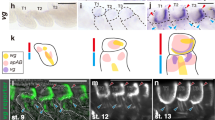
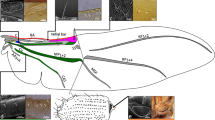
Abstract
The evolution of wings was the central adaptation allowing insects to escape predators, exploit scattered resources, and disperse into new niches, resulting in radiations into vast numbers of species1. Despite the presumed evolutionary advantages associated with full-sized wings (macroptery), nearly all pterygote (winged) orders have many partially winged (brachypterous) or wingless (apterous) lineages, and some entire orders are secondarily wingless (for example, fleas, lice, grylloblattids and mantophasmatids), with about 5% of extant pterygote species being flightless2,3. Thousands of independent transitions from a winged form to winglessness have occurred during the course of insect evolution; however, an evolutionary reversal from a flightless to a volant form has never been demonstrated clearly for any pterygote lineage. Such a reversal is considered highly unlikely because complex interactions between nerves, muscles, sclerites and wing foils are required to accommodate flight4. Here we show that stick insects (order Phasmatodea) diversified as wingless insects and that wings were derived secondarily, perhaps on many occasions. These results suggest that wing developmental pathways are conserved in wingless phasmids, and that ‘re-evolution’ of wings has had an unrecognized role in insect diversification.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hennig, W. Insect Phylogeny (Academic, New York, 1981)
Roff, D. A. The evolution of flightlessness: is history important? Evol. Ecol. 8, 639–657 (1994)
Wagner, D. L. & Liebherr, J. K. Flightlessness in insects. Trends Ecol. Evol. 7, 216–220 (1992)
Dudley, R. The Biomechanics of Insect Flight (Princeton Univ. Press, Princeton, New Jersey, 2000)
Tilgner, E. H., Kiselyova, T. G. & McHugh, J. V. A morphological study of Timema cristinae Vickery with implications for the phylogenetics of Phasmida. Dtsch. Entomol. Z. 46, 149–162 (1999)
Kristensen, N. P. in The insects of Australia: A textbook for Students and Research Workers, 2nd edn (eds Naumann, I. D. et al.) 125–140 (CSIRO, Melbourne Univ. Press, Melbourne, 1991)
Hennig, W. Die Stammesgeschichte der Insekten (Krammer, Frankfurt am Main, Germany, 1969)
Wheeler, W. C., Whiting, M. F., Wheeler, Q. D. & Carpenter, J. M. The phylogeny of the extant hexapod orders. Cladistics 17, 113–169 (2001)
Kamp, J. W. Numerical classification of the orthopteroids, with special reference to the Grylloblattodea. Can. Entomol. 105, 1235–1249 (1973)
Beutel, R. G. & Gorb, S. N. Ultrastructure of attachment specializations of Hexapods (Arthropoda): evolutionary patterns inferred from a revised ordinal phylogeny. J. Zool Syst. Evol. Res. 39, 177–207 (2001)
Whiting, M. F., Carpenter, J. C., Wheeler, Q. D. & Wheeler, W. C. The Strepsiptera problem: phylogeny of the holometabolous insect orders inferred from 18S and 28S ribosomal DNA sequences and morphology. Syst. Biol. 46, 1–68 (1997)
Günther, K. Über die taxonomische Gliederung und geographische Verbreitung der Insektenordnung der Phasmatodea. Beitr. Ent. 3, 541–563 (1953)
Bradler, S. The Australian stick insects—a monophyletic group within the Phasmatodea? Zoology 104, 69 (2001)
Pagel, M. The maximum likelihood approach to reconstructing ancestral characters states of discrete characters on phylogenies. Syst. Biol. 48, 612–622 (1999)
Kim, J. et al. Integration of positional information and identity by Drosophila vestigial gene. Nature 382, 133–138 (1996)
Cohen, B., Simcox, A. A. & Cohen, S. M. Allocation of the thoracic imaginal primordia in the Drosophila embryo. Development 117, 597–608 (1993)
Kutsch, W. & Kittman, R. Flight motor pattern in flying and non-flying Phasmida. J. Comp. Physiol. 168, 483–490 (1991)
