Abstract
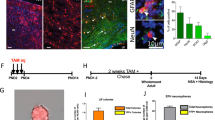
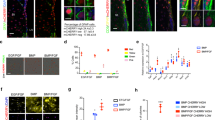
The finding of neurogenesis in the adult brain led to the discovery of adult neural stem cells1. TLX was initially identified as an orphan nuclear receptor expressed in vertebrate forebrains2 and is highly expressed in the adult brain3. The brains of TLX-null mice have been reported to have no obvious defects during embryogenesis4; however, mature mice suffer from retinopathies5, severe limbic defects, aggressiveness, reduced copulation and progressively violent behaviour4,6. Here we show that TLX maintains adult neural stem cells in an undifferentiated, proliferative state. We show that TLX-expressing cells isolated by fluorescence-activated cell sorting (FACS) from adult brains can proliferate, self-renew and differentiate into all neural cell types in vitro. By contrast, TLX-null cells isolated from adult mutant brains fail to proliferate. Reintroducing TLX into FACS-sorted TLX-null cells rescues their ability to proliferate and to self-renew. In vivo, TLX mutant mice show a loss of cell proliferation and reduced labelling of nestin in neurogenic areas in the adult brain. TLX can silence glia-specific expression of the astrocyte marker GFAP in neural stem cells, suggesting that transcriptional repression may be crucial in maintaining the undifferentiated state of these cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gage, F. H. Mammalian neural stem cells. Science 287, 1433–1438 (2000)
Yu, R. T., McKeown, M., Evans, R. M. & Umesono, K. Relationship between Drosophila gap gene tailless and a vertebrate nuclear receptor Tlx. Nature 370, 375–379 (1994)
Monaghan, A. P., Grau, E., Bock, D. & Schutz, G. The mouse homolog of the orphan nuclear receptor tailless is expressed in the developing forebrain. Development 121, 839–853 (1995)
Monaghan, A. P. et al. Defective limbic system in mice lacking the tailless gene. Nature 390, 515–517 (1997)
Yu, R. T. et al. The orphan nuclear receptor Tlx regulates Pax2 and is essential for vision. Proc. Natl Acad. Sci. USA 97, 2621–2625 (2000)
Chiang, M. Y. & Evans, R. M. Reverse Genetic Analysis of Nuclear Receptors, RXRg, RARb, and TLX in Mice (Dissertation, Univ. California, San Diego, 1997)
Gage, F. H. Neurogenesis in the adult brain. J. Neurosci. 22, 612–613 (2002)
Lendahl, U., Zimmerman, L. B. & McKay, R. D. CNS stem cells express a new class of intermediate filament protein. Cell 60, 585–595 (1990)
Reynolds, B. A., Tetzlaff, W. & Weiss, S. A multipotent EGF-responsive striatal embryonic progenitor cell produces neurons and astrocytes. J. Neurosci. 12, 4565–4574 (1992)
Doetsch, F., Petreanu, L., Caille, I., Garcia-Verdugo, J. M. & Alvarez-Buylla, A. EGF converts transit-amplifying neurogenic precursors in the adult brain into multipotent stem cells. Neuron 36, 1021–1034 (2002)
Allen, D. M. et al. Ataxia telangiectasia mutated is essential during adult neurogenesis. Genes Dev. 15, 554–566 (2001)
Taupin, P. et al. FGF-2-responsive neural stem cell proliferation requires CCg, a novel autocrine/paracrine cofactor. Neuron 28, 385–397 (2000)
Nakashima, K. et al. Synergistic signaling in fetal brain by STAT3–Smad1 complex bridged by p300. Science 284, 479–482 (1999)
Lie, D. C. et al. The adult substantia nigra contains progenitor cells with neurogenic potential. J. Neurosci. 22, 6639–6649 (2002)
Palmer, T. D., Markakis, E. A., Willhoite, A. R., Safar, F. & Gage, F. H. Fibroblast growth factor-2 activates a latent neurogenic program in neural stem cells from diverse regions of the adult CNS. J. Neurosci. 19, 8487–8497 (1999)
Shi, Y. et al. Sharp, an inducible cofactor that integrates nuclear receptor repression and activation. Genes Dev. 15, 1140–1151 (2001)
Misawa, K. et al. A method to identify cDNAs based on localization of green fluorescent protein fusion products. Proc. Natl Acad. Sci. USA 97, 3062–3066 (2000)
Morita, S., Kojima, T. & Kitamura, T. Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther. 7, 1063–1066 (2000)
Miyoshi, H., Blomer, U., Takahashi, M., Gage, F. H. & Verma, I. M. Development of a self-inactivating lentivirus vector. J. Virol. 72, 8150–8157 (1998)
Acknowledgements
We thank G. Cabrera, S. Tiep, M. Nelson, H. Juguilon, J. Havstad, B. Miller, R. Summers, A. Consiglio, A. Huynh, L. Moore, A. Dearie and H. Lansford for technical help; M. L. Gage for editing; E. Stevens and E. Ong for administrative assistance; C. Zhang for comments on the manuscript; R. Marr and I. Verma for the lentiviral vector; and T. Kitamura for the pMY vector and PlatE cells. Y.S. is a fellow of the Susan G. Komen Breast Cancer Foundation. D.C.L. was supported by the Deutsche Forschungsgemeinschaft. P.T. was supported by the Pasarow Foundation. K.N. was supported by a Japan Society for the Promotion of Science (JSPS) Postdoctoral Fellowship for Research Abroad. R.M.E. is an Investigator of the Howard Hughes Medical Institute at the Salk Institute and March of Dimes Chair in Molecular and Developmental Biology. F.H.G. is the Adler Professor of Age-Related Neurodegenerative Diseases. This work was supported by the Howard Hughes Medical Institute, the NIH, the Christopher Reeve Paralysis Foundation, the National Institutes of Aging, the Michael J. Fox Foundation, Project ALS and the Lookout Fund.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
41586_2004_BFnature02211_MOESM2_ESM.jpg
Supplementary Figure 2: Quantitation of nestin+ (a) and GFAP+ cells (b) from TLX+/- and TLX-/- culture in growth media. (JPG 41 kb)
41586_2004_BFnature02211_MOESM4_ESM.jpg
Supplementary Figure 4: Quantitation of Ki67+ (a) and GFAP+ cells (b) in TLX-/- cells infected with control or TLX-GFP lentivirus. (JPG 42 kb)
Rights and permissions
About this article
Cite this article
Shi, Y., Chichung Lie, D., Taupin, P. et al. Expression and function of orphan nuclear receptor TLX in adult neural stem cells. Nature 427, 78–83 (2004). https://doi.org/10.1038/nature02211
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02211
This article is cited by
-
Post-transcriptional control of a stemness signature by RNA-binding protein MEX3A regulates murine adult neurogenesis
Nature Communications (2023)
-
Targeting PUS7 suppresses tRNA pseudouridylation and glioblastoma tumorigenesis
Nature Cancer (2021)
-
Combined Atoh1 and Neurod1 Deletion Reveals Autonomous Growth of Auditory Nerve Fibers
Molecular Neurobiology (2020)
-
Tet3 regulates cellular identity and DNA methylation in neural progenitor cells
Cellular and Molecular Life Sciences (2020)
-
The role of adult hippocampal neurogenesis in brain health and disease
Molecular Psychiatry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.