Abstract
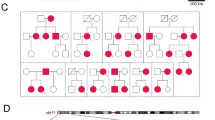
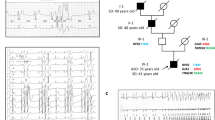
Mutations in ion channels involved in the generation and termination of action potentials constitute a family of molecular defects that underlie fatal cardiac arrhythmias in inherited long-QT syndrome1. We report here that a loss-of-function (E1425G) mutation in ankyrin-B (also known as ankyrin 2), a member of a family of versatile membrane adapters2, causes dominantly inherited type 4 long-QT cardiac arrhythmia in humans. Mice heterozygous for a null mutation in ankyrin-B are haploinsufficient and display arrhythmia similar to humans. Mutation of ankyrin-B results in disruption in the cellular organization of the sodium pump, the sodium/calcium exchanger, and inositol-1,4,5-trisphosphate receptors (all ankyrin-B-binding proteins), which reduces the targeting of these proteins to the transverse tubules as well as reducing overall protein level. Ankyrin-B mutation also leads to altered Ca2+ signalling in adult cardiomyocytes that results in extrasystoles, and provides a rationale for the arrhythmia. Thus, we identify a new mechanism for cardiac arrhythmia due to abnormal coordination of multiple functionally related ion channels and transporters.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others

References
Keating, M. T. & Sanguinetti, M. C. Molecular and cellular mechanisms of cardiac arrhythmias. Cell 104, 569–580 (2001)
Mohler, P. J., Gramolini, A. O. & Bennett, V. Ankyrins. J. Cell Sci. 115, 1565–1566 (2002)
Schott, J. J. et al. Mapping of a gene for long QT syndrome to chromosome 4q25-27. Am. J. Hum. Genet. 57, 1114–1122 (1995)
Tuvia, S., Buhusi, M., Davis, L., Reedy, M. & Bennett, V. Ankyrin-B is required for intracellular sorting of structurally diverse Ca2+ homeostasis proteins. J. Cell Biol. 147, 995–1008 (1999)
Mohler, P. J., Gramolini, A. O. & Bennett, V. The Ankyrin-B C-terminal domain determines activity of Ankyrin-B/G chimeras in rescue of abnormal inositol 1,4,5-trisphosphate and ryanodine receptor distribution in ankyrin-B (-/-) neonatal cardiomyocytes. J. Biol. Chem. 277, 10599–10607 (2002)
Towbin, J. A. & Vatta, M. Molecular biology and the prolonged QT syndromes. Am. J. Med. 110, 385–398 (2001)
Casimiro, M. C. et al. Targeted disruption of the Kcnq1 gene produces a mouse model of Jervell and Lange-Nielsen Syndrome. Proc. Natl Acad. Sci. USA 98, 2526–2531 (2001)
Jenkins, S. M. & Bennett, V. Ankyrin-G coordinates assembly of the spectrin-based membrane skeleton, voltage-gated sodium channels, and L1 CAMs at Purkinje neuron initial segments. J. Cell Biol. 155, 739–746 (2001)
Bennett, V. & Baines, A. J. Spectrin and ankyrin-based pathways: metazoan inventions for integrating cells into tissues. Physiol. Rev. 81, 1353–1392 (2001)
Frank, J. S. & Garfinkel, A. in The Myocardium (ed. Langer, G. A.) 1–32 (Academic, San Diego, California, 1997)
Bers, D. M. Excitation-Contraction Coupling and Cardiac Contractile Force (Kluwer, Dordrecht, 2001)
Marks, A. R., Priori, S., Memmi, M., Kontula, K. & Laitinen, P. J. Involvement of the cardiac ryanodine receptor/calcium release channel in catecholaminergic polymorphic ventricular tachycardia. J. Cell Physiol. 190, 1–6 (2002)
Blaustein, M. P. & Lederer, W. J. Sodium/calcium exchange: its physiological implications. Physiol. Rev. 79, 763–854 (1999)
Reuter, H. et al. The Na+-Ca2+ exchanger is essential for the action of cardiac glycosides. Circ. Res. 90, 305–308 (2002)
Philipson, K. D. & Nicoll, D. A. Sodium-calcium exchange: a molecular perspective. Annu. Rev. Physiol. 62, 111–133 (2000)
Santana, L. F., Kranias, E. G. & Lederer, W. J. Calcium sparks and excitation-contraction coupling in phospholamban-deficient mouse ventricular myocytes. J. Physiol. 503, 21–29 (1997)
Santana, L. F., Gomez, A. M. & Lederer, W. J. Ca2+ flux through promiscuous cardiac Na+ channels: slip-mode conductance. Science 279, 1027–1033 (1998)
Gomez, A. M. et al. Defective excitation-contraction coupling in experimental cardiac hypertrophy and heart failure. Science 276, 800–806 (1997)
duBell, W. H., Lederer, W. J. & Rogers, T. B. Dynamic modulation of excitation-contraction coupling by protein phosphatases in rat ventricular myocytes. J. Physiol. 493, 793–800 (1996)
Acknowledgements
We thank the type 4 long-QT family for participation. Research support was provided by the Howard Hughes Medical Institute, NIH, the Muscular Dystrophy Association, Canadian Institutes of Health, the Institut National de la Santé et de la Recherche Médicale (INSERM), and the programme Hospitalier de Recherche Clinique. We also thank C. Kontos and B. Knollman for discussions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Mohler, P., Schott, JJ., Gramolini, A. et al. Ankyrin-B mutation causes type 4 long-QT cardiac arrhythmia and sudden cardiac death. Nature 421, 634–639 (2003). https://doi.org/10.1038/nature01335
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01335
This article is cited by
-
Isolated Cardiac Ryanodine Receptor Function Varies Between Mammals
The Journal of Membrane Biology (2024)
-
Mouse models of spontaneous atrial fibrillation
Mammalian Genome (2023)
-
Role of Ca2+ in healthy and pathologic cardiac function: from normal excitation–contraction coupling to mutations that cause inherited arrhythmia
Archives of Toxicology (2023)
-
New drug discovery of cardiac anti-arrhythmic drugs: insights in animal models
Scientific Reports (2023)
-
“Ryanopathies” and RyR2 dysfunctions: can we further decipher them using in vitro human disease models?
Cell Death & Disease (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.