Abstract
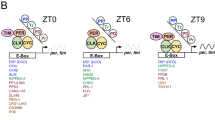
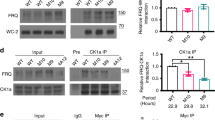
The Drosophila circadian clock is driven by daily fluctuations of the proteins Period and Timeless, which associate in a complex and negatively regulate the transcription of their own genes1,2. Protein phosphorylation has a central role in this feedback loop, by controlling Per stability in both cytoplasmic and nuclear compartments3,4,5,6 as well as Per/Tim nuclear transfer7,8. However, the pathways regulating degradation of phosphorylated Per and Tim are unknown. Here we show that the product of the slimb (slmb) gene9—a member of the F-box/WD40 protein family of the ubiquitin ligase SCF complex that targets phosphorylated proteins for degradation10,11,12,13—is an essential component of the Drosophila circadian clock. slmb mutants are behaviourally arrhythmic, and can be rescued by targeted expression of Slmb in the clock neurons. In constant darkness, highly phosphorylated forms of the Per and Tim proteins are constitutively present in the mutants, indicating that the control of their cyclic degradation is impaired. Because levels of Per and Tim oscillate in slmb mutants maintained in light:dark conditions, light- and clock-controlled degradation of Per and Tim do not rely on the same mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Allada, R., Emery, P., Takahashi, J. S. & Rosbash, M. Stopping time: the genetics of fly and mouse circadian clocks. Annu. Rev. Neurosci. 24, 1091–1119 (2001)
Young, M. W. & Kay, S. A. Time zones: a comparative genetics of circadian clocks. Nature Rev. Genet. 2, 702–715 (2001)
Kloss, B. et al. The Drosophila clock gene double-time encodes a protein closely related to human casein kinase Iε. Cell 94, 97–107 (1998)
Price, J. L. et al. double-time is a novel Drosophila clock gene that regulates PERIOD protein accumulation. Cell 94, 83–95 (1998)
Suri, V., Hall, J. C. & Rosbash, M. Two novel doubletime mutants alter circadian properties and eliminate the delay between RNA and protein in Drosophila. J. Neurosci. 20, 7547–7555 (2000)
Kloss, B., Rothenfluh, A., Young, M. W. & Saez, L. Phosphorylation of period is influenced by cycling physical associations of double-time, period, and timeless in the Drosophila clock. Neuron 30, 699–706 (2001)
Martinek, S., Inonog, S., Manoukian, A. S. & Young, M. W. A Role for the segment polarity gene shaggy/GSK-3 in the Drosophila circadian clock. Cell 105, 769–779 (2001)
Curtin, K. D., Huang, Z. J. & Rosbash, M. Temporally regulated nuclear entry of the Drosophila period protein contributes to the circadian clock. Neuron 14, 365–372 (1995)
Jiang, J. & Struhl, G. Regulation of the Hedgehog and Wingless signalling pathways by the F-box/WD40-repeat protein Slimb. Nature 391, 493–496 (1998)
Skowyra, D., Craig, K. L., Tyres, M., Elledge, S. J. & Harper, J. W. F-box proteins are receptors that recruit phosphorylated substrates to the SCF ubiquitin-ligase complex. Cell 91, 209–219 (1997)
Margottin, F. et al. A novel human WD protein, h-β TrCp, that interacts with HIV-1 Vpu connects CD4 to the ER degradation pathway through an F-box motif. Mol. Cell 1, 565–574 (1998)
Spencer, E., Jiang, J. & Chen, Z. J. Signal-induced ubiquitination of IκBα by the F-box protein Slimb/β-TrCP. Genes Dev. 13, 284–294 (1999)
Winston, J. T. et al. The SCFβ-TRCP-ubiquitin ligase complex associates specifically with phosphorylated destruction motifs in IκBα and β-catenin and stimulates IκBα ubiquitination in vitro. Genes Dev. 13, 270–283 (1999)
Miletich, I. & Limbourg-Bouchon, B. Drosophila null slimb clones transiently deregulate Hedgehog-independent transcription of wingless in all limb discs, and induce decapentaplegic transcription linked to imaginal disc regeneration. Mech. Dev. 93, 15–26 (2000)
Renn, S. C., Park, J. H., Rosbash, M., Hall, J. C. & Taghert, P. H. A pdf neuropeptide gene mutation and ablation of PDF neurons each cause severe abnormalities of behavioral circadian rhythms in Drosophila. Cell 99, 791–802 (1999)
Blanchardon, E. et al. Defining the role of Drosophila lateral neurons in the control of circadian activity and eclosion rhythms by targeted genetic ablation and PERIOD protein overexpression. Eur. J. Neurosci. 13, 871–888 (2001)
