although not diagnostic of, DCS8. The large hepatic cavities found exclusively in the UK cases are atypical of DCS in humans and experimental animals. For logistical reasons, the central nervous system was examined in only two UK cases and the bones were not examined in any. We cannot therefore confirm or refute the presence of lesions consistent with gas embolism in bone or the central nervous system. However, large numbers of gas bubbles were seen in portal veins and sinusoids in the livers from all UK cases examined microscopically, consistent with DCS in humans8.
As cetaceans differ from humans behaviourally (as obligate, repetitive breath-hold divers), physiologically (for example, in their diving reflex)9 and anatomically (as in their retia mirabilia, large portal veins and diaphragmatic sphincters)10,11, it may be too simplistic to assume that the distribution, severity and chronicity of lesions induced by gas emboli will be similar in both human divers and free-living cetaceans. Extensive sublethal bubble formation in human DCS is an acute medical emergency. Without medical intervention, a free-living cetacean suffering the same fate would continue diving for days, weeks or months afterwards unless death or stranding intervened.
Lesion pathogenesis in the stranded cetaceans2 may ultimately be explained by bubble formation, possibly in response to either rapid decompression or acoustic exposure of nitrogen-supersaturated tissues5; however, it is not clear how marine mammals mitigate the accumulation of nitrogen gas while diving and defend themselves against nitrogen-bubble formation. These uncertainties do indeed argue for caution in interpreting the limited studies available.
References
Piantadosi, C. A. & Thalmann, E. D. Nature doi:10.1038/nature02527 (2004).
Jepson, P. D. et al. Nature 425, 575–576 (2003).
Ridgway, S. H. & Howard, R. Science 206, 1182–1183 (1979).
Houser, D. S., Howard, R. & Ridgway, S. H. J. Theor. Biol. 213, 183–195 (2001).
Crum, L. A. & Mao, Y. J. Acoust. Soc. Am. 99, 2898–2907 (1996).
US Department of Commerce and US Navy Joint Interim Report http://www.nmfs.noaa.gov/prot_res/overview/Interim_Bahamas_Report.pdf (2001).
Frantzis, A. Nature 392, 29 (1998).
Francis, T. J. R. & Mitchell, S. J. in Bennett and Elliott's Physiology and Medicine of Diving (eds Brubakk, A. O. & Neuman, T. S.) 5th edn 530–556 (Saunders, Philadelphia, 2003).
Ridgway, S. H. in Mammals of the Sea: Biology and Medicine (ed. Ridgway, S. H.) 590–747 (Thomas, Springfield, Illinois, 1972).
Harrison, R. J. & Tomlinson, D. W. Proc. Zool. Soc. Lond. 126, 205–233 (1956).
Simpson, J. G. & Gardener, M. B. in Mammals of the Sea: Biology and Medicine (ed. Ridgway, S. H.) 298–418 (Thomas, Springfield, Illinois, 1972).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fernández, A., Arbelo, M., Deaville, R. et al. Whales, sonar and decompression sickness (reply). Nature 428, 2 (2004). https://doi.org/10.1038/nature02528a
Issue Date:
DOI: https://doi.org/10.1038/nature02528a
This article is cited by
-
Establishment of a fish model to study gas-bubble lesions
Scientific Reports (2022)
-
Budd-Chiari-like pathology in dolphins
Scientific Reports (2022)
-
Beaked whale auditory evoked potential hearing measurements
Journal of Comparative Physiology A (2006)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.