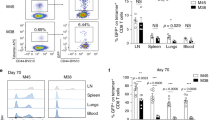
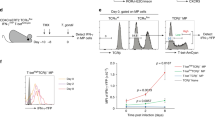
Abstract
A long-standing paradox in cellular immunology concerns the conditional requirement for CD4+ T-helper (TH) cells in the priming of cytotoxic CD8+ T lymphocyte (CTL) responses in vivo. Whereas CTL responses against certain viruses can be primed in the absence of CD4+ T cells, others, such as those mediated through ‘cross-priming’ by host antigen-presenting cells, are dependent on TH cells1,2,3,4. A clearer understanding of the contribution of TH cells to CTL development has been hampered by the fact that most TH-independent responses have been demonstrated ex vivo as primary cytotoxic effectors, whereas TH-dependent responses generally require secondary in vitro re-stimulation for their detection. Here, we have monitored the primary and secondary responses of TH-dependent and TH-independent CTLs and find in both cases that CD4+ T cells are dispensable for primary expansion of CD8+ T cells and their differentiation into cytotoxic effectors. However, secondary CTL expansion (that is, a secondary response upon re-encounter with antigen) is wholly dependent on the presence of TH cells during, but not after, priming. Our results demonstrate that T-cell help is ‘programmed’ into CD8+ T cells during priming, conferring on these cells a hallmark of immune response memory: the capacity for functional expansion on re-encounter with antigen.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Buller, R. M., Holmes, K. L., Hugin, A., Frederickson, T. N. & Morse, H. C. III Induction of cytotoxic T-cell responses in vivo in the absence of CD4 helper cells. Nature 328, 77–79 (1987)
Rahemtulla, A. et al. Normal development and function of CD8+ cells but markedly decreased helper cell activity in mice lacking CD4. Nature 353, 180–184 (1991)
Schoenberger, S. P., Toes, R. E., van der Voort, E. I., Offringa, R. & Melief, C. J. T-cell help for cytotoxic T lymphocytes is mediated by CD40–CD40L interactions. Nature 393, 480–483 (1998)
Bennett, S. R., Carbone, F. R., Karamalis, F., Miller, J. F. & Heath, W. R. Induction of a CD8 + cytotoxic T lymphocyte response by cross-priming requires cognate CD4 + T cell help. J. Exp. Med. 186, 65–70 (1997)
Sprent, J. & Surh, C. D. T cell memory. Annu. Rev. Immunol. 20, 551–579 (2002)
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996)
van Stipdonk, M. J., Lemmens, E. E. & Schoenberger, S. P. Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nature Immunol. 2, 423–429 (2001)
Kaech, S. M. & Ahmed, R. Memory CD8 + T cell differentiation: initial antigen encounter triggers a developmental program in naive cells. Nature Immunol. 2, 415–422 (2001)
Badovinac, V. P., Porter, B. B. & Harty, J. T. Programmed contraction of CD8(+ ) T cells after infection. Nature Immunol. 3, 619–626 (2002)
Keene, J. & Forman, J. Helper activity is required for the in vivo generation of cytotoxic T lymphocytes. J. Exp. Med. 155, 768–782 (1982)
Bennett, S. R. et al. Help for cytotoxic-T-cell responses is mediated by CD40 signalling. Nature 393, 478–480 (1998)
Ridge, J. P., Di Rosa, F. & Matzinger, P. A conditioned dendritic cell can be a temporal bridge between a CD4+ T-helper and a T-killer cell. Nature 393, 474–478 (1998)
Matzinger, P. Tolerance, danger, and the extended family. Annu. Rev. Immunol. 12, 991–1045 (1994)
Lanzavecchia, A. Immunology. Licence to kill. Nature 393, 413–414 (1998)
Schoenberger, S. P. et al. Cross-priming of CTL responses in vivo does not require antigenic peptides in the endoplasmic reticulum of immunizing cells. J. Immunol. 161, 3808–3812 (1998)
Grusby, M. J., Johnson, R. S., Papaioannou, V. E. & Glimcher, L. H. Depletion of CD4 + T cells in major histocompatibility complex class II-deficient mice. Science 253, 1417–1420 (1991)
Oehen, S. & Brduscha-Riem, K. Differentiation of naive CTL to effector and memory CTL: correlation of effector function with phenotype and cell division. J. Immunol. 161, 5338–5346 (1998)
Hudrisier, D., Oldstone, M. B. & Gairin, J. E. The signal sequence of lymphocytic choriomeningitis virus contains an immunodominant cytotoxic T cell epitope that is restricted by both H-2D(b) and H-2K(b) molecules. Virology 234, 62–73 (1997)
Busch, D. H., Pilip, I. M., Vijh, S. & Pamer, E. G. Coordinate regulation of complex T cell populations responding to bacterial infection. Immunity 8, 353–362 (1998)
Parker, D. C. T cell-dependent B cell activation. Annu. Rev. Immunol. 11, 331–360 (1993)
Mintern, J. D., Davey, G. M., Belz, G. T., Carbone, F. R. & Heath, W. R. Cutting edge: precursor frequency affects the helper dependence of cytotoxic T cells. J. Immunol. 168, 977–980 (2002)
Di Rosa, F. & Matzinger, P. Long-lasting CD8 T cell memory in the absence of CD4 T cells or B cells. J. Exp. Med. 183, 2153–2163 (1996)
Klenerman, P., Hengartner, H. & Zinkernagel, R. M. A non-retroviral RNA virus persists in DNA form. Nature 390, 298–301 (1997)
Kaech, S. M., Wherry, E. J. & Ahmed, R. Effector and memory T-cell differentiation: implications for vaccine development. Nature Rev. Immunol. 2, 251–262 (2002)
Suresh, M. et al. Role of CD28-B7 interactions in generation and maintenance of CD8 T cell memory. J. Immunol. 167, 5565–5573 (2001)
Dialynas, D. P. et al. Characterization of the murine T cell surface molecule, designated L3T4, identified by monoclonal antibody GK1.5: similarity of L3T4 to the human Leu-3/T4 molecule. J. Immunol. 131, 2445–2451 (1983)
Matzinger, P. The JAM test. A simple assay for DNA fragmentation and cell death. J. Immunol. Methods 145, 185–192 (1991)
Acknowledgements
This work was supported by grants from the National Institutes of Health, the American Cancer Society, and CaP CURE to S.P.S. The authors thank G. Palioungas for technical assistance and the staff of the LIAI animal facility for animal care.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Janssen, E., Lemmens, E., Wolfe, T. et al. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 421, 852–856 (2003). https://doi.org/10.1038/nature01441
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature01441
This article is cited by
-
CD40 ligand stimulation affects the number and memory phenotypes of human peripheral CD8+ T cells
BMC Immunology (2023)
-
Novel nanoadjuvants balance immune activation with modest inflammation: implications for older adult vaccines
Immunity & Ageing (2023)
-
Immune synapse formation promotes lipid peroxidation and MHC-I upregulation in licensed dendritic cells for efficient priming of CD8+ T cells
Nature Communications (2023)
-
Optimizing the manufacturing and antitumour response of CAR T therapy
Nature Reviews Bioengineering (2023)
-
Potent anti-tumor immune response and tumor growth inhibition induced by HER2 subdomain fusion protein in a mouse tumor model
Journal of Cancer Research and Clinical Oncology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.