Edery, I., Zwiebel, L. J., Dembinska, M. E. & Rosbash, M. Temporal phosphorylation of the Drosophila period protein. Proc. Natl Acad. Sci. USA 91, 2260–2264 (1994)
Myers, M. P., Wager-Smith, K., Rothenfluh-Hilfiker, A. & Young, M. W. Light-induced degradation of TIMELESS and entrainment of the Drosophila circadian clock. Science 271, 1736–1740 (1996)
Zeng, H. K., Qian, Z. W., Myers, M. P. & Rosbash, M. A light-entrainment mechanism for the Drosophila circadian clock. Nature 380, 129–135 (1996)
Kaneko, M., Park, J. H., Cheng, Y., Hardin, P. E. & Hall, J. C. Disruption of synaptic transmission or clock-gene-product oscillations in circadian pacemaker cells of Drosophila cause abnormal behavioral rhythms. J. Neurobiol. 43, 207–233 (2000)
Yang, Z. & Sehgal, A. Role of molecular oscillations in generating behavioral rhythms in Drosophila. Neuron 29, 453–467 (2001)
Hunter-Ensor, M., Ousley, A. & Sehgal, A. Regulation of the Drosophila protein timeless suggests a mechanism for resetting the circadian clock by light. Cell 84, 677–685 (1996)
Lee, C. G., Parikh, V., Itsukaichi, T., Bae, K. & Edery, I. Resetting the Drosophila clock by photic regulation of PER and a PER-TIM complex. Science 271, 1740–1744 (1996)
Naidoo, N., Song, W., Hunter-Ensor, M. & Sehgal, A. A role for the proteasome in the light response of the Timeless clock protein. Science 285, 1737–1741 (1999)
Price, M. A. & Kalderon, D. Proteolysis of the Hedgehog signaling effector Cubitus interruptus requires phosphorylation by glycogen synthase kinase 3 and casein kinase 1. Cell 108, 823–835 (2002)
Jia, J. et al. Shaggy/GSK3 antagonizes Hedgehog signalling by regulating Cubitus interruptus. Nature 416, 548–552 (2002)
Pai, L. M., Orsulic, S., Bejsovec, A. & Peifer, M. Negative regulation of Armadillo, a Wingless effector in Drosophila. Development 124, 2255–2266 (1997)
Stanewsky, R. et al. Temporal and spatial expression patterns of transgenes containing increasing amounts of the Drosophila clock gene period and a lacZ reporter: Mapping elements of the PER protein involved in circadian cycling. J. Neurosci. 17, 676–696 (1997)
Ruel, L., Pantesco, V., Lutz, Y., Simpson, P. & Bourouis, M. Functional significance of a family of protein kinases encoded at the shaggy locus in Drosophila. EMBO J. 12, 1657–1669 (1993)
Cegielska, A., Gietzen, K. F., Rivers, A. & Virshup, D. M. Autoinhibition of casein kinase I ε (CKI ε) is relieved by protein phosphatases and limited proteolysis. J. Biol. Chem. 273, 1357–1364 (1998)
Acknowledgements
We thank M. Boudinot for the Faas software, M. Serrier and L. Collet for help with the figures, M. Rosbash, P. Emery, A. Klarsfeld, J.-F. Julien and E. Petrochilo for their comments and suggestions on the manuscript, as well as J. Champagnat and J.-D. Vincent for their continuous support. We thank I. Miletich for the unpublished UAS-slmb line, and R. Myers, L. Saez, R. Stanewsky and D. Virshup for providing antibodies or constructs. This work was supported by CNRS (ATIPE “Développement” and appel d'offres “Biologie cellulaire”) and Fondation pour la Recherche Médicale. F.R. is supported by INSERM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Grima, B., Lamouroux, A., Chélot, E. et al. The F-box protein Slimb controls the levels of clock proteins Period and Timeless. Nature 420, 178–182 (2002). https://doi.org/10.1038/nature01122
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01122
This article is cited by
-
Molecular mechanisms and physiological importance of circadian rhythms
Nature Reviews Molecular Cell Biology (2020)
-
Proteomic analysis of Drosophila CLOCK complexes identifies rhythmic interactions with SAGA and Tip60 complex component NIPPED-A
Scientific Reports (2020)
-
Effects of MUL1 and PARKIN on the circadian clock, brain and behaviour in Drosophila Parkinson’s disease models
BMC Neuroscience (2019)
-
Molecular modulators of the circadian clock: lessons from flies and mice
Cellular and Molecular Life Sciences (2017)
-
Chickpea transcription factor CaTLP1 interacts with protein kinases, modulates ROS accumulation and promotes ABA-mediated stomatal closure
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.