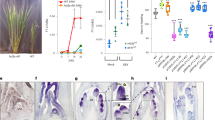
Abstract
The photoperiodic control of flowering is one of the important developmental processes of plants because it is directly related to successful reproduction1. Although the molecular genetic analysis of Arabidopsis thaliana, a long-day (LD) plant, has provided models to explain the control of flowering time in this species2,3,4, very little is known about its molecular mechanisms for short-day (SD) plants. Here we show how the photoperiodic control of flowering is regulated in rice, a SD plant. Overexpression of OsGI5, an orthologue of the Arabidopsis GIGANTEA (GI) gene6,7 in transgenic rice, caused late flowering under both SD and LD conditions. Expression of the rice orthologue8 of the Arabidopsis CONSTANS (CO) gene9 was increased in the transgenic rice, whereas expression of the rice orthologue10 of FLOWERING LOCUS T (FT)11,12 was suppressed. Our results indicate that three key regulatory genes for the photoperiodic control of flowering are conserved between Arabidopsis, a LD plant, and rice, a SD plant, but regulation of the FT gene by CO was reversed, resulting in the suppression of flowering in rice under LD conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Thomas, B. & Vince-Prue, D. Photoperiodism in Plants (Academic, London, 1997)
Koornneef, M., Alonso-Blanco, C., Peeters, A. J. M. & Soppe, W. Genetic control of flowering time in Arabidopsis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 49, 345–370 (1998)
Mouradov, A., Cremer, F. & Coupland, G. Control of flowering time: Interacting pathways as a basis for diversity. Plant Cell Suppl. 14, S111–S130 (2002)
Simpson, G. G. & Dean, C. Arabidopsis, the Rosetta stone of flowering time? Science 296, 285–289 (2002)
Hayama, R., Izawa, T. & Shimamoto, K. Isolation of rice genes possibly involved in the photoperiodic control of flowering by a fluorescent differential display method. Plant Cell Physiol. 43, 494–504 (2002)
Fowler, S. et al. GIGANTEA: A circadian clock-controlled gene that regulates photoperiodic flowering in Arabidopsis and encodes a protein with several possible membrane-spanning domains. EMBO J. 18, 4679–4688 (1999)
Park, D. H. et al. Control of circadian rhythms and photoperiodic flowering by the Arabidopsis GIGANTEA gene. Science 285, 1579–1582 (1999)
Yano, M. et al. Hd1, a major photoperiod sensitivity quantitative trait locus in rice, is closely related to the Arabidopsis flowering time gene CONSTANS. Plant Cell 12, 2473–2484 (2001)
Putterill, J., Robson, F., Lee, K., Simon, R. & Coupland, G. The CONSTANS gene of Arabidopsis promotes flowering and encodes a protein showing similarities to zinc finger transcription factors. Cell 80, 847–857 (1995)
Kojima, S. et al. Hd3a, a rice ortholog of the Arabidopsis FT gene, promotes transition to flowering downstream of Hd1 under short-day conditions. Plant Cell Physiol. 43, 1096–1105 (2002)
Kardailsky, I. et al. Activation tagging of the floral inducer FT. Science 286, 1962–1965 (1999)
Kobayashi, Y., Kaya, H., Goto, K., Iwabuchi, M. & Araki, T. A pair of related genes with antagonistic roles in mediating flowering signals. Science 286, 1960–1962 (1999)
Izawa, T., Oikawa, T., Tokutomi, S., Okuno, K. & Shimamoto, K. Phytochromes confer the photoperiodic control of flowering in rice (a short-day plant). Plant J. 22, 391–399 (2000)
Araki, T. & Komeda, Y. Analysis of the role of the late-flowering locus, GI, in the flowering of Arabidopsis thaliana. Plant J. 3, 231–239 (1993)
Samach, A. et al. Distinct roles of CONSTANS target genes in the reproductive development of Arabidopsis. Science 288, 1613–1616 (2000)
Suarez-Lopez, P. et al. CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature 410, 1116–1120 (2001)
Yanovsky, M. J. & Kay, S. Molecular basis of seasonal time measurement in Arabidopsis. Nature 419, 308–312 (2002)
Izawa, T. et al. Phytochrome mediates the external light signal to repress FT orthologs in photoperiodic flowering of rice. Genes Dev. 16, 2006–2020 (2002)
Martinez-Garcia, J. F., Virgos-Soler, A. & Prat, S. Control of photoperiod-regulated tuberization in potato by the Arabidopsis flowering-time gene CONSTANS. Proc. Natl Acad. Sci. USA 99, 15211–15216 (2002)
Liu, J., Yu, J., McIntosh, L., Kende, H. & Zeevaart, J. A. D. Isolation of a CONSTANS ortholog from Pharbitis nil and its role in flowering. Plant Physiol. 125, 1821–1830 (2001)
Christensen, A. H., Sharrock, R. A. & Quail, P. H. Maize polyubiquitin genes: Structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol. Biol. 18, 675–689 (1992)
Hiei, Y., Ohta, S., Komari, T. & Kumashiro, T. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J. 6, 271–282 (1994)
Tanaka, A. et al. Enhancement of foreign gene expression by a dicot intron in rice but not tobacco is correlated with an increased level of mRNA and an efficient splicing of the intron. Nucleic Acids Res. 18, 6767–6770 (1990)
Acknowledgements
We thank Takeshi Izawa for help in the early part of this work, and members of the Plant Molecular Genetics Lab at NAIST for discussions. This research was supported by Grants-in-Aid for Scientific Research on Priority Areas of the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Hayama, R., Yokoi, S., Tamaki, S. et al. Adaptation of photoperiodic control pathways produces short-day flowering in rice. Nature 422, 719–722 (2003). https://doi.org/10.1038/nature01549
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01549
This article is cited by
-
Transcriptomic analysis reveals the molecular basis of photoperiod-regulated sex differentiation in tropical pumpkins (Cucurbita moschata Duch.)
BMC Plant Biology (2024)
-
QTL epistasis plays a role of homeostasis on heading date in rice
Scientific Reports (2024)
-
Rice PIFs: Critical regulators in rice development and stress response
Plant Molecular Biology (2024)
-
Genetic variation analysis of pleiotropic gene Ghd7 in rice
Plant Growth Regulation (2023)
-
Genetic and signaling pathways of flowering regulation in rice (Oryza sativa L.)
Brazilian Journal of Botany (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.