Halder, G., Callaerts, P. & Gehring, W. J. Induction of ectopic eyes by targeted expression of the eyeless gene in Drosophila. Science 167, 1788–1792 (1995)
Anderson, N. M. in The Origin of Biodiversity in Insects: Phylogenetic Tests of Evolutionary Scenarios (ed. Grandcolas, P.) 91–108, (Mem. Mus. Natl Hist. Nat., Paris, 1997)
Colgan, D. J. et al. Histone H3 and U2 snRNA DNA sequences and arthropod molecular evolution. Aust. J. Zool. 46, 419–437 (1998)
Whiting, M. F. Mecoptera is paraphyletic: multiple genes and phylogeny of Mecoptera and Siphonaptera. Zoologica Scripta 31, 93–104 (2002)
Whiting, M. F. in Molecular Systematics and Evolution: Theory and Practice (eds DeSalle, R., Wheeler, W. C. & Giribet, G.) 69–84 (Birkhauser, Basel, 2002)
Wheeler, W. Fixed character states and the optimization of molecular sequence data. Cladistics 15, 379–386 (1999)
Swofford, D. L. PAUP*: Phylogenetic analysis using parsimony (*and other methods) (version 4.0b10) (Sinauer, Sunderland, Massachusetts, 2000)
Sorenson, M. D. TreeRot (version 2) (Boston Univ. Press, Massachusetts, 1999)
Farris, J. S., Kallersjo, M., Kluge, A. G. & Bult, C. Testing significance of incongruence. Cladistics 10, 315–320 (1994)
Maddison, D. R. & Maddison, W. P. MacClade 4: Analysis of phylogeny and character evolution (version 4) (Sinauer, Sunderland, Massachusetts, 2000)
Pagel, M. Discrete (Univ. Reading Press, Reading, UK, 1999)
Lutzoni, F., Pagel, M. & Reeb, V. Major fungal lineages are derived from lichen symbiotic ancestors. Nature 411, 937–940 (2001)
Acknowledgements
We thank E. Tilgner for providing some specimens; A. Whiting and L. Harmon for assistance in data analysis; and J. Cherry, M. Gruwell, M. Terry, H. Ogden, J. Robertson and K. Jarvis for generating sequence data. Analyses were performed in the Fulton Supercomputer Center, Brigham Young University, and parallel software implementation was performed by M. Clement and Q. Snell. DNA sequences are deposited in GenBank under accession numbers AY121129–AY121186 and AY125216–AY125326.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
41586_2003_BFnature01313_MOESM1_ESM.pdf
Supplementary Figure 1: This is the ingroup portion of the Bayesian tree, with posterior probabilities given above nodes, and the probability that the ancestral state was wingless given below the node. This reconstruction requires 5 independent wing gains and 2 wing losses. (PDF 60 kb)
41586_2003_BFnature01313_MOESM2_ESM.doc
Supplementary Information: This file provides additional details concerning taxon selection, optimization alignment methodology, incongruence length difference metrics, parsimony tree reconstruction, likelihood tree reconstruction, Bayesian analysis, congruence of molecular results with known morphological characters, parsimony character mapping, and likelihood character mapping. (DOC 168 kb)
Rights and permissions
About this article
Cite this article
Whiting, M., Bradler, S. & Maxwell, T. Loss and recovery of wings in stick insects. Nature 421, 264–267 (2003). https://doi.org/10.1038/nature01313
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01313
This article is cited by
-
Independent wing reductions and losses among stick and leaf insects (Phasmatodea), supported by new Cretaceous fossils in amber
BMC Biology (2023)
-
Testing alternative hypotheses on the origin and speciation of Hawaiian katydids
BMC Ecology and Evolution (2022)
-
The transcription factor Zfh1 acts as a wing-morph switch in planthoppers
Nature Communications (2022)
-
De novo transcriptome analysis for examination of the nutrition metabolic system related to the evolutionary process through which stick insects gain the ability of flight (Phasmatodea)
BMC Research Notes (2021)
-
Rapid parallel adaptation despite gene flow in silent crickets
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.