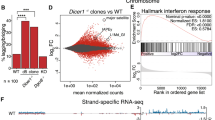
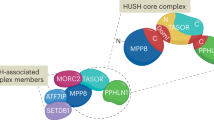
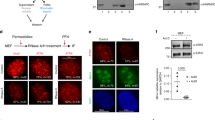
Abstract
Gene repression is crucial to the maintenance of differentiated cell types in multicellular organisms, whereas aberrant silencing can lead to disease. The organization of DNA into chromatin and heterochromatin1 is implicated in gene silencing. In chromatin, DNA wraps around histones, creating nucleosomes. Further condensation of chromatin, associated with large blocks of repetitive DNA sequences, is known as heterochromatin. Position effect variegation (PEV) occurs when a gene is located abnormally close to heterochromatin, silencing the affected gene in a proportion of cells1. Here we show that the relatively short triplet-repeat expansions found in myotonic dystrophy and Friedreich's ataxia confer variegation of expression on a linked transgene in mice. Silencing was correlated with a decrease in promoter accessibility and was enhanced by the classical PEV modifier heterochromatin protein 1 (HP1). Notably, triplet-repeat-associated variegation was not restricted to classical heterochromatic regions but occurred irrespective of chromosomal location. Because the phenomenon described here shares important features with PEV, the mechanisms underlying heterochromatin-mediated silencing might have a role in gene regulation at many sites throughout the mammalian genome and modulate the extent of gene silencing and hence severity in several triplet-repeat diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dillon, N. & Festenstein, R. Unravelling heterochromatin: competition between positive and negative factors regulates accessibility. Trends Genet. 18, 252–258 (2002)
Lachner, M., O'Carroll, D., Rea, S., Mechtler, K. & Jenuwein, T. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 410, 116–120 (2001)
Bannister, A. J. et al. Selective recognition of methylated lysine 9 on histone H3 by the HP1 chromo domain. Nature 410, 120–124 (2001)
Dorer, D. R. & Henikoff, S. Expansions of transgene repeats cause heterochromatin formation and gene silencing in Drosophila. Cell 77, 993–1002 (1994)
Garrick, D., Fiering, S., Martin, D. I. & Whitelaw, E. Repeat-induced gene silencing in mammals. Nature Genet. 18, 56–59 (1998)
Grosveld, F., van Assendelft, G. B., Greaves, D. R. & Kollias, G. Position-independent, high-level expression of the human beta-globin gene in transgenic mice. Cell 51, 975–985 (1987)
Festenstein, R. et al. Locus control region function and heterochromatin-induced position effect variegation. Science 271, 1123–1125 (1996)
Milot, E. et al. Heterochromatin effects on the frequency and duration of LCR-mediated gene transcription. Cell 87, 105–114 (1996)
Kioussis, D. & Festenstein, R. Locus control regions: overcoming heterochromatin-induced gene inactivation in mammals. Curr. Opin. Genet. Dev. 7, 614–619 (1997)
Campuzano, V. et al. Friedreich's ataxia: Autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science 271, 1423–1427 (1996)
Brook, J. D. et al. Molecular basis of myotonic dystrophy: Expansion of a trinucleotide (CTG) repeat at the 3′ end of a transcript encoding a protein kinase family member. Cell 68, 799–808 (1992)
Mahadevan, M. et al. Myotonic dystrophy mutation: an unstable CTG repeat in the 3′ untranslated region of the gene. Science 255, 1253–1255 (1992)
Sun, F. L., Cuaycong, M. H. & Elgin, S. C. Long-range nucleosome ordering is associated with gene silencing in Drosophila melanogaster pericentric heterochromatin. Mol. Cell. Biol. 21, 2867–2879 (2001)
Wang, Y., Amirhaeri, S., Kang, S., Wells, R. D. & Griffith, J. D. Preferential nucleosome assembly at DNA triplet repeats from the myotonic dystrophy gene. Science 265, 669–671 (1994)
Klesert, T. R., Otten, A. D., Bird, T. D. & Tapscott, S. J. Trinucleotide repeat expansion at the myotonic dystrophy locus reduces expression of DMAHP. Nature Genet. 16, 402–406 (1997)
Klesert, T. R. et al. Mice deficient in Six5 develop cataracts: Implications for myotonic dystrophy. Nature Genet. 25, 105–109 (2000)
Carango, P., Noble, J. E., Marks, H. G. & Funanage, V. L. Absence of myotonic dystrophy protein kinase (DMPK) mRNA as a result of a triplet repeat expansion in myotonic dystrophy. Genomics 18, 340–348 (1993)
