Abstract
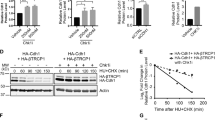
Skp2 and its cofactor Cks1 are the substrate-targeting subunits of the SCFSkp2–Cks1 (Skp1/Cul1/F-box protein) ubiquitin ligase complex that regulates entry into S phase by inducing the degradation of the cyclin-dependent kinase inhibitors p21 and p27 (ref. 1). Skp2 is an oncoprotein that often shows increased expression in human cancers2; however, the mechanism that regulates its cellular abundance is not well understood. Here we show that both Skp2 and Cks1 proteins are unstable in G1 and that their degradation is mediated by the ubiquitin ligase APC/CCdh1 (anaphase-promoting complex/cyclosome and its activator Cdh1). Silencing of Cdh1 by RNA interference in G1 cells stabilizes Skp2 and Cks1, with a consequent increase in p21 and p27 proteolysis. Depletion of Cdh1 also increases the percentage of cells in S phase, whereas concomitant downregulation of Skp2 reverses this effect, showing that Skp2 is an essential target of APC/CCdh1. Expression of a stable Skp2 mutant that cannot bind APC/CCdh1 induces premature entry into S phase. Thus, the induction of Skp2 and Cks1 degradation in G1 represents a principal mechanism by which APC/CCdh1 prevents the unscheduled degradation of SCFSkp2–Cks1 substrates and maintains the G1 state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Reed, S. Ratchets and clocks: the cell cycle, ubiquitylation and protein turnover. Nature Rev. Mol. Cell. Biol. 4, 855–864 (2003)
Pagano, M. & Benmaamar, R. When protein destruction runs amok, malignancy is on the loose. Cancer Cell 4, 251–256 (2003)
Wirbelauer, C. et al. The F-box protein Skp2 is a ubiquitylation target of a Cul1-based core ubiquitin ligase complex: evidence for a role of Cul1 in the suppression of Skp2 expression in quiescent fibroblasts. EMBO J. 19, 5362–5375 (2000)
Peters, J. M. The anaphase-promoting complex: proteolysis in mitosis and beyond. Mol. Cell 9, 931–943 (2002)
Carrano, A. C. & Pagano, M. Role of the F-box protein Skp2 in adhesion-dependent cell cycle progression. J. Cell Biol. 153, 1381–1389 (2001)
Sitry, D. et al. Three different binding sites of Cks1 are required for p27–ubiquitin ligation. J. Biol. Chem. 277, 42233–42240 (2002)
Sudakin, V., Shteinberg, M., Ganoth, D., Hershko, J. & Hershko, A. Binding of activated cyclosome to p13Suc1. Use for affinity purification. J. Biol. Chem. 272, 18051–18059 (1997)
Sutterluty, H. et al. p45Skp2 promotes p27Kip1 degradation and induces S phase in quiescent cells. Nature Cell Biol. 1, 207–214 (1999)
Bloom, J. & Pagano, M. Deregulated degradation of the CDK inhibitor p27 and malignant transformation. Semin. Cancer Biol. 13, 41–47 (2003)
Guardavaccaro, D. et al. Control of meiotic and mitotic progression by the F box protein β-Trcp1 in vivo. Dev. Cell 4, 799–812 (2003)
Fang, G., Yu, H. & Kirschner, M. W. Direct binding of Cdc20 protein family members activates the anaphase-promoting complex in mitosis and G1. Mol. Cell 2, 163–171 (1998)
Bloom, J., Amador, V., Bartolini, F., DeMartino, G. & Pagano, M. Proteasome-mediated degradation of p21 via N-terminal ubiquitinylation. Cell 115, 71–82 (2003)
Acknowledgements
We thank A. Hershko for contributing to this work; W. Kaelin and M. Kirschner for communicating results before publication; J. Bloom, T. Cardozo, L. Gardner, A. Hershko and E. Oxelmark for critically reading the manuscript; and M. Brandeis, B. Dynlacht, G. Draetta, K. Helin and J. Lukas for reagents. M.P. is grateful to T. M. Thor for continuous support. This work was supported by a fellowship from the New York State Breast Cancer Research and Education fund to T.B.; a Federazione Italiana Ricerca sul Cancro fellowship to N.V.D.; a Human Frontiers Science Program fellowship to V.A.; an Italian American Cancer Foundation fellowship and a Susan Komen Breast Cancer Foundation fellowship to D.G.; and a Irma Hirschl Scholarship and NIH grants to M.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Bashir, T., Dorrello, N., Amador, V. et al. Control of the SCFSkp2–Cks1 ubiquitin ligase by the APC/CCdh1 ubiquitin ligase. Nature 428, 190–193 (2004). https://doi.org/10.1038/nature02330
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02330
This article is cited by
-
O-GlcNAcylation of E3 ubiquitin ligase SKP2 promotes hepatocellular carcinoma proliferation
Oncogene (2024)
-
DNA replication initiation factor RECQ4 possesses a role in antagonizing DNA replication initiation
Nature Communications (2023)
-
Regulated interaction of ID2 with the anaphase-promoting complex links progression through mitosis with reactivation of cell-type-specific transcription
Nature Communications (2022)
-
The NUCKS1-SKP2-p21/p27 axis controls S phase entry
Nature Communications (2021)
-
Ubiquitin signaling in cell cycle control and tumorigenesis
Cell Death & Differentiation (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.