Abstract
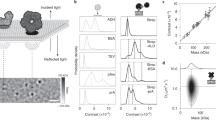
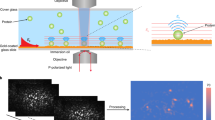
The molecular architecture of—and biochemical processes within—cell membranes play important roles in all living organisms, with many drugs and infectious disease agents targeting membranes. Experimental studies of biochemical reactions on membrane surfaces are challenging, as they require a membrane environment that is fluid (like cell membranes) but nevertheless allows for the efficient detection and characterization of molecular interactions. One approach uses lipid membranes supported on solid substrates such as silica or polymers1,2: although the membrane is trapped near the solid interface, it retains natural fluidity and biological functionality3 and can be implanted with membrane proteins for functional studies4. But the detection of molecular interactions involving membrane-bound species generally requires elaborate techniques, such as surface plasmon resonance 5 or total internal reflection fluorescence microscopy6. Here we demonstrate that colloidal phase transitions of membrane-coated silica beads provide a simple and label-free method for monitoring molecular interactions on lipid membrane surfaces. By adjusting the lipid membrane composition and hence the pair interaction potential between the membrane-supporting silica beads, we poise our system near a phase transition so that small perturbations on the membrane surface induce dramatic changes in the macroscopic organization of the colloid. We expect that this approach, used here to probe with high sensitivity protein binding events at membrane surfaces, can be applied to study a broad range of cell membrane processes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sackmann, E. Supported membranes: Scientific and practical applications. Science 271, 43–48 (1996)
Groves, J. T. Membrane array technology for drug discovery. Curr. Opin. Drug Discov. Dev. 5, 606–612 (2002)
Grakoui, A. et al. The immunological synapse: A molecular machine controlling T cell activation. Science 285, 221–227 (1999)
Fang, Y., Frutos, A. G. & Lahiri, J. Membrane protein microarrays. J. Am. Chem. Soc. 124, 2394–2395 (2002)
Hoffman, T. L., Canziani, G., Jia, L., Rucker, J. & Doms, R. W. A biosensor assay for studying ligand-membrane receptor interactions: binding of antibodies and HIV-1 Env to chemokine receptors. Proc. Natl Acad. Sci. USA 97, 11215–11220 (2000)
Yang, T., Jung, S.-y., Mao, H. & Cremer, P. S. Fabrication of phospholipid bilayer-coated microchannels for on-chip immunoassays. Anal. Chem. 73, 165–169 (2001)
Anderson, V. J. & Lekkerkerker, H. N. W. Insights into phase transition kinetics from colloid science. Nature 416, 811–815 (2002)
Yethiraj, A. & van Blaaderen, A. A colloidal model system with an interaction tunable from hard sphere to soft and dipolar. Nature 421, 513–517 (2003)
Ramos, L., Lubensky, T. C., Dan, N., Nelson, P. & Weitz, D. A. Surfactant-mediated two-dimensional crystallization of colloidal crystals. Science 286, 2325–2328 (1999)
Dinsmore, A. D. et al. Colloidosomes: selectively permeable capsules composed of colloidal particles. Science 298, 1006–1009 (2002)
Manoharan, V. N., Elsesser, M. T. & Pine, D. J. Dense packing and symmetry in small clusters of microspheres. Science 301, 483–487 (2003)
Yang, P. et al. Hierarchically ordered oxides. Science 282, 2244–2246 (1998)
Bayerl, T. M. & Bloom, M. Physical properties of single phospholipid bilayers adsorbed to micro glass beads. Biophys. J. 58, 357–362 (1990)
Buranda, T. et al. Biomimetic molecular assemblies on glass and mesoporous silica microbeads for biotechnology. Langmuir 19, 1654–1663 (2003)
Wong, A. P. & Groves, J. T. Molecular topography imaging by intermembrane fluorescence resonance energy transfer. Proc. Natl Acad. Sci. USA 99, 14147–14152 (2002)
Discher, D. E. & Eisenberg, A. Polymer vesicles. Science 297, 967–973 (2002)
Acknowledgements
We thank R. Parthasarathy and N. Clack for discussions, and D. Discher for advice on the manuscript. M.J. was supported by a Fulbright scholarship and by the Czech Republic Ministry of Education. This work was supported in part by the Burroughs Wellcome Career Award in the Biomedical Sciences (to J.T.G.) and by the US Department of Energy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Baksh, M., Jaros, M. & Groves, J. Detection of molecular interactions at membrane surfaces through colloid phase transitions. Nature 427, 139–141 (2004). https://doi.org/10.1038/nature02209
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature02209
This article is cited by
-
A charge-dependent long-ranged force drives tailored assembly of matter in solution
Nature Nanotechnology (2024)
-
Two-dimensional binary colloidal crystals formed by particles with two different sizes
Scientific Reports (2022)
-
Protein–Protein Interactions on Membrane Surfaces Analysed Using Pull-Downs with Supported Bilayers on Silica Beads
The Journal of Membrane Biology (2022)
-
Membrane fusion studied by colloidal probes
European Biophysics Journal (2021)
-
New Hexagonally Ordered Monolayer Electrode with Monodisperse Carbon/Fe3O4 Microspheres for High Performance Lithium Ion Battery Anodes
Macromolecular Research (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.