Krahe, R. et al. Effect of myotonic dystrophy trinucleotide repeat expansion on DMPK transcription and processing. Genomics 28, 1–14 (1995)
Davis, B. M., McCurrach, M. E., Taneja, K. L., Singer, R. H. & Housman, D. E. Expansion of a CUG trinucleotide repeat in the 3′ untranslated region of myotonic dystrophy protein kinase transcripts results in nuclear retention of transcripts. Proc. Natl Acad. Sci. USA 94, 7388–7393 (1997)
Mankodi, A. et al. Myotonic dystrophy in transgenic mice expressing an expanded CUG repeat. Science 289, 1769–1773 (2000)
Wells, R. D., Collier, D. A., Hanvey, J. C., Shimizu, M. & Wohlrab, F. The chemistry and biology of unusual DNA structures adopted by oligopurine.oligopyrimidine sequences. FASEB J. 2, 2939–2949 (1988)
Sakamoto, N., Ohshima, K., Montermini, L., Pandolfo, M. & Wells, R. D. Sticky DNA, a self-associated complex formed at long GAA.TTC repeats in intron 1 of the Frataxin gene, inhibits transcription. J. Biol. Chem. 276, 27171–27177 (2001)
Greaves, D. R., Wilson, F., Lang, G. & Kioussis, D. Human CD2 3′-flanking sequences confer high-level T-cell specific position independent gene expression in transgenic mice. Cell 56, 979–986 (1989)
Festenstein, R. et al. Heterochromatin protein 1 modifies mammalian PEV in a dose- and chromosomal-context-dependent manner. Nature Genet. 23, 457–461 (1999)
Lake, R. A., Wotton, D. & Owen, M. J. A 3′ transcriptional enhancer regulates tissue-specific expression of the human CD2 gene. EMBO J. 9, 3129–3136 (1990)
Carter, D., Chakalova, L., Osborne, C. S., Dai, Y. F. & Fraser, P. Long-range chromatin regulatory interactions in vivo. Nature Genet. 32, 623–626 (2002)
Nielsen, S. J. et al. Rb targets histone H3 methylation and HP1 to promoters. Nature 412, 561–565 (2001)
Litt, M. D., Simpson, M., Gaszner, M., Allis, C. D. & Felsenfeld, G. Correlation between histone lysine methylation and developmental changes at the chicken beta-globin locus. Science 293, 2453–2455 (2001)
Hwang, K. K., Eissenberg, J. C. & Worman, H. J. Transcriptional repression of euchromatic genes by Drosophila heterochromatin protein 1 and histone modifiers. Proc. Natl Acad. Sci. USA 98, 11423–11427 (2001)
Wijgerde, M., Grosveld, F. & Fraser, P. Transcription complex stability and chromatin dynamics in vivo. Nature 377, 209–213 (1995)
Acknowledgements
We thank P. Fraser and especially C. Osborne (Babraham Insitute) for help with the RNA FISH; S. Sharghi Namini, S. Uribe Lewis and D. Kazazi for technical assistance; M. Walport, N. Dillon, L. Aragorn and D. Kioussis for critical reading of an earlier version of the manuscript; and M. McCarthy for advice on statistical analysis. A.S. was a Wellcome Travelling Fellow and C.E. a Wellcome Clinical Fellow. This work was supported by the Muscular Dystrophy Campaign (UK), the MRC UK and a EU Framework 5 grant—GeneXtra.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Saveliev, A., Everett, C., Sharpe, T. et al. DNA triplet repeats mediate heterochromatin-protein-1-sensitive variegated gene silencing. Nature 422, 909–913 (2003). https://doi.org/10.1038/nature01596
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01596
This article is cited by
-
Patient-derived iPSC models of Friedreich ataxia: a new frontier for understanding disease mechanisms and therapeutic application
Translational Neurodegeneration (2023)
-
A wearable motion capture suit and machine learning predict disease progression in Friedreich’s ataxia
Nature Medicine (2023)
-
Genome-wide SSR markers in bottle gourd: development, characterization, utilization in assessment of genetic diversity of National Genebank of India and synteny with other related cucurbits
Journal of Applied Genetics (2022)
-
Molecular Mechanisms and Therapeutics for the GAA·TTC Expansion Disease Friedreich Ataxia
Neurotherapeutics (2019)
-
Protocol of a randomized, double-blind, placebo-controlled, parallel-group, multicentre study of the efficacy and safety of nicotinamide in patients with Friedreich ataxia (NICOFA)
Neurological Research and Practice (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.